Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZHAO Bo(赵博), LIU Jinhu(刘金虎), SONG Junjie(宋骏杰), CAO Liang(曹亮), DOU Shuozeng(窦硕增)
- Evaluation of removal of the size effect using data scaling and elliptic Fourier descriptors in otolith shape analysis, exemplified by the discrimination of two yellow croaker stocks along the Chinese coast
- Chinese Journal of Oceanology and Limnology, 35(6): 1482-1492
- http://dx.doi.org/10.1007/s00343-017-6012-x
Article History
- Received Jan. 25, 2016
- accepted in principle Apr. 1, 2016
- accepted for publication Sep. 17, 2016
2 University of Chinese Academy of Sciences, Beijing 100049, China;
3 Laboratory for Marine Ecology and Environmental Science, Qingdao National Laboratory for Marine Science and Technology, Qingdao 266071, China
An understanding of stock structure is essential for effective management of fisheries that contain multiple stocks. Studies on fish biology or population dynamics and most estimates of yield are conducted on the basis of a well-established population structure (Campana and Casselman, 1993). Techniques including genetic analysis, otolith elemental fingerprinting and otolith shape analysis or combinations thereof are widely applied for stock discrimination (Bentzen et al., 1996; Begg and Waldman, 1999; Campana, 1999; Thresher, 1999; Longmore et al., 2010; Smith and Campana, 2010). Among these techniques, otolith shape analysis has shown promise as a potential technique for stock identification due to the rapid development of digital photographic technology and statistical tools and their application to shape analysis (Begg and Brown, 2000). Otoliths grow throughout the life of teleosts and are metabolically inert and species specific. The external characteristics of otoliths vary among geographic stocks due to differences in the ontogeny, genetics and environmental history of fish (Castonguay et al., 1991; Lombarte and Lleonart, 1993; Begg and Brown, 2000); thus, otolith morphology is a potential indicator of discrete stocks. Moreover, shape analysis is a relatively inexpensive and time-efficient method compared with both genetic and microchemical techniques (Tracey et al., 2006). As an ideal marker of fish population affiliation, the use of otolith shape to discriminate among stocks has been investigated in many fishes, such as herring Clupea harengus (Burke et al., 2008), Atlantic cod Gadus morhua (Campana and Casselman, 1993; Cardinale et al., 2004), haddock Melanogrammus aeglefinus (Begg and Brown, 2000), and southern blue whiting Micromesistius australis (Legua et al., 2013).
In the analysis of otolith shape, fish size is a potential source of variability in morphometric measures because size is associated with individual ontogeny (Lleonart et al., 2000). Some statistical procedures have been used to eliminate the size effect of fish individuals. The linear model Y=a+bX is widely used to scale variables correlated with fish size, where the coefficient b is equal to the common within-group slope. In this method, the product of the slope (b) and fish length is subtracted from the observed variables to obtain size-free variables (i.e., standardized or scaled variables; Bolles and Begg, 2000; Cardinale et al., 2004). Another method for removal of the size effect is based on an allometric growth model, Y=αXβ (Lleonart et al., 2000). However, the accuracy of these two models in otolith morphology studies and the extent to which the two models remove the size effect on scaled variables have rarely been investigated. Although otolith shape analysis is widely used for stock discrimination, very few studies have attempted to identify weaknesses of this method. For example, variation in fish size (e.g., length range or distribution), even at a small scale, could cause significant discrepancies in stock discrimination; these potential discrepancies have rarely been investigated thus far. To address these questions, we used two known stocks of yellow croaker, Larimichthys polyactis, from Chinese coastal waters as a case study to evaluate and compare the effectiveness of the two scaling models in removing the size effect in otolith shape analysis.
Yellow croaker is an important commercially exploited fish distributed in the seas of China (Guo et al., 2006; Xiao et al., 2009; Xu and Chen, 2010). Both morphological and genetic analyses have shown that the two sample groups of yellow croaker used in this study come from different geographic stocks (Ye, 1991; Jin, 1996; Meng et al., 2003). Using known stock samples for this otolith shape analysis allowed us to effectively evaluate the results of stock discrimination and compare the adjusting effect in a reliable way. This case study was primarily aimed at determining whether frequently applied scaling methods used in otolith shape analysis and normalized elliptic Fourier descriptors (NEFDs) can effectively remove the size effect as expected, and if these methods do not remove the size effect, what measures should be taken to minimize discrepancies during otolith sampling and morphological data analysis.
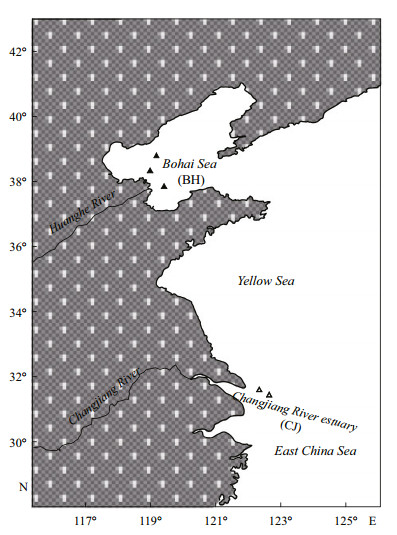
2 MATERIAL AND METHOD 2.1 Sample collectionThe samples used in this study came from the otolith collection of the Institute of Oceanology of the Chinese Academy of Sciences and were collected during fishery research surveys in Chinese coastal waters, as described in Dou et al. (2012). This collection includes yellow croaker samples from the Bohai Sea (BH) and the Changjiang River estuary of the East China Sea (CJ; Fig. 1). Immediately after capture, the fish were labelled and frozen for subsequent biological analysis. In the laboratory, the total length (TL, ±1 mm), weight (±0.1 g), sex, reproductive maturity stage and other biological characteristics of the fish were routinely determined and recorded. The sagittal otoliths on both the left and right sides were removed from each fish, cleaned of adhering tissues in distilled water, and stored dry in sealed glass vials until morphological analysis.
|
| Figure 1 Sampling sites of the two yellow croaker stocks: BH, the Bohai Sea stock; CJ, the Changjiang River estuary stock |
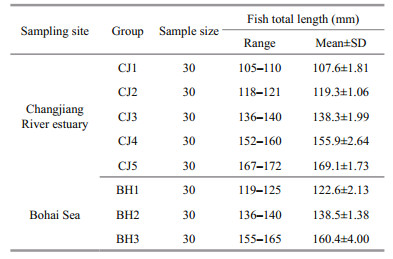
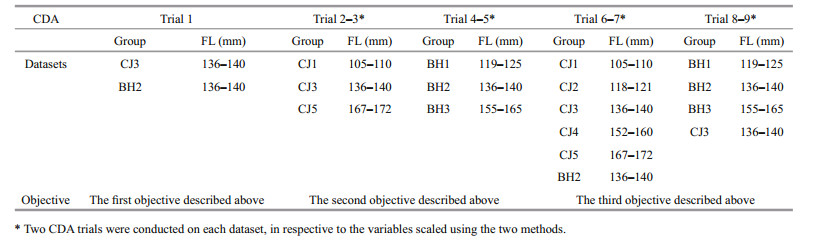
A total of 282 (185 males and 97 females) and 472 (263 males and 209 females) pairs of otoliths from the BH stock and the CJ stock, respectively, were analysed morphologically to test the inter-or intra-stock differences in shape characters between sexes or body sides. Because the objective of the study was to test the effectiveness of the two models in removing the size effect in otolith shape analysis, the otolith sampling programme was designed to include 3 length groups from the BH stock (BH1–BH3) and 5 length groups from the CJ stock (CJ1–CJ5), including 30 specimens in each group with a relatively uniform individual length distribution (the maximum individual length difference was controlled to be less than 1 cm). A total of 8 groups (n=30×8) with different length ranges were used for otolith morphology analysis (Table 1).
|
Prior to imaging, the otoliths were cleaned in distilled water with an ultrasonic cleaner, then airdried and weighed to the nearest 0.1 mg using an electronic balance. Digital images of both the left and right sagittal otoliths of each fish were captured using a video camera linked to a binocular microscope (ACT-2, Nikon SMZ1000; Tokyo, Japan). The otoliths were positioned on a dark background with the sulcus acusticus facing down and the anterior sidepointing to the left for the left otolith, or to the right for the right otolith (Fig. 2).

|
| Figure 2 The right (a) and the left (b) otoliths of a yellow croaker |
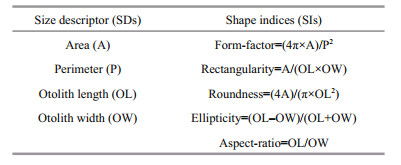
The otolith size descriptors (SDs: area, length, width, perimeter, minimum diameter, maximum diameter, mean diameter, minimum radius, maximum radius and the radius ratio) were obtained with ImagePro Plus (Legua et al., 2013). The shape indices (SIs: circularity, form factor, rectangularity, roundness and ellipticity) were obtained using the specified mathematical equations (Tuset et al., 2003; Table 2).
The digitizing program SHAPE 1.2 software was used to extract the elliptic Fourier descriptors (EFDs)from the images of the otoliths. The coefficients were normalized with the program such that they were invariant to otolith size and orientation. The Fourier power (FP) spectrum was calculated to determine the optimal number of harmonics for the best reconstruction of the otolith outline. For the nth harmonic, the Fourier power (FPn) was calculated according to the following formula (Crampton, 1995):
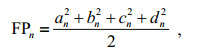
where an, bn, cn and dn are the Fourier coefficients of the nth harmonic. The cumulative variance (FPc) was calculated as follows:

Because the first eight harmonics described more than 99% of the cumulative power, otolith shape was summarized using 8 Fourier harmonics (i.e., 8×4=32 Fourier coefficients). However, normalization of the EFDs (NEFDs) resulted in degeneration of the first three coefficients to fixed values: c1=1, and c2=c3=0; thus, the number of Fourier coefficients was reduced to 29 (c4–c32) in the present study (Tracey et al., 2006).
2.3 Data analysisFor each analysis, the otolith shape variables (NEFDs, SDs and SIs) were first examined for normality and homogeneity using KolmogorovSmirnov (KS) and Levene's tests, respectively. Variables that did not meet the normality or homogeneity test were excluded from the statistical analysis.
A multivariate analysis of covariance (MANCOVA) was employed to test the differences in otolith morphology between sexes or body sides. Fish length was used as a covariate, and the shape variables were used as independent variables. Because no significant difference in otolith morphology were found between the sexes or between the left and right otoliths (MANCOVA, P>0.05, in all cases), the images of the left otoliths of both male and female samples were used in the statistical analyses.
2.3.1 Fish length scalingAn analysis of covariance (ANCOVA) was performed to test the magnitude of the fish length effect on each otolith shape variable and NEFD (dependent variable). Fish length was used as a covariate, and the length group was used as the independent variable. Variables were removed from analysis when adjustment for fish size was unsuccessful in MANOVA to compare otolith morphology between regions (Begg and Brown, 2000). When a significant interaction between the length group and fish length was detected (P < 0.05, groups with unequal slopes), the dependent variable was excluded from subsequent analyses because the variable could not be adjusted or corrected for fish length in a consistent manner. Otherwise, the differences that were detected could exclusively be related to fish length, and not to any other stock or environmental effect (Cardinale et al., 2004). When the remaining variables were significantly correlated with fish length, they were standardized or scaled for the length effect using two data scaling methods. One of these approaches is the most widely used common within-group slope method (Campana and Casselman, 1993; Bolles and Begg, 2000; DeVries et al., 2002; Tracey et al., 2006; Burke et al., 2008; Zhang et al., 2014). This method is based on the linear model of Y=a+bX, where the coefficient b is equal to the common within-group slope. Standardization was conducted using the following formula (Bolles and Begg, 2000; Cardinale et al., 2004):
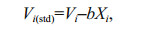
where Vi is the original variable; Vi(std) is the standardized variable; and Xi is the fish length of the ith individual.
The second data scaling method is based on an allometric growth model, Y=aXb, where a and b are constants. After a series of transformations and substitutions, the following data standardization equation was obtained (Lleonart et al., 2000):
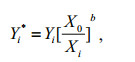
where X0 is the average length of the fish samples.
Therefore, two sets of standardized data were scaled using the two models. To avoid the effect of multicollinearity among the variables and decrease the dimensions of the variables in the subsequent discriminant analysis, principal component analysis (PCA) was performed on the scaled SDs and SIs based on the correlation matrix (Thalib et al., 1999; Yu et al., 2014). Additionally, PCA was conducted on the scaled NEFDs based on the variance-covariance matrix of the coefficients rather than the correlation matrix because the coefficients with low variance and covariance values were of little importance in explaining the observed morphological variation. Several sets of scored variables were produced from the PCA results for the subsequent discriminant analysis.
2.3.2 Canonical discriminant analysis (CDA)As the most commonly employed method for discriminating among groups using multiple observed variables, CDA produces linear combinations that maximize the ratio of 'inter-groups' to 'pooled withingroup' sample variances. A series of CDA trials were conducted on the scaled datasets to test whether the size effect was effectively removed by the two length scaling methods and the NEFDs (Table 3). These trials were performed to realize three specific objectives.
|
The first objective was to determine whether the samples from the different locations could be discriminated on the basis of otolith shape. Samples from the BH2 and CJ3 groups, with identical ranges of fish length and a similar size distribution, were used to realize this goal.
The second objective was to test the effectiveness of the two fish length scaling methods and the NEFDs in removing the size effect. CDA was applied separately to the BH1, BH2 and BH3 groups and the CJ1, CJ3 and CJ5 groups to test the discrimination among the different fish length groups within a single stock, following the scaling of the shape variables with the two methods.
The third objective was to illustrate that otolith shape analysis could produce misleading results in discriminating among stocks, particularly when the size effect was not effectively removed using the two data scaling methods and the NEFDs. Two CDA trials were conducted to discriminate between two interstock length groups. The first trial included the CJ3 and BH1–BH3 groups, whereas the second trial included the BH2 and CJ1–CJ5 groups, among which some groups exhibit the same size but come from different fish stocks (i.e., CJ3 and BH2).
In each CDA, the stepwise method was used to identify the suitable variables for inclusion in the discriminant functions, followed by a jackknife crossvalidation to produce an unbiased estimation of classification success.
All statistical tests were conducted at a significance level of P < 0.05 using SPSS 22 for Macintosh computers.
3 RESULT 3.1 Inter-stock otolith shape variationNo significant difference was found in either fish length (t-test, P>0.50) or the length distribution (KS test, P>0.50) between the BH2 and CJ3 groups. Otolith shape variation caused by fish length can be counteracted to a great extent using samples with similar size distributions. Moreover, the two groups showed an identical length range of 136–140 mm. Therefore, the data were not scaled for between-stock discriminant analysis.
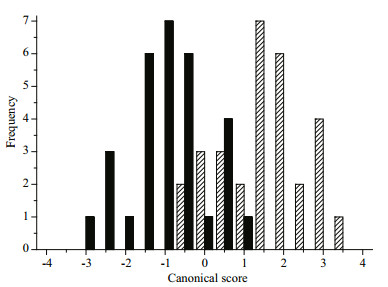
PCA was conducted on the SDs, SIs and NEFDs, and the principle scores were combined and used for CDA. Otolith shape discriminated between the two geographic stocks with an overall classification success rate of 83.3% (Fig. 3). Significant inter-stock otolith variation existed in the two yellow croaker stocks (P < 0.05), which provided a theoretical basis for the follow-up analyses.
|
| Figure 3 Frequency distribution of canonical scores derived from CDA for the BH2 (the solid bars) and CJ3 (the slashed bars) groups |
CDA was applied to the BH (BH1, BH2 and BH3) group and the CJ (CJ1, CJ3 and CJ5) group, respectively. In each analysis, the shape variables were scaled for fish length using the two models.
CDA results indicated that the length groups within the BH or CJ group were well discriminated from each other using both scaling methods (Table 4; Fig. 4).
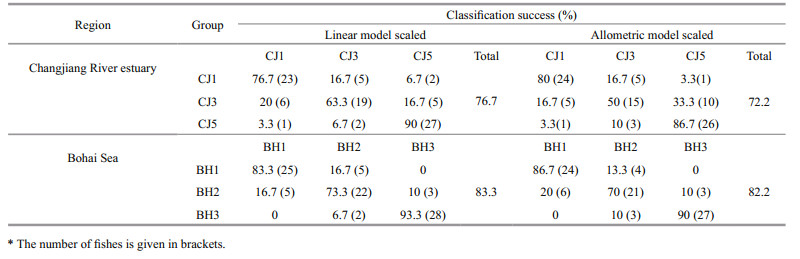
|
|
| Figure 4 Scatter plots showing the CDA scores among within-stock groups: (a) and (b), linear model-scaled; (c) and (d), allometric model-scaled |
In the BH group, CDA of the linearly and allometrically standardized data achieved overall classification success rates of 83.3% (73.3%–93.3%) and 82.2% (70%–90%), respectively (Table 4). For the linearly scaled data, function 1 explained 98.1% of the between-group variance, which was significant for the differentiation of the length groups (Wilks' Lambda=0.152, P < 0.05). Among the 30 individuals in each length group, four BH1 individuals were misclassified into the BH2 group, while five and three BH2 individuals were incorrectly assigned to the BH1 and BH3 groups, respectively. Similarly, using the allometrically scaled data, function 1 explained 98.1% of the between-group variance, which was also significant for the differentiation of the groups (Wilks' Lambda=0.174, P < 0.05). Among the 30 individuals in each length group, four BH1 individuals were incorrectly assigned to the BH2 group; six BH2 individuals to the BH1 group; one BH2 individual to the BH3 group; and three BH3 individuals to the BH2 group.
In the CJ group, CDA of the linearly and allometrically standardized data achieved overall classification success rates of 76.7% (63.3%–90%) and 72.2% (50%–86.7%), respectively (Table 4). For the linearly scaled data, function 1 and function 2 explained 66.6% and 33.4% of the between-group variance, respectively, which was significant for the differentiation of the groups (Wilks' Lambda=0.303, P < 0.05 for function 1; Wilks' Lambda=0.641, P < 0.05 for function 2). Among the 30 individuals in each length group, five and two CJ1 individuals were incorrectly assigned to the CJ3 and CJ5 groups, respectively; six and five CJ3 individuals to the CJ1 and CJ5 groups; one and two CJ5 individuals to the CJ1 and CJ3 groups. Similarly, for the allometrically scaled data, function 1 and function 2 explained 92.4% and 7.6% of the between-group variance, respectively, with both discriminant functions being significant for the differentiation of the groups (Wilks' Lambda=0.321, P < 0.05 for function 1; Wilks' Lambda=0.876, P < 0.05 for function 2). Among the 30 individuals in each length group, five CJ1 individuals were incorrectly assigned to the CJ3 group; three CJ5 individuals to the CJ3 group; and one and three CJ3 individuals to the CJ1 and CJ5 groups, respectively.
3.3 Inter-stock otolith shape variation across different fish length groupsTwo CDA trials were conducted to discriminate among inter-stock length groups. The BH2 group was pooled with the CJ1–CJ5 groups, and the CJ3 group was pooled with the BH1–BH3 groups. Prior to CDA, data scaling and PCA were conducted as described above. The standardized canonical discriminant functions and main components of each principle score used in each CDA trial are listed in Supplementary Table 1.
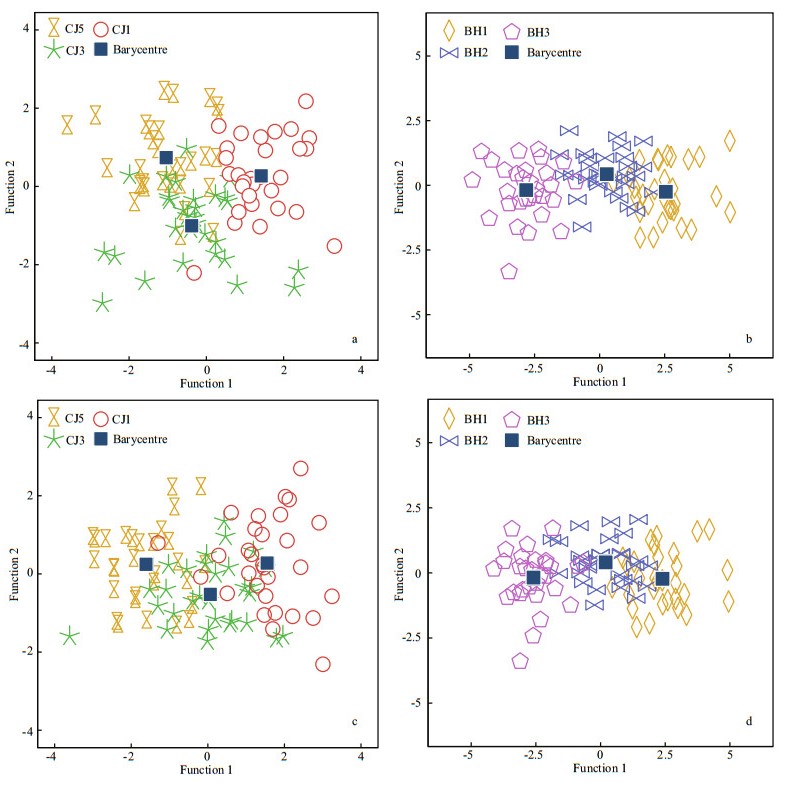
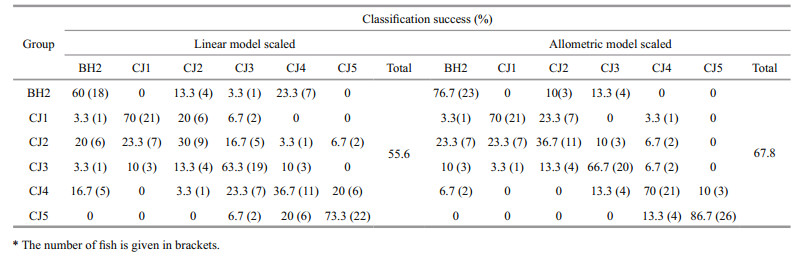
In the BH2 and CJ1–CJ5 groups, the length groups could be separated from each other with modest classification success rates of 55.6% and 67.8% using the linear and allometric scaling methods, respectively, although CJ1–CJ5 were from the same stock (Fig. 5; Table 5). The variation in otolith shape between geographic stocks or between length groups could be observed from the distance between their barycentres in the scatter plots (Fig. 5).
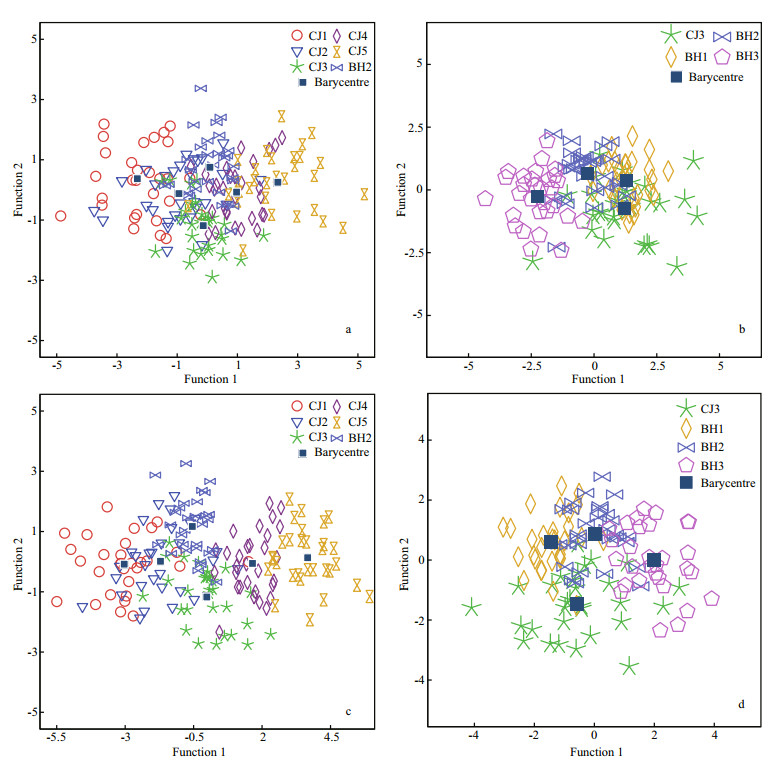
|
| Figure 5 Scatter plots showing the CDA scores across inter-stocks of different fish lengths: (a) and (b), linear model scaled; (c) and (d), allometric model scaled |
|
Similar CDA results were obtained for the CJ3 and BH1-BH3 groups (Fig. 5; Table 6). Using the linear and allometric scaling methods, the two CDA trials achieved modest overall classification successes rate of 62.5% and 67.5%, respectively.
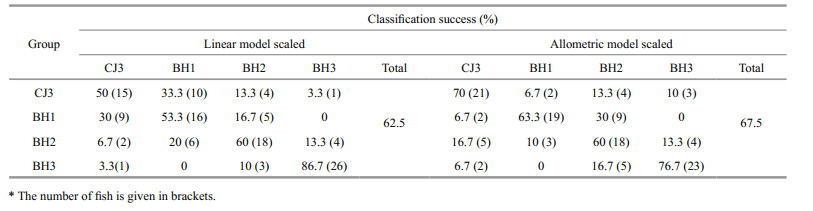
|
The results revealed that inter-stock otolith shape variation might be affected by an interaction with fish length, which led to CDA misclassification when the size effect was not effectively removed. Thus, otolith shape variation caused by fish length could easily result in confusing CDA results.
4 DISCUSSIONOtolith morphology is primarily determined by genetics but is also affected by environmental factors (Begg and Brown, 2000; Cardinale et al., 2004). Therefore, otolith morphometrics are a potential indicator for species or stock discrimination. However, otolith morphology also changes allometrically with fish size, which is one of the decisive factors in the variability in otolith morphometrics, even within a geographic stock (Smith, 1984; Lleonart et al., 2000). The methods for removing the size effect on otolith morphology caused by allometric growth are important and should be cautiously considered when otolith shape analysis is applied to stock discrimination (Cardinale et al., 2004). If the size effect is not taken into account, the validity of the results of morphological analyses of otoliths will be confounded or compromised. The common assumption is that the size effect caused by allometric growth is diminished or even eliminated with application of the appropriate sample selection procedure and statistical analyses (Lleonart et al., 2000). Thus, previous studies have typically attempted to minimize or remove the size effect by restricting fish size to a narrow range in samples evaluated (Neves et al., 2011) and by scaling the variables that are correlated with fish size using either a linear (Bolles and Begg, 2000; Cardinale et al., 2004) or allometric growth model (Lombarte and Lleonart, 1993; Torres et al., 2000).
However, simply minimizing the range of fish sizes in the samples and routinely scaling length data may not always be effective in ensuring that the size effect is removed. A small difference in fish size potentially confounding otolith shape analysis could lead to potential for poor or erroneous stock discrimination. As shown in this study, the intra-stock length groups (with only a 10-mm difference in size) of both the BH and CJ stocks were successfully differentiated from one another based on the otolith shape variables standardized for fish length using both the linear and the allometric scaling methods. This finding suggested that otolith shape variation might exist among the length groups within a geographic stock.
In previous studies, researchers have cautiously attempted to address this problem by selecting samples of similar size during the sample selection step (Castonguay et al., 1991; Campana and Casselman, 1993; Begg and Brown, 2000; DeVries et al., 2002). Some researchers have emphasized that conclusions regarding fish stock structure that are based on otolith shape analysis should be carefully addressed because the discrimination that is achieved could be primarily due to age-or year-class effects, which would therefore highlight sample differences, rather than stock discrimination (Castonguay et al., 1991). Begg and Brown (2000) also suggested that for stock discrimination, otolith shape variables should be recalculated each year for each age group.
To scale otolith shape variables, linear or allometric growth models are commonly used to remove the size effect in studies on otolith shape (Lombarte and Lleonart, 1993; Bolles and Begg, 2000; Torres et al., 2000; Cardinale et al., 2004). Contour morphometry (NEFDs) determined through elliptic Fourier analysis of otoliths is widely used to generate variables that are independent of otolith size variables (Yu et al., 2014). However, the present study demonstrated that these data scaling methods might fail to remove the size effect, as previously suggested, which was clearly shown by CDA of the intra-stock length groups of both geographic stocks. CDA of the inter-stock shape variation of the different length groups (the BH2 group pooled with the CJ groups, or the CJ3 group pooled with the BH groups) further indicated the failure of data scaling to remove the size effect and illustrated how this could result in erroneous stock discrimination. The BH2 and CJ3 groups came from two different geographic stocks with identical fish length ranges and distributions, whereas CJ1–CJ5 and BH1–BH3 each came from their own respective geographic stocks with different fish lengths. From either a statistical or an ecological perspective, intrastock shape variation was reduced when the size effect was effectively removed by length scaling; therefore, CDA should achieve greater classification success in discriminating between inter-stock length groups than among intra-stock groups. However, in both CDA trials, similar or even greater classification success was observed between most of the intra-stock length groups (CJ1–CJ2, CJ4–CJ5; BH1 and BH3) than for the two inter-stock groups with identical fish size (CJ3 and BH2). Additionally, the distance between group centroids in the CDA function plots led to confusing stock discrimination results, which was inconsistent with the results of stock discrimination in previous studies (Ye, 1991; Jin, 1996). These findings suggested that the intra-stock variation in otolith shape caused by fish length could exceed that of the inter-stock geographical separation, even when the otolith shape variables are standardized for fish length using both scaling methods and NEFDs. In this case, data scaling failed to remove the size effect on CDA, which consequently produced a misleading classification in stock discrimination. This finding implied that the length effect might not be effectively removed simply by scaling the data using the two models and NEFDs in either the intra-stock or the inter-stock length groups, particularly when the length variation of the sample groups exceeds a certain level (for yellow croaker, a 10-mm difference in size could not be effectively scaled in the present study).
Nevertheless, data scaling methods and normalized elliptic Fourier coefficients could play an important role in stock discrimination based on otolith shape analysis. Regressions between otolith shape variables (original data and scaled data) and fish length should be conducted to assess the ability of the two scaling methods to select the appropriate shape variables for the subsequent statistical analysis. In this study, all the original shape variables were significantly correlated with fish length and could therefore not be directly included in the subsequent CDA. The allometric model removed the correlation with fish length for all size descriptors (e.g., area, length, width, perimeter, diameter and radius) and shape indices (e.g., circularity, form factor, rectangularity, roundness and ellipticity). In contrast, the linear model removed the correlation of the size descriptors with fish length but failed to achieve the same goal for the shape indices (Supplementary Table 2). Most of the shape indices were proportionally calculated from size descriptors that are generally correlated with fish length. Thus, the linear model did not effectively remove the correlation between the ratios of the size descriptors and fish lengths, leading to exclusion of the shape indices from subsequent statistical analyses of the linearly scaled data. Therefore, the allometric scaling model tended to retain more effective shape variables and, thus, more shape information for subsequent CDA than the linear scaling model, which may affect the results of stock discrimination.
The results indicated the possibility that stock discrimination might be primarily biased by the length effect, which highlighted differences in the samples, rather than stock discrimination, particularly when the size variation of the samples was large relative to fish size. In such cases, data scaling using both growth models and NEFDs may fail to remove the size effect. This problem could result in misleading stock discrimination, particularly in fish species of small size.
5 CONCLUSIONTo help improve the reliability of otolith shape analyses and their ability to discriminate among stocks, a few points should be considered with caution during sample selection and data analysis: (1) even slight variation in length among sample groups can considerably confound the results of otolith shape analysis. Thus, the size variation of samples, both within-and between-groups, must be minimized as much as possible during sample selection; (2) to achieve unbiased discrimination among groups, an equal length distribution of the samples is essential and, to some extent, is more important than a narrow length range or age class. To realize this goal, statistical analysis should be conducted to test the homogeneity of length distributions among sample groups in morphological analyses of otoliths; (3) because many otolith shape variables that are commonly used in otolith shape studies cannot be correctly scaled for the length effect, statistical analysis should be applied to exclude those variables from subsequent statistical analyses for stock discrimination.
Electronic supplementary materialSupplementary material (Supplementary Tables 1–2) is available in the online version of this article at https://doi.org/10.1007/s00343-016-5164-4 .
| Begg G A, Brown R W, 2000. Stock identification of haddock Melanogrammus aeglefinus on Georges bank based on otolith shape analysis. Trans. Am. Fish. Soc., 129(4): 935–945. Doi: 10.1577/1548-8659(2000)129<0935:SIOHMA>2.3.CO;2 |
| Begg G A, Waldman J R, 1999. An holistic approach to fish stock identification. Fish. Res., 43(1-3): 35–44. Doi: 10.1016/S0165-7836(99)00065-X |
| Bentzen P, Taggart C T, Ruzzante D E, Cook D, 1996. Microsatellite polymorphism and the population structure of Atlantic cod (Gadus morhua) in the northwest Atlantic. Can. J. Fish. Aquat. Sci., 53(12): 2706–2721. Doi: 10.1139/f96-238 |
| Bolles K L, Begg G A, 2000. Distinction between silver hake(Merluccius bilinearis) stocks in U. S. waters of the northwest Atlantic based on whole otolith morphometrics.Fish. Bull., 98(3): 451–462. |
| Burke N, Brophy D, King P A, 2008. Shape analysis of otolith annuli in Atlantic herring (Clupea harengus); a new method for tracking fish populations. Fish. Res., 91(2-3): 133–143. Doi: 10.1016/j.fishres.2007.11.013 |
| Campana S E, Casselman J M, 1993. Stock discrimination using otolith shape analysis. Can. J. Fish. Aquat. Sci., 50(5): 1062–1083. Doi: 10.1139/f93-123 |
| Campana S E, 1999. Chemistry and composition of fish otoliths:pathways, mechanisms and applications. Mar.Ecol. Prog. Ser., 188: 263–297. Doi: 10.3354/meps188263 |
| Cardinale M, Doering-Arjes P, Kastowsky M, Mosegaard H, 2004. Effects of sex, stock, and environment on the shape of known-age Atlantic cod (Gadus morhua) otoliths. Can.J. Fish. Aquat. Sci., 61(2): 158–167. Doi: 10.1139/f03-151 |
| Castonguay M, Simard P, Gagnon P, 1991. Usefulness of Fourier analysis of otolith shape for atlantic mackerel(Scomber scombrus) stock discrimination. Can. J. Fish.Aquat. Sci., 48(2): 296–302. Doi: 10.1139/f91-041 |
| Crampton J S, 1995. Elliptic Fourier shape analysis of fossil bivalves:some practical considerations. Lethaia, 28(2): 179–186. Doi: 10.1111/let.1995.28.issue-2 |
| DeVries D A, Grimes C B, Prager M H, 2002. Using otolith shape analysis to distinguish eastern Gulf of Mexico and Atlantic Ocean stocks of king mackerel. Fish. Res., 57(1): 51–62. Doi: 10.1016/S0165-7836(01)00332-0 |
| Dou S Z, Yu X, Cao L, 2012. Otolith shape analysis and its application in fish stock discrimination:a case study. Oceanol. Limnol. Sin., 43(4): 702–712. |
| Guo X P, Jin X S, Dai F Q, 2006. Growth variations of small yellow croaker (Pseudosciaena polyactis Bleeker) in the Bohai Sea. J. Fish. Sci. China, 13(2): 243–249. |
| Jin X S, 1996. Ecology and population dynamics of small yellow croaker (Pseudosciaena polyactis Bleeker) in the Yellow Sea. J. Fish. Sci. China, 3(1): 32–46. |
| Legua J, Plaza G, Perez D, Arkhipkin A, 2013. Otolith shape analysis as a tool for stock identification of the southern blue whiting. Micromesistius australis. Lat. Am. J. Aquat.Res., 41(3): 479–489. |
| Lleonart J, Salat J, Torres G J, 2000. Removing allometric effects of body size in morphological analysis. J. Theor.Biol., 205(1): 85–93. Doi: 10.1006/jtbi.2000.2043 |
| Lombarte A, Lleonart J, 1993. Otolith size changes related with body growth, habitat depth and temperature. Environ.Biol. Fishes, 37(3): 297–306. Doi: 10.1007/BF00004637 |
| Longmore C, Fogarty K, Neat F, Brophy D, Trueman C, Milton A, Mariani S, 2010. A comparison of otolith microchemistry and otolith shape analysis for the study of spatial variation in a deep-sea teleost, . Coryphaenoides rupestris.Environ. Biol. Fishes, 89(3-4): 591–605. Doi: 10.1007/s10641-010-9674-1 |
| Meng Z N, Zhuang Z M, Jin X S, Tang Q S, Su Y Q, 2003. Genetic diversity in small yellow croaker (Pseudosciaena polyactis) by RAPD analysis. Biodiv. Sci., 11(3): 197–203. |
| Neves A, Sequeira V, Farias I, Vieira A R, Paiva R, Gordo L S, 2011. Discriminating bluemouth, Helicolenus dactylopterus (Pisces:Sebastidae), stocks in Portuguese waters by means of otolith shape analysis. J. Mar. Biol.Assoc. U. K., 91(6): 1237–1242. Doi: 10.1017/S002531541000189X |
| Smith R J, 1984. Allometric scaling in comparative biology:problems of concept and method. Am. J. Physiol., 246(2): R152–R160. |
| Smith S J, Campana S E, 2010. Integrated stock mixture analysis for continous and categorical data, with application to genetic-otolith combinations. Can. J. Fish.Aquat. Sci., 67(10): 1533–1548. Doi: 10.1139/F10-078 |
| Thalib L, Kitching R L, Bhatti M I, 1999. Principal component analysis for grouped data-A case study. Environmetrics, 10(5): 565–574. Doi: 10.1002/(ISSN)1099-095X |
| Thresher R E, 1999. Elemental composition of otoliths as a stock delineator in fishes. Fish. Res., 43(1-3): 165–204. Doi: 10.1016/S0165-7836(99)00072-7 |
| Torres G J, Lombarte A, Morales-Nin B, 2000. Sagittal otolith size and shape variability to identify geographical intraspecific differences in three species of the genus Merluccius. J. Mar. Biol. Assoc. U. K., 80(2): 333–342. Doi: 10.1017/S0025315499001915 |
| Tracey S R, Lyle J M, Duhamel G, 2006. Application of elliptical Fourier analysis of otolith form as a tool for stock identification. Fish. Res., 77(2): 138–147. Doi: 10.1016/j.fishres.2005.10.013 |
| Tuset V M, Lombarte A, González J A, Pertusa J F, Lorente M, 2003. Comparative morphology of the sagittal otolith in Serranus spp. J. Fish Biol., 63(6): 1 491–1504. Doi: 10.1111/jfb.2003.63.issue-6 |
| Xiao Y S, Zhang Y, Gao T X, Yanagimoto T, Yabe M, Sakurai Y, 2009. Genetic diversity in the mtDNA control region and population structure in the small yellow croaker Larimichthys polyactis. Environ. Biol. Fishes, 85(4): 303–314. Doi: 10.1007/s10641-009-9497-0 |
| Xu Z L, Chen J J, 2010. Population division of Larimichthys polyactis in China Sea. Chin. J. Appl. Ecol, 21(11): 2856–2864. |
| Ye C C. 1991. Small yellow croaker (Larimichthys polyactis).In:Deng J Y, Zhao C Y eds. Marine Fisheries Biology.China Agriculture Press, Beijing, China. p.164-200. (in Chinese) |
| Yu X, Cao L, Liu J H, Zhao B, Shan X J, Dou S Z, 2014. Application of otolith shape analysis for stock discrimination and species identification of five goby species (Perciformes:Gobiidae) in the northern Chinese coastal waters. Chin. J. Oceanol. Limnol., 32(5): 1060–1073. Doi: 10.1007/s00343-015-4022-0 |
| Zhang C, Ye Z J, Wan R, Ma Q Y, Li Z G, 2014. Investigating the population structure of small yellow croaker(Larimichthys polyactis) using internal and external features of otoliths. Fish. Res., 153: 41–47. Doi: 10.1016/j.fishres.2013.12.012 |
 2017, Vol. 35
2017, Vol. 35