Institute of Oceanology, Chinese Academy of Sciences
Article Information
- WANG Yueyun(王跃云), SUI Jixing(隋吉星), LI Xinzheng(李新正)
- Two new records of Spiochaetopterus Sars, 1856 (Chaetopteridae, Polychaeta) from Chinese waters
- Chinese Journal of Oceanology and Limnology, 35(6): 1465-1470
- http://dx.doi.org/10.1007/s00343-017-6075-8
Article History
- Received Mar. 19, 2016
- accepted in principle May. 10, 2016
- accepted for publication Sep. 22, 2016
2 University of Chinese Academy of Sciences, Beijing 100049, China;
3 Laboratory for Marine Biology and Biotechnology, Qingdao National Laboratory for Marine Science and Technology, Qingdao 266235, China
The genus Spiochaetopterus Sars, 1856, distributed worldwide in subtidal to abyssal waters, comprises about 18 species that build tubes in variety of benthic environments like sand, mud and even deep cold methane seeps, comprising about 18 species (Read and Bellan, 2015). Presently, two species of Spiochaetopterus have been recorded from the China Sea: S. costarum (Claparède, 1868) and S. iheyaensis Nishi, 2008 (Liu, 2008; Nishi, 2008), though the former is likely a misidentification given this species is otherwise known from the Mediterranean Sea and western-Atlantic coast (Bhaud, 1998), and specimens labeled S. costarum deposited in Marine Biological Museum (MBM) of the Chinese Academy of Sciences (CAS) proved to be S. koreana Bhaud, Koh & Hong, 2002.
The body of chaetopterids can be divided into three regions: anterior, mid and posterior. The anterior (A) region has uniramous parapodia; the mid-region (B) has biramous parapodia with flap-shaped notopodia and neuropodial tori; and the posterior (C) region has biramous parapodia with simple pointed notopodia. A4 indicating the fourth chaetiger of region A. A pair of long palps is located on either side of the prostomium.
Examination of polychaetes in MBM collections revealed two species not reported previously from the China Sea: S. koreana (including specimens previously misidentified as S. costarum) and S. sanbanzensis Nishi,Bhaud & Koh, 2004. We redescribe these species based on this new material.
2 MATERIAL AND METHODDuring recent surveys (October and November 2014 and 2015), new chaetopterid specimens (MBM 283918–283924) were collected by box corer from 4-to 75-m depth in Jiaozhou Bay, the Yellow Sea and north of the East China Sea (Fig. 1). Samples were sieved (0.5-mm mesh) in the field, preserved in 75% ethanol, later stained with Rose Bengal and laboratory sorted, with specimens then accessioned into MBM collections. Additional, previously unidentified specimens collected between 1959 and 1980 (MBM 193886, 010882, 010901, 010914, 010915) were also examined.
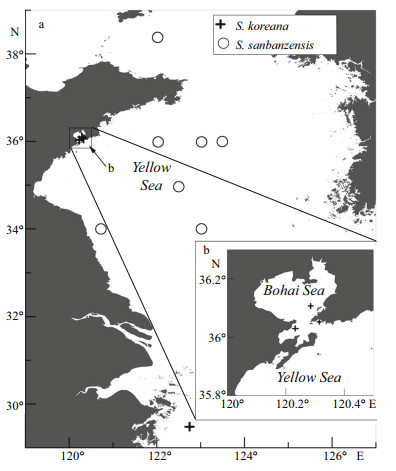
|
| Figure 1 Sampling sites of Spiochaetopterus koreana and S. sanbanzensis, China Sea, 1958–2015 |
Observations were made using a Zeiss Stemi 2000-C Stereo Microscope with AxioCam MRc 5 camera. Some specimens were stained with a saturated solution of methyl green in ethyl alcohol to observe notopodia and neuropodia of region B. The ultrastructure of modified chaetae on the fourth chaetiger (A4) was observed with scanning electron microscope (SEM). The terminology of body regions and modified A4 chaetae follows Crossland (1904) and Bhaud (1998).
3 TAXONOMYFamily Chaetopteridae Audouin and Milne Edwards, 1833
Genus Spiochaetopterus Sars, 1856
Spiochaetopterus koreana Bhaud, Koh& Hong, 2002
Spiochaetopterus koreana Bhaud et al. 2002: 350–358, Fig. 2A–E.
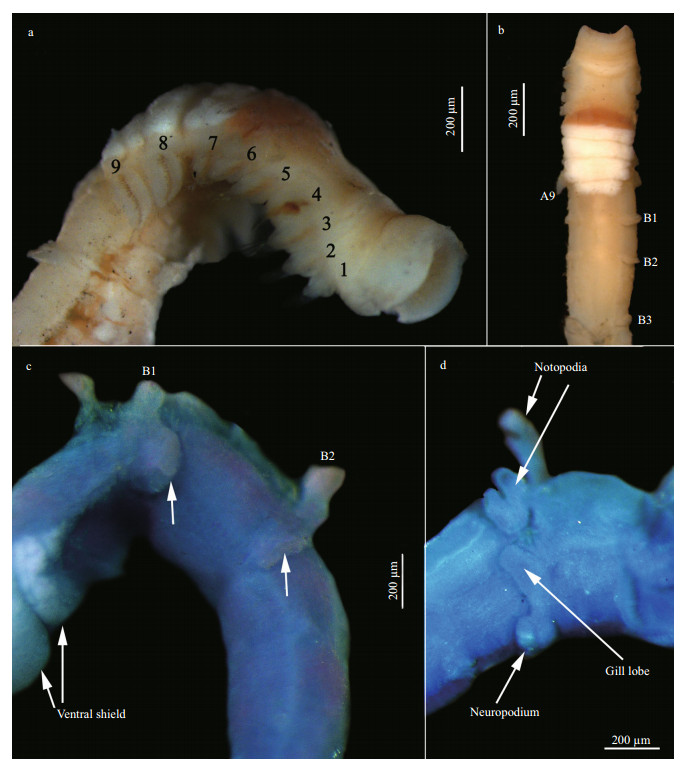
|
| Figure 2 Spiochaetopterus koreana (MBM 193886) a. anterior region, lateral view, palps missing; b. anterior region and part of middle region, ventral view, palps missing; c. first two unilobed neuropodia, lateral view, methyl green stain; d. parapodia of B1, lateral view, methyl green stain. |
Material examined (MBM, number of specimens): 193886, 2, Jiaozhou Bay, 36.064°N, 120.326°E, mud, 6 m, 15 July 1980; 190809, 4, East China Sea, mud, 75 m, 21 July 1976; 190806, 1, East China Sea, mud, 84 m, 21 July 1976; 200618, 1, Beibu Gulf, silt, 32 m, 15 January 1962; 236444, 1, Beibu Gulf, mud, 2 m, 13 October 1981; 283918, 1, Jiaozhou Bay, 36.100°N, 120.291°E, silt, 4 m, November 2014; 283919, 1, Jiaozhou Bay, 36.036°N, 120.233°E, mud, 5 m, November 2014; 283920, 1, north of East China Sea, 29.480°N, 122.750°E, silt, 53 m, 3 November 2015.
Description All material examined (12 specimens) incomplete, missing posterior body region, 1.2– 2.6 mm long in region A and 0.29–0.4 mm wide at the fourth chaetiger of region A.
Region A of preserved specimens often curved to dorsal side, with nine chaetigers (Fig. 2a). Peristomium horseshoe shaped, not overlapping prostomium at dorsal side. Anterior part of peristomium slightly recurved (Fig. 2b). Prostomium rounded, with two conspicuous, dark brown lateral eyespots. Eyespots anterior to palps and visible in dorsal view. Ciliated groove present on mid-dorsal side of region A. Ventral side of region A with long ventral shield (ventral glandular area), distinguished by different color: brown-to-reddish anterior part and white posterior part (Fig. 2a, b), with anterior part crescent-shaped, from A6 to A7 (Fig. 2a), white posterior part from A7 to posterior end of A9, often divided into four sections.
Parapodia of region A uniramous, only notopodia present. Notopodia of A4 stouter than those of otherchaetigers; each notopodium of A4 with single modified cutting chaeta accompanied with common chaetae (other notopodia with common chaetae only). Modified chaetae of A4 with inflated, obliquely truncated distal end, with two equal lobes on the upper oblique plane, heart-shaped in distal view (Fig. 3d, e). Common chaetae lanceolate.

|
| Figure 3 Modified A4 chaetae and uncini (a–c. Spiochaetopterus sanbanzensis, MBM 283922; d–f. S. koreana, MBM 283919) a. head of modified A4 chaeta, ventral view; b. modified A4 chaeta, ventral view; c. uncini plate from B1 neuropodium; d, e. modified A4 chaetae of same specimen, apical view; f. uncinal plate from B1 neuropodium. Scale: a=400 μm; b=1.0 mm; c=10 μm; d, e=200 μm; f=10 μm. |
Region B with more than four chaetigers. Neuropodia uniramous on B1 and B2, each neuropodium with one posterior-oriented lobe (Fig. 2c). Other neuropodia of region B bilobed, comprising a smaller upper and larger lower lobe. Notopodia all bilobed, with Y-shaped inner lobe and triangular gill lobe (Fig. 2d), with base of gill lobe connected to neuropodium lobe (Fig. 2d). Uncinal plate of B1 small and triangular, with 18–19 small teeth (Fig. 3f).
Region C incomplete, with only few chaetigers present. Notopodia unilobed with slightly inflated distal part; chaetae lanceolate. Neuropodia bilobed, comprising a smaller upper and larger lower lobe. Pygidium missing.
Distribution Yellow Sea, East China Sea, 2–84 m.
Remarks Our specimens agree well with those in the type description (Bhaud et al., 2002). This species is similar to S. okinawaensis Nishi and Bhaud, 2000 in that both have eyespots, nine chaetigers on regionA, a ventral shield divided into two parts by color, and heart-shaped modified A4 chaetae. However, these two species differ in that the first two neuropodia of region B are unilobed in S. koreana, but only B1 neuropodia is unilobed in S. okinawaensis, the number of B1 uncinal plate teeth in S. okinawaensis is 14–16 whereas it is 18–19 in S. koreana, and the pair of brown spots usually present on the lateral side of region B of S. okinawaensis are absent in S. koreana.
Spiochaetopterus sanbanzensis Nishi, Bhaud & Koh, 2004

|
| Figure 4 Spiochaetopterus sanbanzensis (a, b: MBM 010882; c–e: MBM 283924) a. anterior region, dorsal view; b, c. anterior region, lateral view, showing color pattern (arrows show crescent ventral shield); d. neuropodia (NE) of first two chaetigers of middle region, lateral view; e. notopodia of middle region, dorsal-lateral view. |
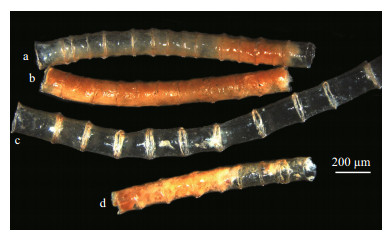
|
| Figure 5 Tube of Spiochaetopterus sanbanzensis (MBM 010882) a, d. tube with intervals shorter than width of tube; b. tube containing worm; c. empty tube. |
Spiochaetopterus sanbanzensis Nishi et al. 2004: 457–464, Figs. 2–3, 6E.
Material examined (MBM, number of specimens): 010882, 3, Yellow Sea, 35.00°N, 122.50°E, mud, 50 m, 20 July 1959; 010914, 1, Yellow Sea, 36.00°N, 123.00°E, mud, 58 m, October 1958; 010915, 1, Yellow Sea, 34.00°N, 120.75°E, mud, 52 m, October 1958; 010901, 1, Yellow Sea, 34.00°N, 123.00°E, mud, 47 m, 21 September 1959; 283921, 1, Yellow Sea, 36.00°N, 123.50°E, mud, 75 m, 20 October 2014; 283922, 1, Yellow Sea, 36.00°N, 122.00°E, mud, 41.7 m, 20 October 2014; 283923, 1 (complete, with tube), Yellow Sea, 36.00°N, 123.00°E, mud, 71 m, 20 October 2014; 283924, 1 (complete), Yellow Sea, 38.40°N, 122.00°E, mud, 50 m, 18 October 2014.
Description Two of 10 available specimens complete. Region A with nine chaetigers, 1.4–2.0 mm long excluding palps, 0.3–1.2 mm wide at fourth chaetiger; last two chaetigers slightly longer than anterior ones (Fig. 4a, b). Peristomium well developed, horseshoe shaped, with two triangular lateral ends (Fig. 4a, c) nearly covering small prostomium. Two long dorsally grooved palps, usually coiled, arising from latero-posterior base of prostomium, with pair of brown eyespots on lateral side of prostomium, visible when palps removed (Fig. 4a).
Parapodia of region A uniramous. Notopodia of A4 with modified enlarged chaeta accompanied with many common lanceolate chaetae, other notopodia of region A with lanceolate chaetae only. Modified A4 chaeta with inflated head asymmetrically cordate in posterior view, with two unequal lobes (Fig. 3a, b). Left side of lateral oblique edge of upper plane with several small teeth (Fig. 3a). Modified A4 chaeta very large, with teeth distinguishable under high resolution stereoscopic microscope. Shaft of modified chaeta stout, with shallow groove on ventral side of anterior portion (Fig. 3b).
Ventral side of region A remarkable for ventral shield and color pattern. Ventral shield long, extending from peristomium to A9, divided into two differentcolored parts: light brown before A7 and milky-white from A7 to A9, with brown crescent-shaped area on A6–A7 followed by white crescent-shaped area on A7–A8 (Fig. 4b, c). Ventral shield on A8–A9 separated into two parts. Significant brown pigmentation on ventral surface of peristomium to chaetiger A7 (Fig. 4b,c). Pigmentation varying among specimens: some dark, others light. Brown dots on ventral surface of notopodia, separating each chaetiger (Fig. 4b,c), with dots on first seven chaetigers very conspicuous, thereafter sometimes absent.
Region B with more than three chaetigers (one specimen with more than 40 chaetigers). First three chaetigers longer than following ones. Neuropodia unilobed in B1 and B2 (Fig. 4d, e). Following neuropodia bilobed, smaller than first two. Notopodia all bilobed, with Y-shaped inner lobe and triangularshaped gill lobe on lateral side of body (Fig. 4d). Uncinal plate of B1 triangular, with 17–19 small teeth.
Region C slender and fragile, with more than 60 chaetigers. Neuropodia bilobed, with lateral lobe smaller than ventral one. Notopodia unilobed, knoblike, with single inner chaeta.
Tube very long, vertical in muddy sediment, transparent (Fig. 4b, d), unbranched, with conspicuous bamboo-like articulations. Length of intervals variable (Fig. 4a, c), with distance between intervals usually equal to or greater than tube width.
Distribution Yellow Sea; Japan. Muddy sediments, depths 41.7–75 m.
Remarks Spiochaetopterus sanbanzensis resembles S. koreana and S. okinawaensis in that all have nine chaetigers in region A and two eyespots on the prostomium. However, the first two neuropodia in region B are unilobed in S. sanbanzensis and S. koreana, but only the first is unilobed in S. okinawaensis. The color pattern S. sanbanzensis is remarkable in that it has numerous brown spots on the ventral surface of region A, extending from the peristomium to A7. This trait seems to be unique to S. sanbanzensis and remains apparent on preserved specimens collected in the 1950s. The head of modified A4 chaeta of S. sanbanzensis has two unequal lobes, and crenulated external edges, while that of S. koreana has two equal lobes. The ventral shield on A7–A9 of S. sanbanzensis is divided into three parts, while the ventral shield on A7–A9 of S. koreana is usually divided into four parts.
Our specimens have more chaetigers in region B and C than the type specimens, the latter with 5–10 and more than 10 in regions B and C respectively (Nishi et al., 2004). Moreover, the uncinal plate of B1 in our specimen has more teeth. Specimens of S. sanbanzensis from Tokyo Bay have 12 or 13 small teeth on the uncinal plate of B1, whereas there are 17–19 small teeth on this plate in specimens from the Yellow Sea.
4 DISCUSSIONSpiochaetopterus is a poorly known genus because its representatives are small and fragile. Despite its near ubiquitous distribution, the systematic status of some species referred to it remains problematic, for example, S. tropicus Grube, 1877 and S. patagonicus Kinberg, 1866. The original description (Grube, 1877) of S. tropicus reports a pair of foliaceous organs on the bases of the palps, but these may be tentacular cirri typical of Phyllochaetopterus (Gitay, 1969). Spiochaetopterus patagonicus is a nomen dubium because the short original description inadequately characterizes the species and the type specimen is ruined and offers no characters of systematic value (Kinberg, 1866; Gitay, 1969).
Ten species have been recorded from the Pacific Ocean: S. pottsi (Berkeley, 1927), S. monroi (Monro, 1933), S. okudai (Okuda, 1935), S. sagamiensis Nishi, Miura & Bhaud, 1999, S. okinawaensis Nishi and Bhaud, 2000, S. sesokoensis Nishi and Bhaud, 2000, S. koreana, S. izuensis Nishi, Bhaud & Koh, 2004, S. sanbanzensis, and S. iheyaensis. Among them, after our records, three species are reported from China Seas: S. sanbanzensis (type locality Tokyo Bay), S. koreana (type localities west and south coasts of South Korea) and S. iheyaensis (types from Iheya Seamount, off Okinawa, East China Sea). Spiochaetopterus iheyaensis is present in chemosynthetic communities (Nishi, 2008). It has one or two pear-shaped modified A4 chaetae and two chaetigers in region B, while S. sanbanzensis and S. koreana have heart-shaped modified A4 chaetae and more than five chaetigers in region B. Spiochaetopterus koreana inhabits silty-sand sheltered areas according to the original description (Bhaud et al., 2002). Our specimens are found in muds and silts from 2–84 m depth. Spiochaetopterus sanbanzensis was described from 2 m in muds, but our specimens of S. sanbanzensis were found in muds from 41.7–75 m depth. Spiochaetopterus sanbanzensis is widely distributed in the Yellow Sea (Fig. 1). This study extends the geographical distribution of S. koreana and S. sanbanzensis from northwest Pacific Ocean.
5 ACKNOWLEDGMENTWe would like to thank Eijiroh Nishi for valuable discussions regarding species identification. We are grateful to the managers of the MBMCAS for assistance in sorting specimens.
| Audouin J V, Milne-Edwards H, 1833. Classification des Annélides, et description de celles qui habitent les côtes de la France. Annales des Sciences Naturelles, Paris, 30: 411–425. |
| Berkeley E, 1927. A new genus of Chaetopteridae from the northeast Pacific:with some remarks on allied genera. Proceedings of the Zoological Society of London, 97: 441–445. |
| Bhaud M R, 1998. Species of Spiochaetopterus (Polychaeta, Chaetopteridae) in the Atlantic-Mediterranean biogeographic area. Sarsia, 83(3): 243–263. Doi: 10.1080/00364827.1998.10413685 |
| Bhaud M R, Koh B S, Hong J S, 2002. Contribution to the present status of Spiochaetopterus costarum:description of Spiochaetopterus koreana, a new species of Chaetopteridae (Polychaeta) from the West Coast of Korea. Proceedings of the Biological Society of Washington, 115(2): 350–358. |
| Claparède E, 1870. Les Annélides Chétopodes du Golfe de Naples.Annélides sédentaires. Mémoires de la Société de physique et d'histoire naturelle de Genève, 20: 1–225. |
| Crossland C, 1904. The Polychaeta of the Maldive Archipelago from the Collections made by Stanley Gnrdiner in 1899. Proceedings of the Zoological Society of London, 1(18): 270–286. |
| Gitay A, 1969. A contribution to the revision ofSpiochaetopterus(Chaetopteridae, Polychaeta). Sarsia, 37(1): 9–20. Doi: 10.1080/00364827.1969.10411142 |
| Grube A E, 1877. Anneliden-Ausbeute S.M. S.Gazelle.. Monatsbericht der Koniglich Preussischer Akademie der Wissenschaften zu Berlin, 1877: 509–554. |
| Kinberg J G H, 1866. Annulata nova. Öfversigt af Königlich Vetenskapsakademiens förhandlingar, Stockholm, 23(4): 97–103. |
| Liu R Y, 2008. Checklist of Marine Biota of China Seas. Science Press, Beijing, China1267p. |
| Monro C A, 1933. The Polychæta Sedentaria collected by Dr. C. Crossland at Colón, in the Panama region, and the Galapagos Islands during the expedition of the S.Y. ‘St. George’. Proceedings of the Zoological Society of London, 103(4): 1039–1092. |
| Nishi E, 2008. A new species of Spiochaetopterus(Chaetopteridae:Polychaeta) from the Iheya seamount off Okinawa in East China Sea, Japan. Proceedings of the Biological Society of Washington, 121(2): 289–294. Doi: 10.2988/06-30.1 |
| Nishi E, Bhaud M R, 2000. Two new species ofSpiochaetopterus(Polychaeta:Chaetopteridae) from Okinawa, Japan, with notes on Pacific Spiochaetopterus. Pacific Science, 54(1): 15–26. |
| Nishi E, Bhaud M R, Koh B S, 2004. Two new species of Spiochaetopterus (Annelida:Polychaeta) from Sagami Bay and Tokyo Bay, central Japan with a comparative table of species from Japanese and adjacent waters. Zoological Science, 21(4): 457–464. Doi: 10.2108/zsj.21.457 |
| Nishi E, Miura T, Bhaud M, 1999. A new species of Spiochaetopterus (Chaetopteridae:Polychaeta) from a cold-seep site off Hatsushima in Sagami Bay, central Japan. Proceedings of the Biological Society of Washington, 112: 210–215. |
| Read G, Bellan G, 2015. Spiochaetopterus M Sars, 1856. In:Read G, Fauchald K eds. World Polychaeta database. |
| Okuda S, 1935. Chaetopterids from Japanese waters. Journal of the Faculty of Science Hokkaido Imperial University Series 6, 4(2): 87–102. |
| Sars M, 1856. Nye Annelider. Fauna littoralis Norvegiae 2. 24. Fredrik D. Beyer, Bergen. |
 2017, Vol. 35
2017, Vol. 35

