Institute of Oceanology, Chinese Academy of Sciences
Article Information
- YIN Jiehui(尹洁慧), ZHANG Guangtao(张光涛), LI Chaolun(李超伦), WANG Shiwei(王世伟), ZHAO Zengxia(赵增霞), WAN Aiyong(万艾勇)
- Community composition, abundance and biomass of zooplankton in Zhangzi Island waters, Northern Yellow Sea
- Chinese Journal of Oceanology and Limnology, 35(5): 1144-1151
- http://dx.doi.org/10.1007/s00343-017-6135-0
Article History
- Received May. 4, 2016
- accepted in principle Jun. 27, 2016
- accepted for publication Aug. 3, 2016
2 Jiaozhou Bay Marine Ecosystem Research Station, Institute of Oceanology, Chinese Academy of Sciences, Qingdao 266071, China;
3 Key Laboratory of Marine Ecology and Environmental Sciences, Institute of Oceanology, Chinese Academy of Sciences, Qingdao 266071, China
Zhangzi Island is one of the most important aquaculture bases in northern China, located in the south of the Changshan Archipelago (CA), 50 km from the coastline in the northern Yellow Sea. Since 1998 the CA area has been an important mariculture area for the Japanese scallop Patinopecten yessoensis. Off Zhangzi Island, waters shallower than 50 m have been used for bottom-sowing culture. A recent study suggested that chlorophyll a (Chl a) concentration might be depressed because of dense bivalve culture (Zhang et al., 2008). The cascade effects on zooplankton have not been investigated until now.
The zooplankton community in the Zhangzi Island area may be influenced by various physical events in addition to mariculture activities. First, the Yellow Sea Cold Bottom Water (YSCBW) is the most important physical character in summer. Its distribution, defined with a 12.5℃ isothermal line at 25 m, covers all Yellow Sea areas below this depth (Song et al., 2009). The strength of the YSCBW was observed correlated with survivorship of cultured scallop (Du et al., 1996). As an over-summering cites, the YSCBW was suggested to be the most important factor influencing zooplankton distribution in the Southern Yellow Sea in summer (Sun et al., 2002). Second, the Yellow Sea Warm Current (YSWC), which flows into the Yellow Sea from the south of Cheju Island, can reach as far as the Bohai Strait (Bao et al., 2009). It has been established that zooplankton assemblage in the southern Yellow Sea can be influenced significantly by the YSWC (Wang and Zuo, 2004). Third, rivers in northern China undergo severe seasonal fluctuation in freshwater discharge, with frequent episodic flood events in summer. During the flood season, surface dilution can occupy a large area in the northern Yellow Sea and exert obvious negative influences both on mariculture and on planktonic species.
Despite the environmental complexity, data on annual zooplankton variation are limited, and we found no annual investigation since the Census of Marine Life in 1958. In present study, an area extending from the mariculture region to the YSCBW areas was covered, in order to understand zooplankton species composition and the relationship between environmental factors and zooplankton abundance.
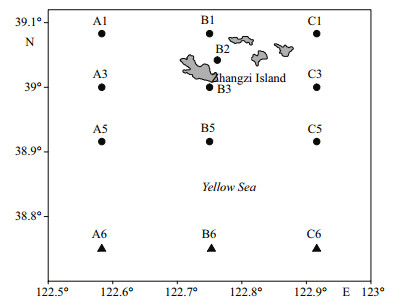
2 MATERIAL AND METHOD 2.1 Sample collectionSamples were collected monthly at the same set of stations (Fig. 1) in Zhangzi Island waters from July 2009 to June 2010, using a conical plankton net (mouth diameter: 0.8 m; mesh size: 500 μm) towed vertically from bottom to surface. The samples were fixed with 5% formalin solution, and then counted under a dissecting microscope. Temperature and salinity were measured with AAQ1183-1F CTD (Alec Electronics Co., Japan). To measure total Chl a concentrations, 500 mL natural seawater was filtered through 0.45 μm cellulose acetate (CA). The membranes were extracted from 90% acetone (v/v) in a refrigerator for 24 h (≤0℃), and then measured with a Turner Designs Model 7200 fluorometer.
|
| Figure 1 Sample sites of cruises from 2009 to 2010 off Zhangzi Island Black dots: sites in mariculture area; black triangle: sites in open water |
Thirteen stations were divided into two groups according to geographic differences. Sites A6, B6, and C6 were located in the YSCBW area away from the nearshore (referred to here as 'open water stations'); the remainder were in the bottom seeding culture area (the 'mariculture area').
The dominance index (Y) was calculated according to the following equation (Dufrêne and Legendre 1997):
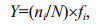 (1)
(1)where ni represented the abundance of species i, N was the total abundance of all species, fi was the appearance frequency of species i and Y>0.02 was adapted as the criterion for a dominant species.
To measure biomass, zooplankton samples were filtered through bolting silk and jellyfish were separated as these contain considerable moisture and impurities. After vacuum filtering and weighing on an electronic balance, according to the filtrate volume, translate them to wet weight (mg/m3).
Biodiversity analysis used PRIMER V5.2 (Plymouth Routines In Multivariate Ecological Research) which was exploited by Plymouth Marine Laboratory (PML).
The Shannon-Weaver Index (H′) species diversity index (Shannon and Weaver, 1949) provided community richness and uniformity information, using the equation:
 (2)
(2)where Pi represented the proportion of species i in the samples. For example, if the total number of the sample was N, the individual number of species i was ni, so Pi=ni/N.
Margalef's index (d) was used to provide information on species numbers and abundance. The equation was:
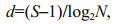 (3)
(3)where S represented the number of species in samples and N was the abundance of zooplankton.
Pielou's evenness index (J) was used:
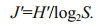 (4)
(4)The sample stations map was generated in Golden Software surfer 9.0. The difference in zooplankton abundance in different areas was evaluated statistically using Independant samples t-test with SPSS V19.0.
3 RESULT 3.1 Environmental conditionsAnnual average surface sea temperature (SST) ranged from 5.29℃ to 23.09℃ (Fig. 2), with the highest of 26.13℃ observed in August. The bottom sea temperature (BST) varied between 2.22℃ and 15.7℃. According to surface and bottom temperatures, thermal stratification existed between July and September, with the largest thermal difference in July. The SST and BST difference decreased as the YSCBW disappeared.
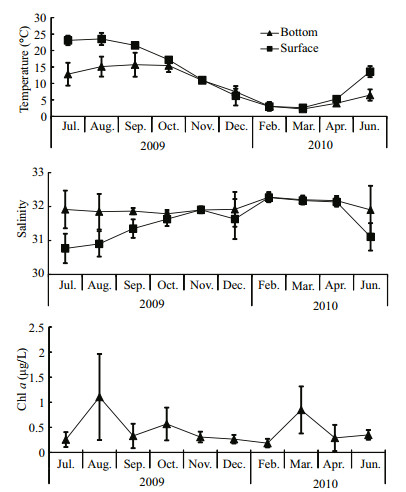
|
| Figure 2 Monthly variations in temperature, salinity and Chl a concentration in Zhangzi Island waters |
Annual average surface salinity ranged from 30.77 to 32.26. Obvious dilution by fresh water was observed in June, July and August with salinities of 31.10, 30.77 and 30.90, respectively. The average bottom salinity was less variable, ranged from 31.79 to 32.28.
Chl a concentration ranged between 0.26 and 0.57 mg/L, with the exception of an average of 1.10 mg/L in August and an average of 0.84 mg/L in March. The highest Chl a concentration value 7.32 mg/L, was recorded in August at B2 station
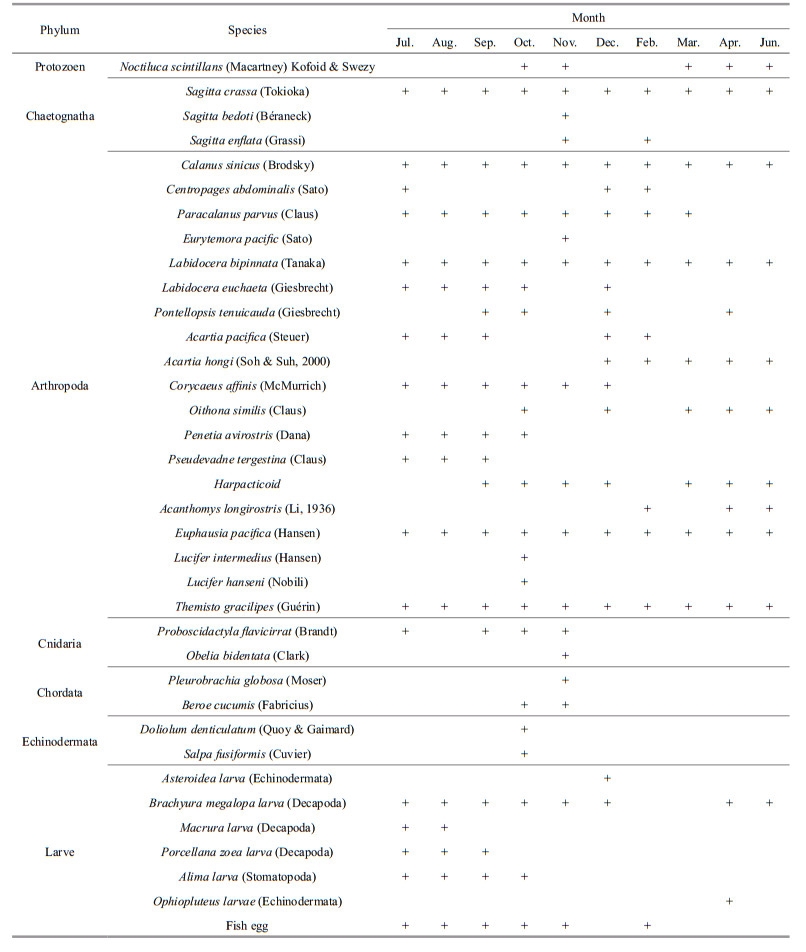
3.2 Zooplankton compositionWe identified 36 zooplankton species in Zhangzi Island waters, including the larval stages of different groups (Table 1). Most were Arthropods (19 species), which formed 52.7% of the total number, followed by Chaetognatha, represented by three species. Cnidaria, Ctenophora and Chordata were each represented by two species. Protozoa were represented by only one species.
Calanus sinicus, Saggitta crassa, Labidocera bipinnata, Themisto gracilipes and Euphausia pacifica were encountered as perennials throughout the whole year. Cladocera with a wide range of tolerance to salinity and temperature, such as Penetia avirostris and Pseudevadne tergestina, mainly appeared from July to October. In general, more warm oceanic species such as Sagitta bedoti, S. enflata, Lucifer intermedius, Eurytemora pacific and Doliolum denticulatum were only recorded in October and November. Jellyfish, including Proboscidactyla flavicirrata, Obelia bidentata, Pleurobrachia globosa and Beroe cucumis, Pontellopsis tenuicauda, Centropages abdominalis, Labidocera euchaeta and Corycaeus affinis appeared only in July, September, October and November. Harpacticoid copepod species and fish eggs were also found. Warm oceanic species in the strict sense can hardly be distinguished.
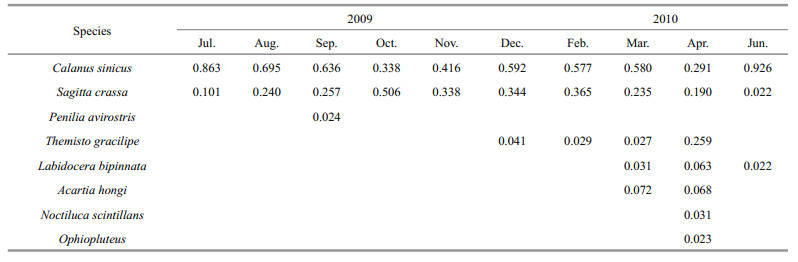
3.3 Dominant zooplankton speciesCalanus sinicus and Sagitta crassa were the dominant species throughout the survey (Table 2), and C. sinicus was the most abundant species off Zhangzi Island. The degrees of preponderance reached 0.926, 0.863 and 0.636 in June, July and September, respectively. Sagitta crassa had a degree of preponderance degree as high as 0.506 and 0.365 in October and February, respectively. Themisto gracilipe was the dominant species in winter and spring, and its highest degree of preponderance was 0.259. Labidocera bipinnata, Acartia hongi, Noctiluca scintillans and Ophiopluteus spp. larvae were the dominant species in spring, with highest degrees of preponderance for each species being 0.063, 0.072, 0.031 and 0.023, respectively. Penilia avirostris, which prefers low salinity, was the dominant species only in September, with a degree of preponderance of 0.024.
Zooplankton biomass and abundance varied seasonally in Zhangzi Island waters. Two peaks of non-jellyfish zooplankton biomass were observed (Fig. 3), the major one being in June (496.1 mg/m3) and the minor one in December (366.77 mg/m3). The highest zooplankton abundance was recorded in June (531.6 ind./m3). Both lowest zooplankton biomass (95.44 mg/m3) and abundance (38.19 ind./m3) occurred in August. The annual averages of zooplankton biomass and abundance were 217.5 mg/m3 and 131.3 ind./m3, respectively.
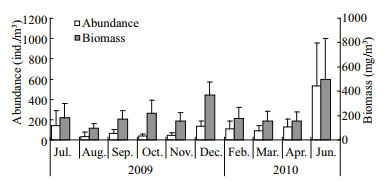
|
| Figure 3 Monthly abundance and biomass of zooplankton in Zhangzi Island waters |
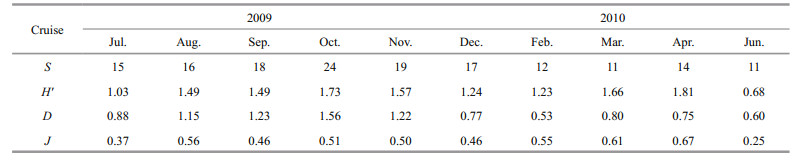
Not only can zooplankton diversity reveal the characteristics of the zooplankton community, but it can also reveal habitat traits. The average species diversity indices (H′), richness (d) and evenness (J) were 1.39, 0.95 and 0.49, respectively (Table 3). The species diversity was identical with evenness; species number was in conformity with richness.
The peak value of species number appeared in October, with a high richness index of 1.56. During this period, the intensity of the YSCBW receded, and some warm oceanic species, such as jellyfish and tunicates, appeared. The high value of species diversity and evenness indices emerged in April, with values as high as 1.81 and 0.67, respectively.
4 DISCUSSION 4.1 Effect of environmental factors on zooplankton compositionThe investigation found that constituents of the zooplankton were clearly different from the nearshore of Xiao Changshan Island (Wang, 2003), maybe the sample stations we set with deep water had long distance from the off-shore. Low-temperature highsalinity species such as Calanus sinicus dominated our whole survey. Some neritic low-salinity species such as Penetia avirostris, Labidocera bipinnata and Acartia hongi emerged as dominant species only in summer and early spring.
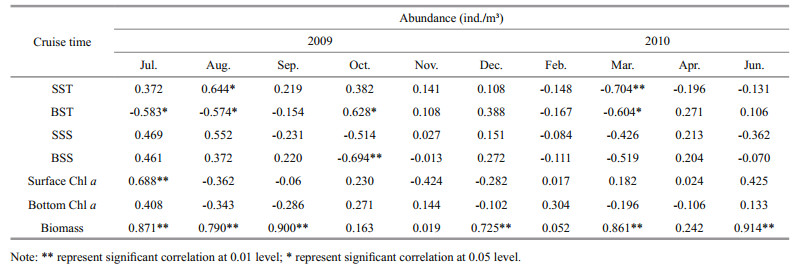
In summer (July to September 2009), seasonal rivers such as the Yalu River were in a period of high flow (Gao et al., 2003). With the freshwater input to the ocean, significant salinity decrease was detected; the dominance of cladocera more suited to a low salinity environment indicated that river discharge may influence zooplankton assemblage in this area. In winter, YSWC can reach as far as the Bohai Strait (Bao et al., 2009), and may bring tropical species with the warm current. In our study, with the disappearance of the YSCBW, some warm-ocean species, Sagitta bedoti, S. enflata and Lucifer intermedius, appeared in October and November, 2009. Positive and significant correlation was found between sea bottom temperature and zooplankton abundance in October (Table 4), but tropical species with obvious characteristic in strict sense can hardly be distinguished; perhaps the intensity of the warm current was not strong enough to bring the tropical species to the northern Yellow Sea. In early spring (March and April 2010), Labidocera bipinnata and Acartia hongi appeared as dominant species off Zhangzi Island. Labidocera bipinnata is a typical species of neritic low salinity (Qi, 2008). Recent study suggested that A. hongi was a dominant species in coastal waters of the Yellow Sea that were characterized by low salinity, low temperature and high eutrophication (Shim and Choi, 1996; Youn and Choi, 2003). The abundance increased from February to April, with a peak value in May in Kyeonggi Bay (Youn and Choi, 2007). In our study, L. bipinnata appeared throughout the year, but as a dominant species only in spring. Acartia hongi appeared in winter and spring, but as a dominant species only in spring. This trend was similar to previous studies.
|
In summer, the YSCBW played an important part in zooplankton distribution. Throughout the survey, zooplankton abundance in the open water area was obviously lower than in the mariculture area except in July and August (P < 0.05) (Fig. 4), when YSCBW exist with high intension. The abundance of Calanus sinicus and Themisto gracilipe in the mariculture area was lower than in the open water area. Calanus sinicus occupied 85.5% and 70.0% of total zooplankton abundance in July and August, respectively. The abundance of C. sinicus was 54.85, 0.79 ind./m3 in July and August in the mariculture area; and 341.51, 82.87 ind./m3 in July and August in the open water area. In the southern Yellow Sea C. sinicus was found to be concentrated inside the YSCBW, using this strategy to avoid high-temperature damage to benefit the population (Zhang, 2003). In the northern Yellow Sea. YSCBW played similar role as over-summering city T. gracilipe was a typical species in the YSCBW, its distribution being consistent with the depth of the YSCBW (Zhu, 2008). In our study, the abundance of T. gracilipe was 0.57, 0.27 ind./m3 in the mariculture area; and 3.42, 1.49 ind./m3 in the open water area in July and August. Zooplankton abundance in July and August had significantly negative correlation with sea bottom temperature (Table 4).

|
| Figure 4 Annual zooplankton abundance in mariculture and open water areas off Zhangzi Island * represent significant difference at 0.05 level. |
Zooplankton is preyed on by jellyfish, and a bloom of Beroe may decrease the abundance of other zooplankton species. The number of jellyfish was low in July and September. A bloom of the ctenophore Beroe was observed in October and November (Fig. 5), with an average biomass of 38.91 mg/m3 and 216.14 mg/m3, respectively.
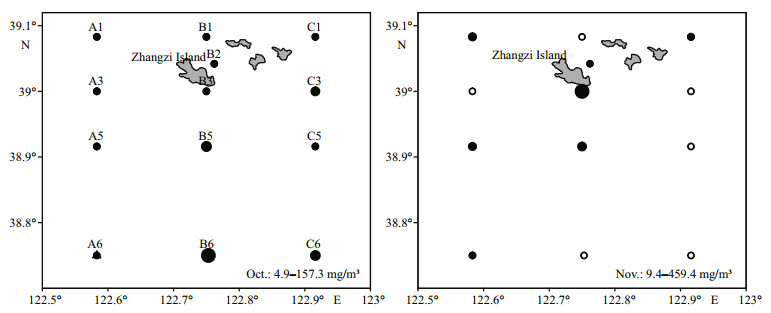
|
| Figure 5 Geographical biomass of jellyfish in Zhangzi Island waters in October and November Open circles represent stations at which no jellyfish were found, and sizes of the filled circles are in proportion to the biomass of jellyfish (mg/m3). |
Copepods formed a large proportion of the zooplankton, ranging from 34% to 92% over the whole year. Copepod abundance ranged from 14.36 ind./m3 to 497.04 ind./m3 over the whole year. In October and November, the abundance was 14.36 ind./m3 and 16.38 ind./m3, respectively, lower than the abundance in other months. The influence of jellyfish on the Chaetognatha, Noctiluca scintillans and larve was not obvious (Fig. 6).

|
| Figure 6 Copepods Chaetognatha, Noctiluca scintillans and larve abundance in Zhangzi Island waters |
According to our results, significant negative correlation was observed between jellyfish and zooplankton abundance by geographical distribution in October. In November, no relationship was found: jellyfish biomass was as high as 383.2 mg in station C1, but the abundance of zooplankton was not low. This illustrated that jellyfish influenced zooplankton abundance over a long time period.
4.4 Zooplankton abundance and biomass variationsDuring this investigation, zooplankton abundance and biomass in summer were higher than in winter (Fig. 3). The seasonal variable trend was in line with C. sinicus, which greatly influenced the total abundance. With the input runoff from the Yalu River, some low-salinity neritic species, such as Penetia avirostris and Pseudevadne tergestina, moved into offshore station, enhancing species abundance. The dilution caused by the freshwater and a short period of rainfall resulted in rapid phytoplankton growth. In July, positive and significant correlation was found between Chl a concentration and zooplankton abundance and biomass (Table 4). Chl a concentration was an important factor in regulating zooplankton biomass in summer.
4.5 Long-term comparisonThe first annual investigation of zooplankton in the northern Yellow Sea was the marine census of 1959. At this time the dominant species were Calanus sinicus, Sagitta crassa, Themisto gracilipe, Labidocera bipinnata and Centropages abdominal. Although the main dominant species didin't change, some low salinity neritic species such as Penetia avirostris and Pseudevadne tergestina didn't appear in the whole year. Acartia spp. were not dominant in summer, although numbers were higher in the northern Yellow Sea than off Zhangzi Island. During a 2006 investigation, zooplankton abundance was 266.3 ind./m3 in winter higher than in Zhangzi Island (133.1 ind./m3), and 525.1 ind./m3 in summer, lower than off Zhangzi Island (531.6 ind./m3) (Zhu, 2008). The species diversity index in the northern Yellow Sea was higher than off Zhangzi Island both in winter and in summer, over the entire area investigated.
5 CONCLUSIONCalanus sinicus and Sagitta crassa were dominant species off Zhangzi Island. No tropical species were brought by the YSWC. In summer, the YSCBW played an important part in protecting the C. sinicus population in the northern Yellow Sea. Jellyfish may decrease zooplankton abundance. We found no direct significant influence from mariculture on the zooplankton community. Annual average zooplankton biomass and abundance were 217.5 mg/m3 and 131.3 ind./m3. The average species diversity index (H′), richness index (d) and evenness index (J) were 1.39, 0.95 and 0.49, respectively. The highest species diversity (1.81) and evenness index (0.67) emerged in April. The richness index was highest in October (1.56).
6 ACKNOWLEDGEMENTWe thank Mr. ZANG Y. C., and the crew of the research vessel of the Dalian Zhangzidao Fishery Group Co. Ltd., for their support with field sampling. We also thank the Open Cruise of Chinese Offshore Oceanography Research by Institute of Oceanology, Chinese Academy of Science for giving us the sampling opportunity in December 2010.
| Bao X W, Li N, Yao Z G, Wu D X, 2009. Seasonal variation characteristics of temperature and salinity of the north Yellow Sea. Periodical of Ocean University of China, 39(4): 553–562. |
| Du B, Zhang Y J, Shan Y C, 1996. The characteristics of cold water mass variation at the bottom of the Northern Yellow sea and its hydrological effects on the mortality of shellfish cultured in the waters of outer Chang-shan Islands. Marine Science Bulletin, 15(4): 17–28. |
| Dufrêne M, Legendre P, 1997. Species assemblages and indicator species:the need for a flexible asymmetrical approach. Ecol. Monogr., 67(3): 345–366. |
| Gao J H, Gao S, Cheng Y, Dong L X, Zhang J, 2003. Sediment transport in Yalu River Estuary. Chinese Geographical Science, 13(2): 157–163. Doi: 10.1007/s11769-003-0010-y |
| Qi Y P. 2008. Study on the community ecology of zooplankton of in the Yellow Sea and East China Sea in summer and winter. Ocean University of China, Qingdao, China. (in Chinese) |
| Shannon C E, Weaver W. 1949. The Mathematical Theory of Communication. The University of Illinois Press, Urbana, IL. 125p. |
| Shim M B, Choi J K, 1996. A study on the seasonal succession of copepod community in Kyeonggi Bay, Yellow Sea through the short interval surveys. J. Int. Soc. Yellow Sea Res., 2: 65–73. |
| Song X, Lin X P, Wang Y, 2009. The preliminary study of long-term variability of the Yellow sea cold water in summer and its possible reasons. Journal of Guangdong Ocean University, 29(3): 59–63. |
| Sun S, Wang R, Zhang G T, Yang B, Ji P, Zhang F, 2002. A Preliminary study on the over-summer strategy of Calanus sinicus in the Yellow Sea. Oceanologia et Limnologia Sinica(S): 92–100. |
| Wang R, Zuo T, 2004. The Yellow Sea warm current and the Yellow Sea cold bottom water, their impact on the distribution of zooplankton in the Southern Yellow Sea. J. Soc. Oceanogr. Korean, 39(1): 1–13. |
| Wang Z L, 2003. Preliminary studies on zooplankton community in waters around Xiao Changshan Island. Journal of DaLian Fisheries University, 18(4): 296–300. |
| Youn S H, Choi J K, 2003. Seasonal changes in zooplankton community in the coastal waters off Incheon. Ocean Science Journal, 38(3): 111–121. |
| Youn S H, Choi J K, 2007. Egg production of the copepod Acartia hongi in Kyeonggi Bay. Journal of Marine Systems, 67(3-4): 217–224. Doi: 10.1016/j.jmarsys.2006.05.017 |
| Zhang G T. 2003. Reproductive strategy of Calanus sinicus in the Southern Yellow Sea. The Institute of Oceanology, Chinese Academy of Sciences, Qingdao, China. (in Chinese) |
| Zhang J H, Fang J G, Jiang Z J, Wang W, Wang S H, Sun S, 2008. Seasonal variation of primary production and spatial-temporal distribution of chlorophyll a in mariculture area of Zhangzidao Island. Marine Fisheries Research, 29(4): 22–28. |
| Zhu Y Z. 2008. Study on the Macro-zooplankton and Mesozooplankton Community Ecology in the North Yellow Sea in Summer and Winter. Ocean University of China, Qingdao, China. (in Chinese) |
 2017, Vol. 35
2017, Vol. 35