Institute of Oceanology, Chinese Academy of Sciences
Article Information
- KONG Ning(孔宁), LIU Xiao(刘晓), LI Junyuan(李俊元), MU Wendan(穆文丹), LIAN Jianwu(连建武), XUE Yanjie(薛艳洁), LI Qi(李琪)
- Effects of temperature and salinity on survival, growth and DNA methylation of juvenile Pacific abalone, Haliotis discus hannai Ino
- Chinese Journal of Oceanology and Limnology, 35(5): 1248-1258
- http://dx.doi.org/10.1007/s00343-016-5185-z
Article History
- Received Jun. 24, 2015
- accepted for publication Sep. 16, 2015
2 Ningbo University, Ningbo 315211, China;
3 University of Chinese Academy of Sciences, Beijing 100049, China;
4 Ocean University of China, Qingdao 266003, China
In China, the Pacific abalone, Haliotis discus hannai Ino, is a commercially important mollusk, whose production was around 50 000 metric tons in 2010, accounting for approximately 90% of the worldwide yield (FAO, 2012). Beginning in the early 1980s, its commercial-scale mariculture was mainly distributed in its natural habitats, the Liaodong and Shandong Peninsulas (Zhang et al., 2004). Since the 2000s, farming of H. discus hannai Ino has extended substantially from the northern Yellow Sea to the East China Sea (Li et al., 2012), which exposes the abalone to various environmental conditions. Even within the same sea area, the ambient environment can vary considerably according to the season.
In marine mollusks, temperature and salinity are regarded as the most potent environmental factors (Kinne, 1963), which not only limit their natural distribution and farming area (Allan et al., 2006; Cáceres-Puig et al., 2007), but also induce variation in survival, growth and hence overall mariculture production (Laing, 2002; Searle et al., 2006; Cáceres-Puig et al., 2007; Xue et al., 2010). Thus, it is necessary to explore the effects of temperature and salinity on abalone performance to optimize development of the industry. A number of studies have dealt with the individual effects of temperature and salinity on H. discus hannai Ino (Nie et al., 1984; Gao et al., 1990; Liu et al., 2005; Zhang et al., 2005; Wang, 2012), but their interaction, which would be more physiologically valid, has not been covered. The present study aimed to examine the individual and combined effects of temperature and salinity on the survival and growth of juvenile H. discus hannai Ino and determine the optimal rearing conditions for abalone mariculture.
Much effort has been devoted to uncovering the mechanism underlying phenotypic variation in response to environmental heterogeneity (Gao et al., 2010). Growing evidence indicates that epigenetic modification, especially DNA methylation, plays an important role in helping individuals cope with different environments (Gao et al., 2010; Lira-Medeiros et al., 2010; Tan, 2010; Morán et al., 2013). DNA methylation, predominantly occurring at the 5-position of cytosine, is crucial for epigenetic control of gene expression (Li et al., 2009). It can be altered by environmental stimuli and may contribute to phenotypic variation by regulating transcriptional opportunities of stress-responsive genes (Roberts and Gavery, 2012). Navarro-Martín et al. (2011) observed that high temperature increased the DNA methylation of the gonadal aromatase promoter, which was involved in temperature-dependent sex ratio shifts in European sea bass. Marconi et al. (2013) found that salinity could induce genome-wide changes in DNA methylation, which might result in the differential expression of stress-related genes. So far, the environment-induced methylation alteration has been well characterized in vertebrates and plants, but little is known in invertebrates, especially in H. discus hannai Ino.
Methylation-sensitive amplified polymorphism (MSAP) analysis, a modification of the amplification fragment length polymorphism (AFLP) technique, represents an efficient and cost-effective approach to profile genomic methylation patterns without requiring any genome information (Lira-Medeiros et al., 2010). MSAP is based on the use of isoschizomers HpaⅡ and MsPⅠ, which recognize the same restriction site 5'-CCGG-3' but display differential sensitivity to cytosine methylation. Recently, the MSAP technique has been applied extensively in Crassostrea gigas (Jiang et al., 2013), Chlamys farreri (Sun et al., 2014), Salmo trutta (Morán et al., 2013) and Apostichopus japonicus (Zhao et al., 2015), and has proved useful to uncover epigenetic variability in marine organisms. In this study, we first analyzed the extent and pattern of DNA methylation in abalone reared at different temperatures and salinities using the MSAP technique, expecting to uncover the epigenetic regulatory mechanism underlying the phenotypic variation and adaptive responses to thermal and salt stresses.
The results of our study may provide a physiological basis for the mariculture of juvenile H. discus hannai Ino, and also lay a foundation for epigenetic research in the abalone.
2 MATERIAL AND METHOD 2.1 Experimental animalsThe abalone used in our study were derived from a selective breeding population known as P-97, which had experienced six generations of selection for rapid growth since 1997. Three-month-old juveniles of uniform size were collected from Xunshan Fishery Company, Shandong Province, and acclimated in a closed circulating water tank (temperature: 20±0.5℃, salinity: 32±0.5) for 2 weeks. Water was totally replaced every other day and vigorous aeration was provided. The animals were fed daily with an artificial diet (250 μm sieved Gold Feed, Weihai, China). Of these, 200 randomly chosen individuals, which were not used in the following culture experiment, were measured on the day of stocking to obtain the initial values of growth-related traits. The abalone were gently dried using blotting paper before being measured. Shell length and width were measured to the nearest 0.01 mm using a vernier caliper, and the wet weight was measured to the nearest 0.01 g using an electronic balance.
2.2 Experimental systemThe individual and combined treatments of temperature and salinity were implemented simultaneously. The experiment was carried out in closed circulating water tanks, each with an effective water volume of 450 L and a turnover rate of 7 200 L/ day. The tanks acted as water baths and aeration ensured complete mixing of the water. Each tank was equipped with a thermostat-controlled immersion heater or a cooling titanium pipe to regulate the water temperature. Each tank had space for partially submerged 2-L plastic beakers, which were used to hold the abalone and ensure they were exposed to continuous aeration. An 11 cm×6 cm black corrugated sheet was placed in each beaker as substrate.
Temperature was controlled using a water bath, and salinity was adjusted by adding a commercial sea salt (Tropical Marine, Cnsic Marine Biotechnology Co., Tianjin, China) or dechlorinated tap water to ambient seawater. Temperature and salinity were adjusted daily to keep the variations within ±0.5℃ and ±0.5, respectively, using a mercury thermometer and a portable refractometer. Dissolved oxygen (5.70–7.90 mg/L) and pH (7.90–8.20) were monitored three times a week using a water-quality meter (YSI Model 556MPS, Yellow Springs Instrument, Yellow Springs, OH, USA).
2.3 Temperature and salinity treatmentsTo study the individual effect, juveniles were exposed to 16, 18, 20, 22, 24, 26, 28, 30 and 32℃ at a constant salinity of 32, and 16, 18, 20, 22, 24, 26, 28, 30, 32, 34, 36, 38, 40, 42, 44 and 46 at a constant temperature of 20℃. To study the combined effect, four temperatures (16, 20, 24 and 28℃) and five salinities (24, 28, 32, 36 and 40) were tested in a full factorial experiment.
After the acclimation, 60 abalone in triplicates (n=20/replicate) were randomly selected for each treatment. Each day at 17:00, the abalone were fed with the artificial diet (as above) to excess. The following morning, each beaker was drained, washed and refilled with appropriate temperature/salinityadjusted water. Abalone were cultured for 77 days. At harvest, the shell length, shell width and wet weight were individually measured using the abovementioned method. Specific growth rate (SGR, %/d) was calculated using the formula: SGR=100×(lnXt– lnX0)/t (the days of the experiment), with Xt and X0 as final and initial values of shell length, shell width and wet weight. Mortalities were recorded daily and used to calculate the survival rate.
2.4 DNA isolation and MSAP analysisTo study the effect of temperature and salinity on DNA methylation, we performed MSAP analysis on the abalone from selected temperature (20, 24 and 30℃) and salinity (22, 32 and 42) treatments in the single-factor experiment. Treatments of 24℃ and 32, which had the best survival and growth, were regarded as control groups, with the other treatments used as the corresponding stress groups. Sixteen individuals were sampled from each treatment, except for 30℃, which only provided seven abalone for the analysis.
Genomic DNA was extracted from the foot muscle using a modified phenol-chloroform protocol (Li et al., 2006). DNA samples were checked on 1% agarose gels and normalized to 100 ng/μL using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, USA). For each individual, 100 ng DNA were independently digested with EcoRI/HpaⅡ and EcoRI/MsPⅠ (2 U each) in a restriction buffer totaling 10 μL, and subsequently ligated with adapters. Pre-selective amplification was performed in a 10 μL reaction using 2 μL of 1:10 ligation dilution, 1×PCR buffer, 0.2 mmol/L of dNTPs, 1.5 mmol/L of MgCl2, 2.5 pmol of EcoRI-A and HpaⅡ/MsPⅠ-T pre-selective primers (Jiang et al., 2013) and 2.5 U of Taq polymerase. PCR conditions for pre-selective amplification were as follows: 72℃ for 2 min, 20 cycles of 94℃ for 20 s, 56℃ for 30 s, 72℃ for 2 min and a final step of 60℃ for 30 min. Selective amplification was performed in a 10 μL reaction using 1.5 μL of 1:20 pre-selective amplification dilution. Ten selective primer combinations were chosen to generate all the MSAP profiles. PCR contained 1×PCR buffer, 0.2 mmol/L of dNTPs, 1.5 mmol/L of MgCl2, 2.5 pmol of each selective primer and 2.5 U of Taq polymerase. Cycling conditions for selective amplification were 94℃ for 2 min, 10 cycles of 94℃ for 20 s, 66℃ (decreasing by 1℃ each cycle) for 30 s, and 72℃ for 2 min; followed by 20 cycles of 94℃ for 20 s, 56℃ for 30 s, and 72℃ for 2 min, ending with 60℃ for 30 min. PCR products were run on an ABI 3130 Genetic Analyzer (Applied Biosystems, Foster City, CA, USA) and the fragment analysis was performed using GeneMapper v.4.0 software (Applied Biosystems, Foster City, CA, USA). DNA fragments less than 50 bp in length, longer than 500 bp or less than 200 RFU (Relative Fluorescent Units) were excluded from the analysis because of low levels of reproducibility.
2.5 Data analysisAnalysis of variance (ANOVA) was used to test the individual and combined effects of temperature and salinity on the survival and growth of juvenile H. discus hannaiIno. Data were assessed for homogeneity of variance using Levene's test. When significant differences were found (P < 0.05), Student-NewmanKeuls test was used to compare means of various treatments. Response-surface contour plots were generated to graphically demonstrate the combined effect using Surfer v.8.0 software (Golden Software, Golden, CO, USA).
In the MSAP analysis, three types of bands were identified by presence (1) or absence (0) of EcoRI/HpaⅡ and EcoRI/MsPⅠ digests, respectively: (a) present in both (1-1), non-methylation; (b) present in only EcoRI/HpaⅡ (1-0, hemi-methylation) or EcoRI/MsPⅠ (0-1, full methylation), methylation; (c) absent in both (0-0), uninformative because of the absence of a fragment or hyper-methylation. The MSAP locus, at which the observed proportion of methylated scores exceeded a 5% threshold, was classified as a "methylation-susceptible locus" (MSL); an MSL was classified as a "methylationsusceptible polymorphism locus" (MSP) if the nonmethylated or missing scores exceeded a 5% threshold. The MSP binary matrix was scored as 1 (1-0 or 0-1), 0 (1-1) and missing (0-0) (Liu et al., 2012).
Methylation level (%) in different treatments was calculated and compared using the Kruskal-Wallis test. There might be a situation where the treated abalone could differ in methylation at many loci, but the direction might not be consistently toward hypoor hyper-methylation. Therefore, we tested the difference in methylation frequency at each MSP between the control and stress treatments via a twotailed Fisher's exact test using a 5% criterion (Blouin et al., 2010). Multivariate analysis was also used to investigate individual profiles because it is a bandbased approach that does not assume Hardy-Weinberg equilibrium. Principal component analysis (PCA) on inter-profile covariance matrix followed by betweengroup eigen analysis (BPCA) was computed on MSP binary profiles using ADE-4 software. BPCA (i.e. PCA among groups based on the PCA among individuals) divides the variance into within-and between-group components and is regarded as analogous to F-statistics (called βST here) given that it is a Euclidean approach. Statistical significance was assessed by the Romesburg randomization test (104 permutations).
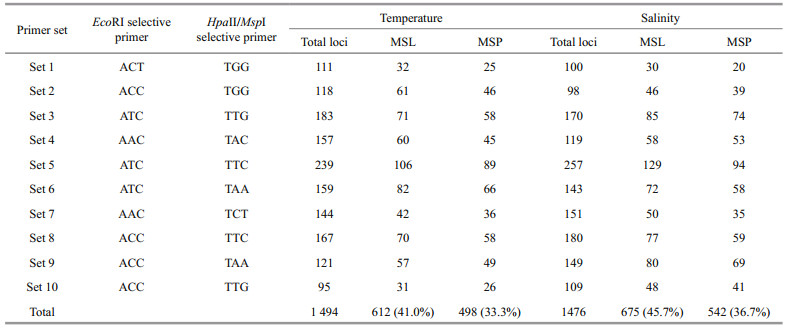
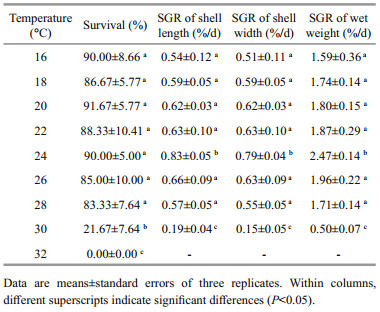
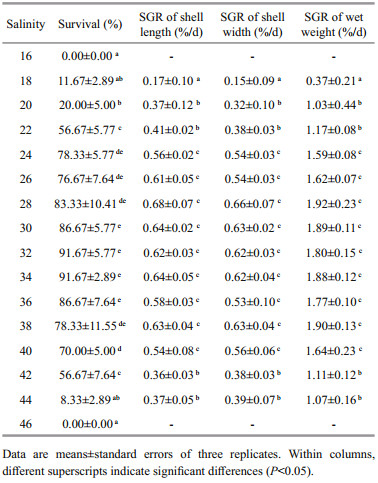
3 RESULT 3.1 Individual effect of temperature and salinity on survival and growthTables 1 and 2 show the summarized data for temperature and salinity treatments, respectively. One-way ANOVA indicated that both temperature (P < 0.001) and salinity (P < 0.001) had significant effects on the survival and growth of juvenile H. discus hannai Ino.
|
|
All juveniles reared at 32℃ died within 4 days. Survival at temperatures from 16 to 28℃, ranging from 83.33% to 91.67%, was significantly higher than that at 30℃. With temperature rising from 16 to 30℃, the specific growth rates first increased and then decreased. Maximum specific growth rates of shell length, shell width and wet weight were obtained at 24℃, which were significantly higher than those in the other treatments, with the values being 0.83%/d, 0.79%/d and 2.47%/d, respectively. Survival at salinities from 24 to 40 was not less than 70%, whereas no juvenile survived at 16 or 46. Specific growth rates at salinities from 24 to 40 were similar and significantly higher than those in the other treatments. Based on the above data, the suitable ranges of temperature and salinity for juvenile H. discus hannai Ino were determined as 16–28℃ at a constant salinity of 32 and 24–40 at a constant temperature of 20℃, respectively.
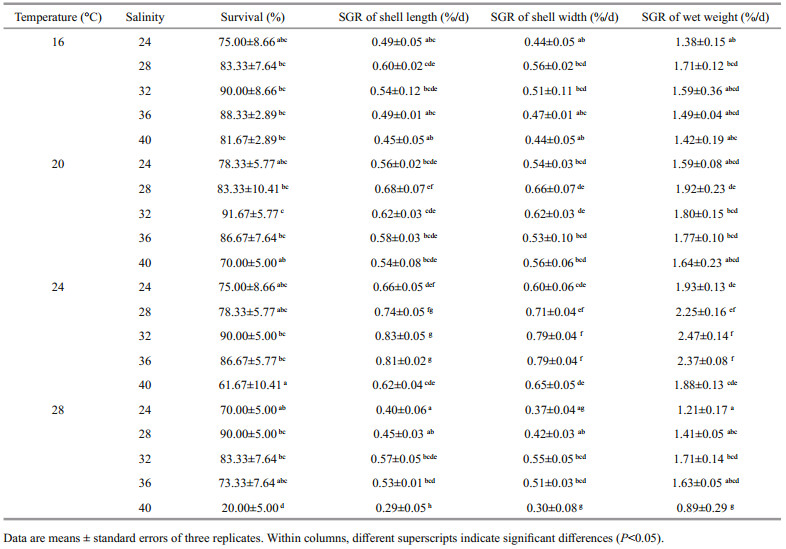
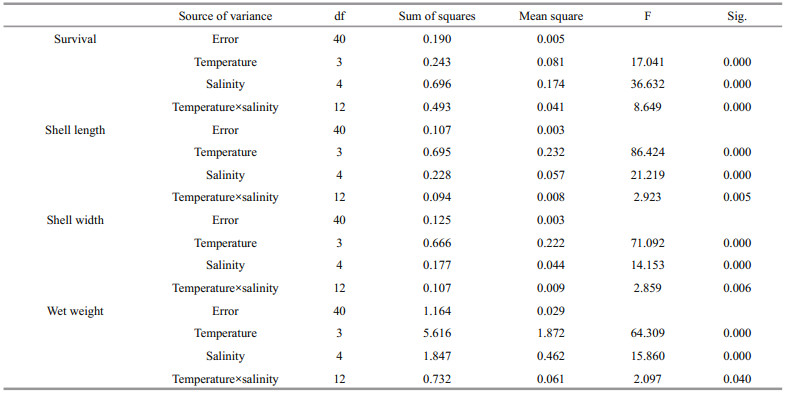
3.2 Combined effect of temperature and salinity on survival and growthTable 3 shows the summarized data for combined treatments of temperature and salinity. Two-way ANOVA indicated that temperature (P < 0.001), salinity (P < 0.001) and their interaction (P < 0.05) all significantly affected the survival and growth of juveniles (Table 4). The mean survival rates were 83.67%, 82.00%, 78.33% and 67.33% at 16, 20, 24 and 28℃, respectively. Regardless of temperature, survival was higher at 28, 32, 36 (83.75%–88.75%) than at 24 (74.58%) and 40 (58.34%). Juveniles reared at the highest salinity of 40 had a relatively low survival at 24℃ and 28℃. At all salinities, the specific growth rates increased as temperature ascended from 16 to 24℃, and decreased at 28℃. Specific growth rates at salinities from 28 to 36 were greater than those in the other treatments regardless of temperature. The poorest growth was observed at the combination of 28℃ and 40. Response-surface contour plots in Fig. 1 show that the optimal conditions for survival and growth coincided best at 23–25℃ and 30–36.
|
|
|
| Figure 1 Response-surface contour plots showing survival (a) and specific growth rates of shell length (b), shell width (c) and wet weight (d) of juvenile H. discus hannai Ino reared in different combinations of temperature and salinity for 77 days The isobars show percentage survival in (a) and specific growth rates in (b) (%/d), (c) (%/d) and (d) (%/d). |
The 10 primer combinations assayed with the MSAP analysis provided a total of 1 494 loci in the temperature treatments, with 612 and 498 of these loci being classified as MSL and MSP, respectively. In the salinity treatments, 675 loci were classified as MSL among 1 476 screened loci, with 542 of them showing methylation-susceptible polymorphism (Table 5).
Table 6 shows the level of different DNA methylation states observed in selected treatments. The average level of full methylation among all treatments was greater (22.25%) than hemimethylation (10.95%), indicating that fully methylated in internal cytosine was the dominant methylation pattern in the H. discus hannai Ino genome. In temperature treatments, the total methylation (hemiand full methylation) level ranged from 31.19% to 33.96%, with a mean of 32.50%. No significant difference was detected in the level of hemimethylation (Kruskal-Wallis χ2=0.328, df=2, P=0.849), full methylation (Kruskal-Wallis χ2=0.692, df=2, P=0.708) or total methylation (Kruskal-Wallis χ2 =0.661, df=2, P=0.719). Total methylation levels were also quite similar among salinity treatments, with values of 33.49%, 33.83% and 34.34% for 22, 32 and 42, respectively. There was no significant difference in the level of hemi-methylation (KruskalWallis χ2=1.208, df=2, P=0.574), full methylation (Kruskal-Wallis χ2=0.472, df=2, P=0.790) or total methylation (Kruskal-Wallis χ2=0.196, df=2, P=0.907).

|
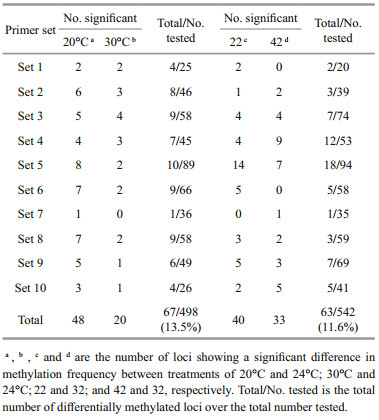
However, in Fisher's exact tests, 67 differentially methylated loci were identified among 498 MSP in temperature treatments with a percentage of 13.5%, which was higher than expected by chance (at 5% criterion). Among these loci, 48 loci were detected in 20℃ treatment and 20 loci in 30℃ treatment when compared with the control treatment of 24℃. Similarly, 63 out of 542 (11.6%) loci showed significant differences in methylation frequency attributable to salinity, with 40 and 33 loci detected in 22 treatment and 42 treatment, respectively (Table 7).
|
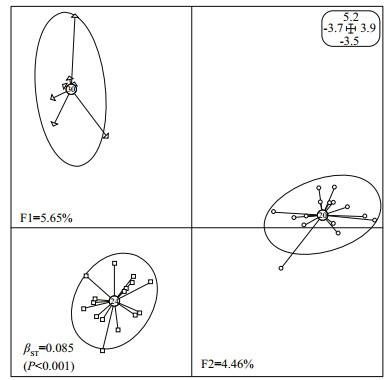
Figure 2 shows the epigenetic structure differentiation attributable to temperature. The PCA on the covariance matrix based on the MSP binary profile summarized 10.11% of total inertia in the first two axes. The three treatments showed pronounced separation, with 30℃ treatment exhibiting greater variation than the others. The multivariate BPCA based on the PCA scores resulted in a significant βST of 0.085 (P < 0.001), suggesting that epigenetic variance could be divided into distinct between-(8.5%) and within-(91.5%) group components.
|
| Figure 2 Between-group eigen analysis of the three temperature-treatment groups of juvenile H. discus hannai Ino using PCA values based on the epigenetic covariance matrix (MSP) F1 and F2 values show the contribution of the two principal components summarizing the total variance of each data set. βST was calculated by BPCA for epigenetic profiles and tested with 104 Romesburg randomization permutations. Circles, squares and triangles represent individuals from 20, 24 and 30℃ treatments, respectively. The group label shows the centroid for the points cloud in each group. Ellipses represent the dispersion of those points around their center. |
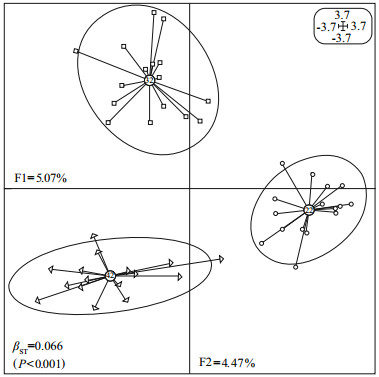
In the salinity treatments, three groups also showed obvious clustering in four quadrants by the first two axes (summarizing 9.54% of the total inertia), and the multivariate BPCA output a significant βST of 0.066 (P < 0.001), which indicated that the proportion of between-group epigenetic variance caused by salt stress was 6.6% (Fig. 3).
|
| Figure 3 Between-group eigen analysis of the three salinitytreatment groups of juvenile H. discus hannai Ino using PCA values based on the epigenetic covariance matrix (MSP) F1 and F2 values show the contribution of the two principal components summarizing the total variance of each data set. βST was calculated by BPCA for epigenetic profiles and tested with 104 Romesburg randomization permutations. Circles, squares and triangles represent individuals from 22, 32 and 42 treatments, respectively. The group label shows the centroid for the points cloud in each group. Ellipses represent the dispersion of those points around their center. |
Traditionally, H. discus hannai Ino is regarded as a stenothermal and stenohaline species, preferring lowtemperature and high-salinity waters. However, the single-factor data in our study confirmed a relatively wide tolerance to temperature (16–28℃) and salinity (24–40), which may have resulted from the selective breeding and long-term adaptation of the species to environmental heterogeneity in mass production. Compared with previous studies (Gao et al., 1990; Zhang et al., 2005; Cho and Kim, 2012), the abalone used in our study exhibited much greater tolerance to high temperature, with survival exceeding 80% at 28℃ and the best growth obtained at 24℃. Differen temperature tolerances and optima may result from variations in size and genetic background. Searle et al. (2006) suggested that the thermal tolerance and temperature optima of H. iris Gmelin were sizespecific and decreased as abalone size increased. Liu et al. (2005) indicated that the differences in genetic background mainly contributed to the variation in temperature optima of H. discus hannai Ino. It is also worth noting that the abalone in our study had experienced six generations of selection for rapid growth. There is a possibility that the fast-growing abalone have stronger environmental tolerances, expanding the span of growth time when they are exposed to adverse conditions. Hence, during the selection for rapid growth, the thermal tolerance of H. discus hannai Ino was also improved.
It is the first time that the combined effect of temperature and salinity on H. discus hannai Ino has been investigated. Two-way ANOVA indicated that the survival and growth of juvenile H. discus hannai Ino were significantly influenced by temperature, salinity and their interaction. Calabrese (1969) stated that the interactive effect of temperature and salinity was significant when the tolerance of either of these was approached. Consistent with this statement, the two factors in our study showed strong interaction at extreme combinations such as 28℃ and 40, at which both the survival and growth were the poorest. At salinity of 40, the survival rates at 16–20℃ were higher than those at 24–28℃, suggesting that the juveniles had greater tolerance to extreme salinities at lower temperatures than at higher temperatures. This observation is of value to abalone mariculture, recommending the culturists lower the seawater temperature appropriately to ensure good survival when the ambient salinity fluctuates considerably. Two-way ANOVA showed that temperature accounted for greater variance in specific growth rates than salinity, while it was the reverse in survival. Ruscoe et al. (2004) obtained similar results to ours, in finding that the growth of Scylla serrata was influenced more by temperature than by salinity, although both had significant effects.
Pronounced variation in the survival and growth of H. discus hannai Ino attributable to temperature and salinity has been observed in this and previous studies (Nie et al., 1984; Gao et al., 1990; Liu et al., 2005; Zhang et al., 2005; Wang, 2012), but the underlying epigenetic mechanism remains uncharacterized. In the present study, we first compared the extent and pattern of DNA methylation in H. discus hannai Ino reared in optimal and adverse conditions using the MSAP technique. The results showed that the methylation level of H. discus hannai Ino was 33.19%, which was higher than that of C. gigas (26.4%) (Jiang et al., 2013) and C. farreri (20.9%– 21.7%) (Sun et al., 2014). The abalone used in our study were derived from the same selective breeding population, indicating that there was high homogeneity in their genetic backgrounds. Hence, it is reasonable to hypothesize that the extensive phenotypic variation among different temperature and salinity treatments was largely affected by the epigenetic events.
Kruskal-Wallis tests showed that neither temperature nor salinity induced evident changes in the global methylation level, in contrast to the results in Oreochromis niloticus (Zhu et al., 2013) and Brassica napus var. oleifera (Marconi et al., 2013). It is possible that the methylation alteration occurred in a number of key genes, without inducing a large change in the methylation level. Therefore, the difference in methylation frequency at each MSP was tested. As we speculated, 67 and 63 loci exhibiting differential methylation frequencies were identified in temperature and salinity treatments, respectively. These loci may be involved in the stress acclimation of H. discus hannai Ino, and thus constitute a core set of epimarker resources that would facilitate further epigenetic studies in this species (Sun et al., 2014). It is worth noting that much more differential loci were found in the low-temperature treatment of 20℃ (48 loci) than in the high-temperature treatment of 30℃ (20 loci), and this might be related to the phenotypic differences in the two treatments. The result indicated that more stress-related genes were involved in the cold acclimation, ultimately resulting in better survival and growth of H. discus hannai Ino in the low-temperature treatment.
The BPCA plots based on methylation profiles showed that the abalone from different treatments were obviously separated, suggesting that both temperature and salinity could induce epigenetic differentiation in H. discus hannai Ino. Significant βST values also indicated that 8.5% and 6.6% of epigenetic variation was structured into the between-group component when stressed by temperature and salinity, respectively. The results provided evidence of the correlation between epigenetic reprogramming and phenotypic variation in H. discus hannai Ino, and verified the important role of DNA methylation in enabling abalone acclimation to environmental stresses.
5 CONCLUSIONOur study established the optimal rearing conditions of juvenile H. discus hannai Ino in terms of temperature and salinity, which may provide important guide to farming practice and industry development. We also analyzed the DNA methylation alteration in abalone reared in different conditions, presenting the first examination of the epigenetic regulatory mechanism of abalone in response to thermal and salt stresses.
6 ACKNOWLEDGEMENTWe wish to thank Dr. Q. JIANG and Dr. C. CHEN for providing support and help in the experimental process.
| Allan E L, Froneman P W, Hodgson A N, 2006. Effects of temperature and salinity on the standard metabolic rate(SMR) of the caridean shrimp Palaemon peringueyi. Journal of Experimental Marine Biology and Ecology, 337(1): 103–108. Doi: 10.1016/j.jembe.2006.06.006 |
| Blouin M S, Thuillier V, Cooper B, Amarasinghe V, Cluzel L, Araki H, Grunau C, 2010. No evidence for large differences in genomic methylation between wild and hatchery steelhead (Oncorhynchus mykiss). Canadian Journal of Fisheries and Aquatic Sciences, 67(2): 217–224. Doi: 10.1139/F09-174 |
| Cáceres-Puig J I, Abasolo-Pacheco F, Mazón-Suastegui J M, Maeda-Martínez A N, Saucedo P E, 2007. Effect of temperature on growth and survival of Crassostrea corteziensis spat during late-nursery culturing at the hatchery. Aquaculture, 272(1-4): 417–422. Doi: 10.1016/j.aquaculture.2007.06.030 |
| Calabrese A, 1969. Individual and combined effects of salinity and temperature on embryos and larvae of the coot clam, Mulinia lateralis (Say). Biological Bulletin, 137(3): 417–428. Doi: 10.2307/1540164 |
| Cho S H, Kim D S, 2012. Effects of feed type and temperature on growth of juvenile abalone, Haliotis discus hannai Ino. Journal of the World Aquaculture Society, 43(1): 114–119. Doi: 10.1111/jwas.2012.43.issue-1 |
| FAO (Food and Agriculture Organization of the United Nations), 2012. FAO Yearbook. Fishery and Aquaculture Statistics 2010. FAO Fisheries and Aquaculture Department, Food and Agriculture Organization of the United Nations, Rome: 109p. |
| Gao L X, Geng Y P, Li B, Chen J K, Yang J, 2010. Genomewide DNA methylation alterations of Alternanthera philoxeroides in natural and manipulated habitats:implications for epigenetic regulation of rapid responses to environmental fluctuation and phenotypic variation. Plant, Cell & Environment, 33(11): 1820–1827. |
| Gao X S, Liu Y F, Liu Y X, Liu J, 1990. Influence of temperature on feeding and growth of the young abalone. Oceanologia et Limnologia Sinica, 21(1): 20–26. |
| Jiang Q, Li Q, Yu H, Kong L F, 2013. Genetic and epigenetic variation in mass selection populations of Pacific oyster Crassostrea gigas. Genes & Genomics, 35(5): 641–647. |
| Kinne O, 1963. The effects of temperature and salinity on marine and brackish water animals. Ⅰ. Temperature.Oceanography and Marine Biology, 1: 301–340. |
| Laing I, 2002. Effect of salinity on growth and survival of king scallop spat (Pecten maximus). Aquaculture, 205(1-2): 171–181. Doi: 10.1016/S0044-8486(01)00663-9 |
| Li J Q, He Q G, Sun H, Liu X, 2012. Acclimation-dependent expression of heat shock protein 70 in Pacific abalone(Haliotis discus hannai Ino) and its acute response to thermal exposure. Chinese Journal of Oceanology and Limnology, 30(1): 146–151. Doi: 10.1007/s00343-012-1026-x |
| Li Q, Yu H, Yu R H, 2006. Genetic variability assessed by microsatellites in cultured populations of the Pacific oyster (Crassostrea gigas) in China. Aquaculture, 259(1-4): 95–102. Doi: 10.1016/j.aquaculture.2006.05.030 |
| Li X L, Lin Z X, Nie Y C, Guo X P, Zhang X L, 2009. Methylation-sensitive amplification polymorphism of epigenetic changes in cotton under salt stress. Acta Agronomica Sinica, 35(4): 588–596. |
| Lira-Medeiros C F, Parisod C, Fernandes R A, Mata C S, Cardoso M A, Gomes Ferreira P C, 2010. Epigenetic variation in mangrove plants occurring in contrasting natural environment. PLoS One, 5(4): e10326. Doi: 10.1371/journal.pone.0010326 |
| Liu S, Sun K P, Jiang T L, Ho J P, Liu B, Feng J, 2012. Natural epigenetic variation in the female great roundleaf bat(Hipposideros armiger) populations. Molecular Genetics and Genomics, 287(8): 643–650. Doi: 10.1007/s00438-012-0704-x |
| Liu X, Deng Y W, Zhang G F, 2005. Growth of eight Pacific abalone families at three temperatures. Acta Oceanologica Sinica, 24(3): 148–153. |
| Marconi G, Pace R, Traini A, Raggi L, Lutts S, Chiusano M, Guiducci M, Falcinelli M, Benincasa P, Albertini E, 2013. Use of MSAP markers to analyse the effects of salt stress on DNA methylation in rapeseed (Brassica napus var.oleifera). PLoS One, 8(9): e75597. Doi: 10.1371/journal.pone.0075597 |
| Morán P, Marco-Rius F, Megías M, Covelo-Soto L, PérezFigueroa A, 2013. Environmental induced methylation changes associated with seawater adaptation in brown trout. Aquaculture, 392(-395): 77–83. |
| Navarro-Martín L, Viñas J, Ribas L, Díaz N, Gutiérrez A, Di Croce L, Piferrer F, 2011. DNA methylation of the gonadal aromatase (cyp19a) promoter is involved in temperaturedependent sex ratio shifts in the European sea bass. PLoS Genetics, 7(12): e1002447. Doi: 10.1371/journal.pgen.1002447 |
| Nie Z Q, Chen W H, Ji M F, 1984. Studies on rearing conditions of abalone, Haliotis discus hannai Ino. I. the effects of temperature and food on the growth of larvae and youngs. Marine Fisheries Research(6): 35–40. |
| Roberts S B, Gavery M R, 2012. Is there a relationship between DNA methylation and phenotypic plasticity in invertebrates. Frontiers in Physiology, 2: 116. |
| Ruscoe I M, Shelley C C, Williams G R, 2004. The combined effects of temperature and salinity on growth and survival of juvenile mud crabs (Scylla serrata Forskål). Aquaculture, 238(1-4): 239–247. Doi: 10.1016/j.aquaculture.2004.05.030 |
| Searle T, Roberts R D, Lokman P M, 2006. Effects of temperature on growth of juvenile blackfoot abalone, Haliotis iris Gmelin. Aquaculture Research, 37(14): 1441–1449. Doi: 10.1111/are.2006.37.issue-14 |
| Sun Y, Hou R, Fu X T, Sun C S, Wang S, Wang C, Li N, Zhang L L, Bao Z M, 2014. Genome-wide analysis of DNA methylation in five tissues of Zhikong scallop, Chlamys farreri. PLoS One, 9(1): e86232. Doi: 10.1371/journal.pone.0086232 |
| Tan M P, 2010. Analysis of DNA methylation of maize in response to osmotic and salt stress based on methylationsensitive amplified polymorphism. Plant Physiology and Biochemistry, 48(1): 21–26. Doi: 10.1016/j.plaphy.2009.10.005 |
| Wang S W, 2012. Analysis of the toleration to different salinities in Haliotis discus hannai Ino. Institute of Oceanology. Chinese Academy of Science, Qingdao. |
| Xue M, Ke C H, Wang D X, Wei Y J, Xu Y B, 2010. The combined effects of temperature and salinity on growth and survival of hatchery-reared juvenile spotted babylon, Babylonia areolata (Link 1807). Journal of the World Aquaculture Society, 41(S1): 116–122. |
| Zhang G F, Que H Y, Liu X, Xu H S, 2004. Abalone mariculture in China. Journal of Shellfish Research, 23(4): 947–950. |
| Zhang M, Wang Z S, Gao X S, 2005. Comparison on adaptability of abalone Haliotis discus hannai Ino to temperatures at different growing stages. Journal of Fishery Sciences of China, 12(6): 720–725. |
| Zhao Y, Chen M Y, Storey K B, Sun L N, Yang H S, 2015. DNA methylation levels analysis in four tissues of sea cucumber Apostichopus japonicus based on fluorescencelabeled methylation-sensitive amplified polymorphism(F-MSAP) during aestivation. Comparative Biochemistry and Physiology, Part B, 181: 26–32. Doi: 10.1016/j.cbpb.2014.11.001 |
| Zhu H P, Lu M X, Huang Z H, Gao F Y, Ke X L, Liu Z G, Li Q Y, Liu Y J, 2013. Effect of low temperature on genomic DNA methylation in Nile tilapia (Oreochromis niloticus). Journal of Fisheries of China, 37(10): 1460–1467. Doi: 10.3724/SP.J.1231.2013.38611 |
 2017, Vol. 35
2017, Vol. 35