Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZHANG Xiaolin(张晓林), LI Nan(李楠), QIN Ting(秦婷), HUANG Bei(黄贝), NIE Pin(聂品)
- Involvement of two glycoside hydrolase family 19 members in colony morphotype and virulence in Flavobacterium columnare
- Chinese Journal of Oceanology and Limnology, 35(6): 1511-1523
- http://dx.doi.org/10.1007/s00343-017-6160-z
Article History
- Received Jun. 3, 2016
- accepted in principle Jul. 14, 2016
- accepted for publication Sep. 3, 2016
2 University of Chinese Academy of Sciences, Beijing 100049, China;
3 College of Fisheries, Jimei University, Xiamen 361021, China
Flavobacterium columnare, the causative agent of columnaris disease, infects a wide range of freshwater fish in aquaculture and natural habitats, ornamental freshwater fish in aquaria can also be infected with the bacterium (Declercq et al., 2013). The bacterial infection normally causes skin and fin lesions, and gill necrosis, resulting in high mortality and severe economic losses in the aquaculture industry. Bacterial adhesion to the skin and gills is considered an important, initial step in F. columnare infection in fish (Decostere et al., 1999); Chondroitin lyases and several other extracellular proteases are considered virulence factors of the bacterium (Stringer-Roth et al., 2002; Xie et al., 2004, 2005; Suomalainen et al., 2006; Beck et al., 2015). However, it was not until very recently that the pathogenesis of two chondroitin lyase-encoding genes, cslA and cslB, was investigated through mutation in F. columnare with the development of an in-frame gene deletion system (Li et al., 2015).
In a recent report by Kharade and McBride (2014), a chitinase gene, chiA, was found in F. johnsoniae also in the genus Flavobacterium, whose encoded protein is secreted through a type IX secretion system and can digest chitin in culture conditions. Chitinases are glycosyl hydrolases mainly in glycoside hydrolase families 18 and 19, with either a conserved Glyco_ hydro_18 domain (GH18 domain) or Glyco_ hydro_19 domain (GH19 domain), respectively. Chitinase activity is also observed in the protein families GH20 and GH48 (Kubota et al., 2004; Fujita et al., 2006). Chitinases can degrade glyosidic bonds in chitin, a polymer of β-1, 4-linked N-acetyl glucosamine (Rinaudo, 2006; Hoell et al., 2010; Beier and Bertilsson, 2013). Chitin is abundant in nature and is a common element in the external structures of organisms, e.g., insect cuticles, crustacean shells, fish scales, and fungi cell walls (Hackman, 1962; Rinaudo, 2006; Kumari and Rath, 2014). Chitinases are present in a wide range of organisms, such as bacteria (Gooday, 1990), some algae (e.g. Vrba et al., 1996), and rotifers (Štrojsová and Vrba, 2005), and may be involved in the pathogenesis of bacterial and fungal pathogens (Chaudhuri et al., 2013; Staats et al., 2013).
In an effort to understand the pathogenesis of columnaris disease, the whole F. columnare strain G4 (unpublished data) genome was searched for possible chitinase genes. Bioinformatic analysis revealed two proteins, which contain a GH19 domain, these were named GH19 domain containing protein-1 and -2 (Ghd-1 (GenBank accession number KT288110) and Ghd-2 (KT288111)). Ghd-1 also has a predicted N-terminal signal peptide and a predicted C-terminal sorting domain, which is a C-terminal signal essential for translocation of the protein across the outer membrane via the T9SS (McBride and Zhu, 2013). Ghd-1 and ghd-2 may encode chitinases and be F. columnare virulence genes. To understand the function of ghd-1 and ghd-2, single-and double-gene deletion mutants were constructed using the gene deletion system as reported in a previous study by Li et al. (2015). However, F. columnare G4 could not degrade the colloidal chitin (unpublished data), which was unexpected. This finding was in contrast to that in another species in the same genus, F. johnsoniae (Kharade and McBride, 2014). Thus, the ghd-1 and ghd-2 genes have no chitin degrading capacity. But the mutants exhibited some specific growth, colony morphotype, and virulence characteristics. The differences in growth and colony morphotypes, skimmed milk degradation, and virulence against zebrafish between these mutants are reported here.
2 MATERIAL AND METHOD 2.1 Bioinformatic analysisThe whole F. columnare virulent strain G4 genome (unpublished data) was analyzed for possible chitinase genes; two genes (GenBank accession numbers KT288110 and KT288111) containing Glyco_ hydro_19 domain (GH19), which is conserved in chitinases, were identified. The nucleotide sequences of these two genes were translated to amino acids (aa) using ExPASy translate tools (http://web.expasy.org/translate/). The conserved domains were searched using the NCBI conserved domains searching tools (http://www.ncbi.nlm.nih.gov/Structure/cdd/wrpsb.cgi), and the presence and location of a signal peptide of the deduced aa were predicted with SignalP 4.1 Server (http://www.cbs.dtu.dk/services/SignalP/). Multiple alignments of aa sequences were performed with the ClustalW2 program (http://www.ebi.ac.uk/Tools/clustalw2/). The similarity of the nucleotide sequences and deduced aa sequences were determined in BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi).
2.2 Determination of F. columnare G4 chitin degradationColloidal chitin was prepared according to Murthy and Bleakley (2012). In brief, 2 L concentrated HCl was added to 100 g crude chitin (Sigma-Aldrich, USA) and agitated for 24 h at 4℃, before being filtered through glass wool. The colloidal chitin was precipitated by adding 18 L distilled water at room temperature. After centrifugation, the precipitate was resuspended with 2 L distilled water. The pH value was then adjusted to 7 with 5 N NaOH. The autoclaved colloidal chitin was stored at 4℃ until use. Next, 1% (v/v) colloidal chitin was added to either modified Shieh agar (0.25 g peptone and 0.025 g yeast extract per 100 mL Shieh medium), Shieh agar without peptone and yeast extract in the chitin degradation experiment, or MYA agar (MgSO4 0.5 mmol/L, FeSO4 0.05 mmol/L, EDTA 0.04 mmol/L, potassium phosphate (pH 7.3) 20 mmol/L, yeast extract 0.1 g, agar 15 g, ddH2O 1 L) (Kharade and McBride, 2014). Wild type strains were cultured and adjusted to an OD600 value of 0.4 (4×108 colony forming units (CFU)/mL) and 10 μL (4×106 CFU/mL) bacterium was spotted on colloidal chitin plates and cultured at 28℃ for 60 h, before being observed for the formation of possible clear zones around the colony.
2.3 Construction of mutants and complementation strainsThe F. columnare G4 strain, which was isolated from the grass carp Ctenopharyngodon idella during an outbreak of gill-rot disease (Lu et al., 1975), was used as the wild type and to create mutants in the present study. F. columnare strains were grown at 28℃ in either Shieh medium or agar as previously described (Shieh, 1980). Two GH19 domain containing genes, ghd-1 and ghd-2, were found in the F. columnare G4 genome sequence data (unpublished data). To obtain deletion mutants of both genes, the shuttle plasmid pCP23 derived from F. psychrophilum D12 plasmid pCP1 (Alvarez et al., 2004) and the suicide plasmid vector pMS75 created in a previous study (Li et al., 2015) were employed. The ghd-1 and ghd-2 in-frame deletion mutants were generated through sacB-based exchange (Edwards et al., 1998). The insertion plasmid pMS75-ghd1 was generated and transferred into S17-1 λpir to conjugate with wild type strain G4, and the ghd-1 deletion mutant, Δghd-1, was then obtained.
The ghd-2 gene deletion mutant, Δghd-2, was obtained by the same method. The double mutant strain Δghd-1 Δghd-2 was obtained by transferring pMS75-ghd1 into the mutant strain Δghd-2. The deleted sequence base number must be trinary in all mutant strains to prevent frame shifting in the following sequences. The SacB gene was used as a counterselectable marker (Gay et al., 1983) to screen negative clones that did not grow on Shieh agar with 10% sucrose (Sigma-Aldrich).
The complementation strains of each mutant strain were constructed based on the plasmid pCP23 as indicated above. For the complementation of ghd-1, a 1.7 kb fragment containing the ribosome-binding site was amplified using the primers ghd1-comp-For (with KpnI site) and ghd1-comp-Rev (with BamHI site). This fragment was introduced into complementation vector pCP23, which was digested beforehand with KpnI and BamHI. The constructed plasmid pCP23-ghd1-comp was verified by sequencing before being transferred into E. coli S17-1 λpir to conjugate with the mutant strain Δghd-1. The Δghd-1, Δghd-1-comp complementation was then obtained. The positive clones were verified by PCR using the primers PompAKpnF and oriTR. The Δghd-2 and Δghd-1 Δghd-2 complementation was also performed using this method. The bacteria and plasmids used in this study are listed in Table 1, and the primers used in the construction of mutant and complementation strains are listed in Table 2.
To obtain extracellular protein, the wild type, mutant, and complementation strains were grown to mid-log phase (OD600=0.4) in Shieh broth at 28℃ with shaking. A 20-mL bacterial suspension (8×109 CFU/mL) was pelleted by centrifugation at 4 000×g for 10 min, and the supernatant was filtered through 0.22 μmol/L polyvinylidene difluoride filters (Merck Millipore, Germany) to remove residual cells. The filtered supernatant was concentrated with an Amicon Ultra-15 centrifugal filtration device with a 30-kDa-molecular-mass-cutoff filter (Millipore) to a final volume of 100 μL, 20 μL of which was then separated by SDS-PAGE and transferred onto a PVDF membrane before being probed with affinity-purified antibodies (developed with the antigen peptide ASTEYPANPTKGYY for Ghd-1 and QADNSTGKQYSKKE for Ghd-2 by GenScript, Nanjing, China; all diluted at 1:1 000), and goat antirabbit IgG antibody was then conjugated with peroxidase (Sigma) (diluted at 1:5 000). Afterwards, the membrane was stained with ImmobilonTM Western Chemiluminescent HRP Substrate (Millipore) and examined under the ECL Western blot system (BioRad, USA).
2.5 Determination of growth rate and saccharide and protein substrate degradation analysisA single colony of each mutant strain was grown overnight in Shieh broth at 28℃ with shaking. The culture was then diluted 1:100 into fresh Shieh broth for culture at 28℃ and rotated at 180 r/min. Growth rate was determined every hour by measuring OD600 for a period of 10 h with a spectrophotometer (Eppendorf Biophotometer, Germany).
Three kinds of saccharides, including glucose, sucrose, and starch (Sinopharm, Shanghai, China), were added to Shieh agar at a concentration of 1%, 1%, and 0.2% (m/v) in Shieh agar, respectively, to observe saccharide degradation for the wild type and mutants. We also used 2% (m/v) skimmed milk (Sinopharm) as a substrate in the analysis.
2.6 Determination of median lethal dose (LD50) and zebrafish survivorshipZebrafish (Danio rerio) from a quarantined source in the China Zebrafish Resource Center at the same institute were employed for the LD50 determination to ensure that they were uninfected. The wild type G4 and mutant strains including rhizoid colony strains, Δghd-1, Δghd-2, and Δghd-1 Δghd-2, and non-rhizoid colony strains, NΔghd-1, NΔghd-2, and NΔghd-1 Δghd-2 were cultured separately in Shieh broth at 28℃ overnight. The cultures were diluted 1:100 and grown to the mid-log phase (OD600=0.4). Immersion infection of zebrafish with each strain was performed by immersing the fish in 1 000-, 10 000-, and 100 000-fold dilution of the original culture with 30 individuals in each group for 30 min. The fish were then transferred into clean water and mortality and disease symptoms were recorded every 6 h for 7 days. To enumerate the viable cells in each original culture, a tenfold serial dilution for each strain was made and 100 μL of the final three dilutions (10-4, 10-5, and 10-6) was transferred to duplicate Shieh agar plates and spread evenly over the entire surface of the plate. All of the plates were incubated at 28℃ and the colonies that had grown on each plate were counted after 48 h. The LD50 value of each strain was calculated using the statistical approach of Reed and Muench (1938). The formula used in the LD50 calculation was:

where, Xk is the logarithm of maximum concentration, d is the logarithm differentials between adjacent concentrations, and ∑Pi is sum of mortalities of different concentration groups.
To compare the survivorship of fish in each group, the survival curve of each strain was determined within seven days after infection. Two-hundred and ten zebrafish were divided into seven groups, and each group with 30 fish was reared in a 10-L plastic tank with aeration and maintained at 25±1℃ for one week before infection. The fish were fed once a day with artificial feed during the acclimatization and approximately 3 L water was changed daily following feeding. F. columnare G4 wild type and mutant strains were cultured separately overnight in Shieh broth at 28℃. The cultures were diluted 1:100 and grown to the mid-log phase (OD600=0.4). Zebrafish were immersed in a 10, 000-fold dilution for 30 min with each bacterial strain and zebrafish survival rate was recorded daily over a 7-day period. The concentration of viable cells in each original culture was determined by making a serial dilution and spreading 100 μL of each dilution on the Shieh agar plates. All of the remaining fish were euthanized.
All animal experiments were conducted according to the principles and procedures of the laboratory animal cares approved by the institute.
2.7 Transcriptome analysis of mutants with rhizoid and non-rhizoid coloniesThe wild type and mutant strains were inoculated into Shieh broth and shaken at 28℃ until the OD600 value had reached 0.4. The bacterial cells were collected by centrifugation at 4 000×g for 10 min. Total RNA was extracted separately from the wild type and mutant strains using TRIzol® Reagent (Invitrogen, USA) according to the manufacturer's protocol. The rRNA was removed with a Ribo-Zero Magnetic Kit (G-Negative Bacteria, EpiCentre, USA) after total RNA collection. The quality of total RNA samples was checked using a NanoDrop2000 (Thermo Fisher, USA) and agarose gel electrophoresis. Approximately 5 μg total RNA from each strain for reciprocal crosses were sent to Majorbio-Shanghai for transcriptome library preparation, and the libraries were sequenced on separate plates, one library per plate, using an Illumina HiSeq 2500 system. The unigene expression was calculated in FPKM (reads per kilobase (kb) of exon model per million mapped reads) (Mortazavi et al., 2008), which can eliminate the influence of different gene length and sequencing discrepancies in the gene expression calculation. Therefore, the differences in gene expression among samples can be compared. The differentially expressed genes (DEGs) were screened out using the software EdgeR (http://www.bioconductor.org/packages/2.12/bioc/html/edgeR.html ) on the basis of FDR < 0.05, |logFC|>=1. The gene ontology (GO) function annotation and KEGG (Kyoto encyclopedia of genes and genomes) pathway analysis were carried out using Blastall software against the GO (http://www.geneontology.org/ ) and KEGG (http://www.genome.jp/kegg/) databases.
2.8 RNA extraction, cDNA synthesis, and quantitative real-time PCR (qRT-PCR) validationThree-mL bacterial suspensions (1.2×109 CFU/ mL) of wild type and each rhizoid and non-rhizoid mutant strains were used separately for RNA extraction. Total RNA was obtained using an RNeasy Mini Kit (Qiagen, Germany) according to the manufacturer's protocol. Each RNA sample was treated with RNase-free DNase (TaKaRa, Dalian, China) following the manufacturer's instructions to remove residual genomic DNA (gDNA). DNasetreated RNA (1 mg) was subjected to reverse transcriptase reactions using oligo-dT primer and PrimeScript Reverse Transcriptase (TaKaRa) according to the manufacturer's protocol.
F. columnare 16S rDNA was used as an internal gene. The qRT-PCR was performed using iQTM SYBR Green Supermix (Bio-Rad, Singapore) on a Bio-Rad CFX96 Real-Time System. The relative expression of target genes was normalized to the expression of the 16S rDNA gene. Each non-rhizoid strain sample was compared to a rhizoid strain sample to calculate the fold changes, using the comparative Ct method (2-∆∆Ct). The primer sequences are listed in Table 2.
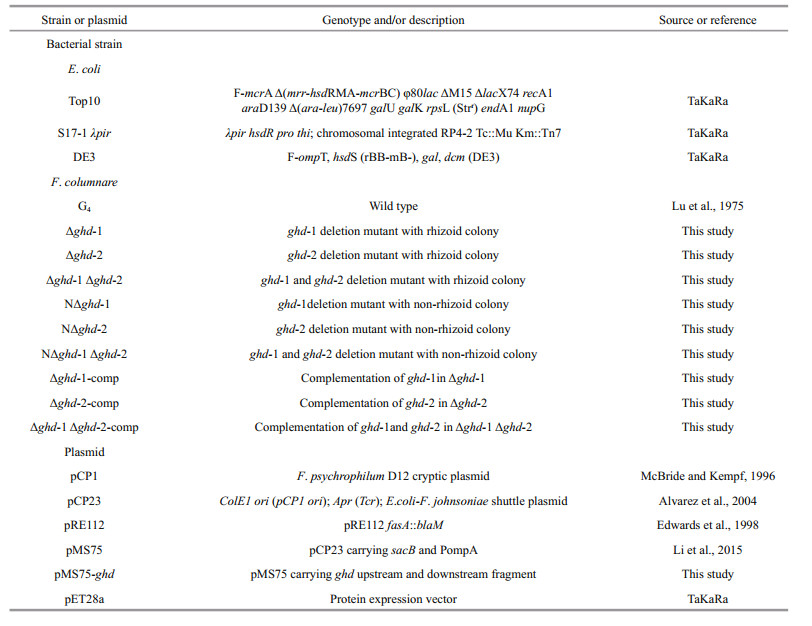
In the F. columnare G4 strain, two genes (GenBank accession numbers: KT288110 and KT288111) containing GH19 domains were identified. One of them, ghd-1 (KT288110) was 100% identical to the gene FCOL_10530, which has been annotated as chitinase in the F. columnare strain ATCC49512; its open reading frame (ORF) was 1 356 bp coding for 451 aa with a signal peptide located at the first 23 aa at the N-terminal and a Por_Secre_tail domain located at the last 68 aa at the C-terminal, which indicates that the protein may be secreted through the type IX secretion system (T9SS), a novel protein secretion system found in members of the phylum Bacteroidetes (McBride and Zhu, 2013) (Figs. 1a, S1, and Table S1). However, the other gene, ghd-2 (KT288111), encodes a putative protein with only a GH19 domain and neither a signal peptide at the N-terminal nor a Por_ Secre_tail domain at the C-terminal, indicating a possible cytoplasmic protein feature in the G4 strain (Fig. 1a). Additionally, ghd-2 was not homologous to any sequences in the F. columnare ATCC49512 genome (Fig.S1).
|
| Figure 1 Identification of Glyco_hydro_19 (GH19) domain containing proteins, Ghd-1 and Ghd-2 (a) in F. columnare G4, examination of flavobacterial chitin degradation ability, (b) and ghd-1 and ghd-2 gene expression (c) a. comparison of Glyco_hydro_19 and Glyco_hydro_18 (GH18) domain containing proteins between Ghd-1 and Ghd-2 in F. columnare G4, and predicted chitinase encoded by FCOL_10530 in F. columnare ATCC 49512, chitinase ChiA in F. johnsoniae, and chitinase C-1 in Streptomyces griseus. SP: signal peptide; GH19 domain: Glyco_hydro_19 domain; GH18 domain: Glyco_hydro_19 domain; CTD: C-terminal domain involved in secretion by the type IX secretion system; CBD: chitin-binding domain. An uppercase letter followed by a number indicates the first and last amino acids of each signal peptide and predicted domain; b. F. johnsoniae UW101 degraded chitin but F. columnare G4 did not. Approximately 103 F. columnare G4 and F. johnsoniae UW101 cells were spotted on the MYA-chitin solid medium for 60 h incubation at 28℃. No clear circle was observed surrounding the G4 strain colony; a clear zone surrounded the F. johnsoniae UW101 colony (red circle); c. both ghd-1 and ghd-2 were transcribed in F. columnare G4. Reverse transcription-PCR was performed using RNA extracted from F. columnare G4 as a template. The target bands of 2 393 bp for ghd-1 and 1 637 bp for ghd-2 were visualized under UV light. M represents a nucleic acid marker with the size indicated on the left. |
Based on the bioinformatics analysis, an attempt was made to examine F. columnare G4 chitin degradation capability on solid medium with colloidal chitin as either the only or primary carbon source. Surprisingly, the G4 strain was not use chitin effectively because no clear circle formed around the G4 strain colony; however, an obvious clear zone was observed around F. johnsoniae UW101 on the same plate (Fig. 1b). Reverse transcription PCR (Fig. 1c) and western blot analysis (Fig. 2a and b) were carried out to examine whether or not the two GH19 domain protein containing genes were transcribed. The ghd-1 and ghd-2 genes were indeed transcribed normally, and the former, which encodes a protein with a Por_ Secre_tail domain, could even be detected in the extracellular milieu, indicating its secretory feature (Fig. 2c). The normal expression of these two genes in F. columnare G4 may indicate that their encoded products have no function in chitin degradation.
|
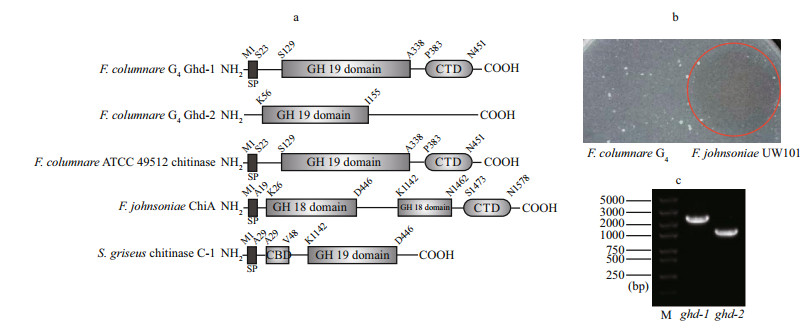
| Figure 2 wo single-gene deletion mutants, Δghd-1, Δghd-2, and the double-gene deletion mutant, Δghd-1 Δghd-2 were verified by PCR (a) and western blot analysis (b) M lane represents a nucleic acid marker with the size indicated on the left; G4: F. columnare G4 wild type strain; a 1 056-bp deletion was detected in Δghd-1 and Δghd-1 Δghd-2, and an 870 bp deletion in Δghd-2 and Δghd-1 Δghd-2. Extracellular products (ECP) of wild type G4, single-gene mutant Δghd-1 and its complementary strains Δghd-1-comp, double-gene mutant Δghd-1 Δghd-2 and its complementary strain Δghd-1 Δghd-2-comp. The anti-GH19 domain containing protein Ghd-1 antibody (anti-Ghd-1) was used for the western blot analysis. The arrow indicates a 49 kDa band. |
To investigate the functions of ghd-1 and ghd-2, the single-and double-deletion mutants were constructed using the suicide plasmid pMS75 and were verified by PCR (Fig. 2a), the corresponding deletion mutants were named Δghd-1, Δghd-2, and Δghd-1 Δghd-2. A 1 056-bp deletion was detected in mutant Δghd-1 and an 870 bp in mutant Δghd-2 compared to the wild type G4 (Fig. 2a); a similar result was observed in the double mutant Δghd-1 Δghd-2 (Fig. 2a). Meanwhile, the G4 extracellular proteins, Δghd-1, Δghd-1-comp, Δghd-1 Δghd-2, and Δghd-1 Δghd-2-comp, were concentrated and analyzed by western blot. No specific Ghd-1 bands were detected in the mutants Δghd-1 and Δghd-1 Δghd-2 (Fig. 2b), revealing the successful deletion of ghd-1 from the wild type F. columnare G4. The presence of a signal peptide in Ghd-1 and the presence of Ghd-1 in extracellular proteins may indicate that Ghd-1 is a secretory protein. However, Ghd-2 was not detected in the extracellular proteins (data not shown), which, in light of the absence of a signal peptide, may indicate that Ghd-2 is not secretory, but can be expressed at the transcription level (Fig. 1c).
3.2 Rhizoid and non-rhizoid colonies in Δghd-1, Δghd-2, and Δghd-1 Δghd-2The wild type and mutant bacterial cells were diluted at the log phase and inoculated on Shieh agar plates. After incubation for 48 h at 28℃, the wild type and mutant colony shapes were observed under a stereomicroscope. As shown in Fig. 3, F. columnare wild type G4 is a typical rhizoid, spreading colony. Surprisingly, both rhizoid and non-rhizoid colonies were observed for the single-gene mutants, Δghd-1 (Fig. 3a and b) and Δghd-2 (Fig. 3c and d), and the double-gene mutant, Δghd-1 Δghd-2 (Fig. 3e and f). The non-rhizoid colonies were obviously smaller and thicker than the rhizoid, spreading colonies, and were named NΔghd-1, NΔghd-2, and NΔghd-1 Δghd-2.

|
| Figure 3 Rhizoid and non-rhizoid colony morphotypes of single-and double-gene deletion mutants of ghd-1 and ghd-2 in Flavobacterium columnare A bacterial dilution was incubated on a Shieh plate for 24 h until a single colony was seen and then photographed under a stereomicroscope. a, b. rhizoid and non-rhizoid ghd-1 gene mutants, Δghd-1 and NΔghd-1; c, d. Δghd-2 and NΔghd-2; e, f. Δghd-1 Δghd-2 and NΔghd-1 Δghd-2; g. F. columnare wild type G4 strain. Scale bar=1 mm. |
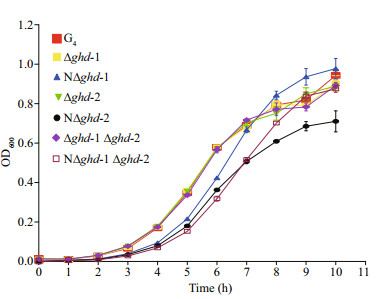
The wild type G4 strain and mutant growth rates, including the rhizoid and non-rhizoid colonies, were examined in Shieh medium (Fig. 4). The shapes of the growth curves were very similar (Fig. 4), implying that the wild type and all of the mutants exhibited a similar pattern of growth in the medium. However, after statistical analysis (one-way ANOVA, P < 0.05), two strains differed significantly from the others in terms of growth rate: NΔghd-1 Δghd-2 grew significantly slower than the wild type and the other mutants, except after a seven hour culture, and NΔghd-2 had a significantly lower growth rate 5–10 hours into the culture (Fig. 4). However, NΔghd-1 grew significantly faster in the later culture stages (Fig. 4).
|
| Figure 4 Growth curves of single-and double-gene deletion mutants of ghd-1 and ghd-2 in Flavobacterium columnare Bacteria were grown in Shieh medium at 28℃ for 10 h, and the growth rate was determined for each strain every hour by measuring OD600. G4 represents the wild type. Δghd-1, Δghd-2, NΔghd-1, and NΔghd-2, represent rhizoid and non-rhizoid ghd-1 and ghd-2 mutants, respectively. Δghd-1 Δghd-2, and NΔghd-1 Δghd-2 represent the rhizoid and non-rhizoid double-gene mutants. |
As no chitinase degradation activity was detected in the wild type, other saccharides including glucose, sucrose, starch, and skimmed milk were tested in the substrate degradation analysis. No difference was observed in the degradation of glucose, sucrose, and starch (data not shown). However, a larger and wider transparent circle was observed in the skimmed milk degradation for the non-rhizoid colony strain, NΔghd-2, when compared with the wild type G4 strain, rhizoid, and other non-rhizoid strains (Fig. 5).
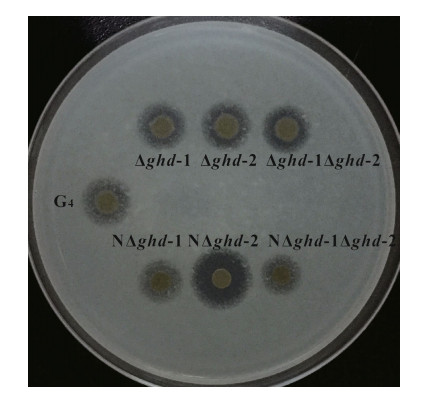
|
| Figure 5 Skimmed milk degradation in rhizoid and non-rhizoid colony morphotype strains of two single-and doublegene ghd-1 and ghd-2 mutants in Flavobacterium columnare G4 Mutants with rhizoid and non-rhizoid colony morphotypes are indicated as in Fig. 4. |
Mutant virulence was measured in zebrafish, and the LD50 values of the wild type G4 and the mutants Δghd-1, Δghd-2, Δghd-1 Δghd-2, NΔghd-1, NΔghd-2, and NΔghd-1 Δghd-2 were 104.917, 105.162, 105.138, 105.013, 105.283, 105.517, and 105.492, respectively. The non-rhizoid mutants NΔghd-1, NΔghd-2, and NΔghd-1 Δghd-2 had higher LD50 values when compared with the wild type G4 and other rhizoid mutants.
Furthermore, zebrafish survivorship was determined by infection with the wild type G4 and mutant strains at a concentration of 4×105 CFU/mL (Fig. 6). The wild type G4 strain caused rapid death, with all fish dying within 3 days post-infection (dpi). Following infection with the mutant rhizoid colony strains, Δghd-1, Δghd-2, and Δghd-1 Δghd-2, zebrafish survival rate decreased and all of the fish died at 4 dpi. However, fish infected with NΔghd-1, NΔghd-2, and NΔghd-1 Δghd-2 exhibited higher survival rates, and at 7 dpi the survival rate was approximately 10% for NΔghd-1 and NΔghd-1 Δghd-2 infected fish. The survival rate was even higher in fish infected with the non-rhizoid mutant NΔghd-2, with a 50% survival rate at the end of the experiment (Fig. 6).
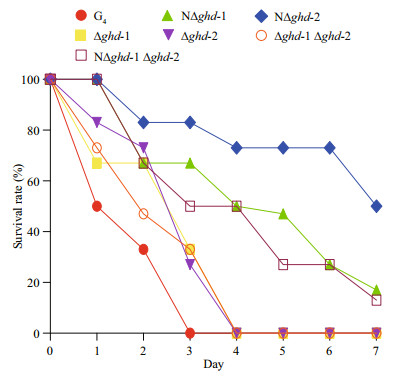
|
| Figure 6 Survivorship of zebrafish infected with F. columnare wild type G4 and its ghd-1 and ghd-2 deletion mutants Thirty zebrafish were immersed in diluted bacterial culture (4×105 CFU/mL) for 30 min and then transferred into clean water. Mortality was recorded every 24 hours for 7 days and zebrafish survival rate was calculated daily. Mutants with rhizoid and nonrhizoid colony morphotypes are indicated as in Fig. 4. |
In the non-rhizoid mutants, NΔghd-1, NΔghd-2, and NΔghd-1 Δghd-2, a total of 26 (13+9+1+4), 60 (40+0+16+4), and 71 (42+9+16+4) up-regulated and 56 (2+18+1+35), 96 (54+1+6+35), and 83 (24+18+6+35) down-regulated genes were detected when compared with the rhizoid mutants, Δghd-1, Δghd-2, and Δghd-1 Δghd-2, respectively (Fig. 7; Table S2). Two up-and two down-regulated genes were further verified through qRT-PCR (Fig.S2). It is obvious that more down-regulated genes were observed in non-rhizoid mutants, and NΔghd-2 had the most down-regulated genes (Fig. 7). Overall, 35 down-regulated and four up-regulated genes were common in all three non-rhizoid mutants.
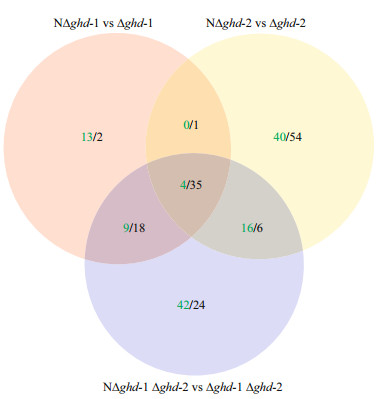
|
| Figure 7 Venn map comparison of differentially expressed genes between non-rhizoid and rhizoid colony morphotype strains in Δghd-1, Δghd-2, and Δghd-1 Δghd-2 from Flavobacterium columnare G4 Numbers in red and black indicate up-regulated and down-regulated genes in non-rhizoid strains, respectively. Mutants with rhizoid and non-rhizoid colony morphotypes are indicated as in Fig. 4. |
However, most of the differentially expressed genes detected were hypothetical proteins (Table S2), which reflects the current lack of data on F. columnare, despite its importance in aquaculture. Among the 35 down-regulated expression genes, 24 encode hypothetical proteins, the others encode phage tail proteins, rearrangement hotspot (rhs) element Vgr protein, TonB-dependent outer membrane receptor precursor, glycosyl transferase family 2, glycosyl hydrolase, metalloprotease, ATPase AAA, and thiolactivated cytolysin. In all of the annotated genes among the three comparisons, metabolic processes, cellular processes, and single-organism processes were commonly identified (data not shown); metabolic pathways for the enrichment of nitrogen metabolism, and genetic information processing were the most common functional representatives (data not shown).
4 DISCUSSIONUsing a newly developed gene deletion strategy, two F. columnare chondroitin lyase-encoding genes were examined for chondroitin sulfate A digestion and virulence, providing the first report on the pathogenesis of possible virulence factors in the bacterium (Li et al., 2015). In the present study, F. columnare G4 ghd-1 and ghd-2 were deleted separately and in combination from the bacterium. Both genes contained a GH19 domain, a functional domain present in chitinases, but had no role in chitin degradation. Surprisingly, the deletion mutants exhibited two colony morphotypes, one like the wild type was rhizoid and the other was non-rhizoid and relatively less virulent, as revealed by their higher LD50 values and higher zebrafish survival rates.
In a recent study on chitinase in F. johnsoniae, Kharade and McBride (2014) detected its chitin degradation role and secretion through the IX secretion system. This chitinase contains two GH18 domains (Kharade and McBride, 2014); while the two annotated genes ghd-1 and ghd-2 in the F. columnare G4 genome encode putative proteins with a GH19 rather than a GH18 domain. The F. columnare strain ATCC 49512, which was the first strain of the species sequenced (Tekedar et al., 2012), contains a chitinase (FCOL_10530) exactly identical to Ghd-1 in F. columnare G4. Therefore, we presume that the chitinase in F. columnare ATCC 49512 cannot use chitin either, and suggest that the gene be renamed as in the present study.
In plant genomes, there are many sequences that encode inactive chitinase to protect plants from chitin-containing pathogens, these are known as chitinase-like proteins (CLPs) (Kesari et al., 2015). Although these proteins share high sequence and structural homology with GH18 and GH19 family chitinases, they may lack either binding or catalytic activity as a result of either a mutation in the chitinase consensus sequence or functional diversification (Kesari et al., 2015). In F. columnare G4, ghd-1 and ghd-2 may have functions other than chitin degradation, but their exact role needs further investigation. However, the wide geographical distribution of F. columnare G4 and ATCC 49512, which were isolated from different continents (Lu et al., 1975; Bernardet, 1989), implies that this bacterium does not utilize chitin; other genomovars of the bacterium were not examined and chitinase is functional in chitin utilization in other flavobacteria such as F. johnsoniae (Kharade and McBride, 2014) and may even serve as a virulence factor in other pathogenic bacteria such as Listeria monocytogenes (Chaudhuri et al., 2013). Indeed, the search for a chitinase gene in the F. psychrophilum genome, another fish pathogen causing bacterial cold water disease (CWD) in salmonid fish (Duchaud et al., 2007), revealed the absence of any chitinase gene. The ability to degrade chitin might have been lost in this group of fish bacterial pathogens.
It is, however, rather surprising that the two single ghd genes and the double gene mutant constructed in the present study all exhibited rhizoid and non-rhizoid colony morphotypes. Rhizoid and non-rhizoid colonies have been frequently reported in F. columnare (Kunttu et al., 2009, 2011; Laanto et al., 2014), and the rhizoid colony morphotype positively correlates with virulence (Kunttu et al., 2011; Laanto et al., 2014). The F. columnare rhizoid colony morphotype can be more adhesive, which may facilitate bacterial infection (Kunttu et al., 2011). Additionally, the organized structure observed in the rhizoid colony morphotype, with bacterial cells connected to each other through numerous thin fimbriae-like strings, may protect the bacteria from stressors (Laanto et al., 2014). However, these rhizoid and non-rhizoid colony morphotypes, when derived from the same strain, were genetically identical as has been revealed by amplified fragment length polymorphism (AFLP) and automated ribosomal intergenic spacer analysis (ARISA; Kunttu et al., 2011). Laanto et al. (2014) characterized six proteins in the membrane vesicle contents and extracellular proteins in a rhizoid colony morphotype using the same F. columnare strain, among which OmpA and SprF are the only known proteins. In contrast to the strains used by Kunttu et al. (2011) and Laanto et al. (2014), non-rhizoid colony morphotypes have never been observed in the F. columnare G4 strain in the laboratory (unpublished data). The observed rhizoid and non-rhizoid mutants following the single and double-gene deletions from F. columnare G4 implies that these two morphotypes can exist for a genetically identical strain as observed by Kunttu et al. (2011) and Laanto et al. (2014), and that when deleted, ghd genes may cause these changes in bacterial colony morphotypes. The observed difference in LD50 and survivorship between rhizoid and non-rhizoid mutants in the present study also indicates that F. columnare rhizoid mutants are more virulent than the non-rhizoid morphotype.
The OmpA identified by Laanto et al. (2014) has been reported in relation to virulence in many other bacterial pathogens (Dabo et al., 2003; Smith et al., 2007) and OmpA has been recognized as a candidate immunogen against CWD in F. psychrophilum (Dumetz et al., 2008). SprB, which exhibits gliding motility, is required along with SprC, SprD, and SprF, in the formation of rhizoid, spreading colonies in F. johnsoniae (Rhodes et al., 2011). However, genes encoding these two proteins were not detected in the three sets of transcriptome comparisons between the non-rhizoid and rhizoid mutants in the present study. The gld genes that are involved in F. johnsoniae gliding motility (McBride, 2001; McBride and Nakane, 2015) were not found in the list of differentially expressed genes in the present study either.
However, in a recent study, Dong et al. (2015) found that the F. columnare rhizoid and non-rhizoid isolates did not differ in their ability to adhere to fish, but the lack of virulence in the non-rhizoid isolate may have been the result of an inability to invade and persist in fish following adhesion. Rhizoid isolates may spread quickly and, thus, may be more infectious, but the mechanism involved in F. columnare infection is unknown and requires further investigation.
A large number of other genes were detected differentially in rhizoid and non-rhizoid mutants in the present study, with more genes being detected in Δghd-2 mutants. Despite that, the majority of differentially expressed genes common in the three mutants only encode hypothetical proteins, a few genes annotated as phage tail proteins and Rhs element proteins were up-regulated in the rhizoid mutants, Δghd-1, Δghd-2, and Δghd-1 Δghd-2, and may be related to either bacterial virulence or colony morphotype. Phage tail proteins are members of a cell membrane penetrating mechanism for bacterial secretion (Pukatzki et al., 2007). The Rhs elements are widely distributed in bacteria and have functions in mediating intercellular competition and sensing population density (Youderian and Hartzell, 2007; Koskiniemi et al., 2013). Additioanlly, a thiolactivated cytolysin and TonB-dependent outer membrane receptor precursor are related to bacterial virulence (Michel et al., 1990; Pauer et al., 2013). The genes that were up-regulated in the non-rhizoid mutants also included TonB-dependent receptors and nitrous-oxide reductase. However, the function of these genes in F. columnare are unknown.
Skimmed milk degradation by NΔghd-2 was unexpected, and a few genes annotated as either protein or peptide enzyme genes were up-regulated in this mutant. However, the mechanism involved in skimmed milk degradation is at present unknown and certainly requires further investigation.
5 CONCLUSIONTwo genes, ghd-1 and ghd-2, encoding proteins with a GH19 domain, a conserved domain in glycoside hydrolase family 19 proteins, were identified in the F. columnare G4 genome. However, the wild type G4 strain did not degrade chitin, and, thus, these two genes are not involved in chitin degradation. However, the single-and double-gene mutants exhibited variations in colony morphotype, growth, virulence, and transcriptomes, and the non-rhizoid colony mutants exhibited reduced virulence.
Electronic supplementary materialSupplementary material (Supplementary Figs.S1, S2 and Tables S1, S2) is available in the online version of this article at https://doi.org/10.1007/s00343-017-6160-z.
| Alvarez B, Secades P, McBride M J, Guijarro J A, 2004. Development of genetic techniques for the psychrotrophic fish pathogen Flavobacterium psychrophilum. Appl.Environ. Microbiol., 70(1): 581–587. Doi: 10.1128/AEM.70.1.581-587.2004 |
| Beck B H, Li C, Farmer B D, Barnett L M, Lange M D, Peatman E, 2015. A comparison of high-and lowvirulence Flavobacterium columnare strains reveals differences in iron acquisition components and responses to iron restriction. J. Fish Dis., 39(3): 259–268. |
| Beier S, Bertilsson S, 2013. Bacterial chitin degradation mechanisms and ecophysiological strategies. Front.Microbiol., 4: 149. |
| Bernardet J F, 1989. 'Flexibacter columnaris':first description in France and comparison with bacterial strains from other origins. Dis. Aquat. Org., 6: 37–44. Doi: 10.3354/dao006037 |
| Chaudhuri S, Gantner B N, Ye R D, Cianciotto N P, Freitag N E, 2013. The Listeria monocytogenes ChiA chitinase enhances virulence through suppression of host innate immunity. mBio, 4(2): e00617–12. |
| Dabo S M, Confer A W, Quijano-Blas R A, 2003. Molecular and immunological characterization of Pasteurella multocida serotype A:3 OmpA:evidence of its role in P.multocida interaction with extracellular matrix molecules.. Microb. Pathog., 35(4): 147–157. Doi: 10.1016/S0882-4010(03)00098-6 |
| Declercq A M, Haesebrouck F, van den Broeck W, Bossier P, Decostere A, 2013. Columnaris disease in fish:a review with emphasis on bacterium-host interactions. Vet. Res., 44: 27. Doi: 10.1186/1297-9716-44-27 |
| Decostere A, Haesebrouck F, van Driessche E, Charlier G, Ducatelle R, 1999. Characterization of the adhesion of Flavobacterium columnare (Flexibacter columnaris) to gill tissue. J. Fish Dis., 22(6): 465–474. Doi: 10.1046/j.1365-2761.1999.00198.x |
| Dong H T, Senapin S, LaFrentz B, Rodkhum C, 2015. Virulence assay of rhizoid and non-rhizoid morphotypes of Flavobacterium columnare in red tilapia, Oreochromis sp. , fry. J. Fish Dis., 39(6): 649–655. |
| Duchaud E, Boussaha M, Loux V, Bernardet J F, Michel C, Kerouault B, Mondot S, Nicolas P, Bossy R, Caron C, Bessières P, Gibrat J F, Claverol S, Dumetz F, Le Hénaff M, Benmansour A, 2007. Complete genome sequence of the fish pathogen Flavobacterium psychrophilum. Nat.Biotechnol., 25(7): 763–769. Doi: 10.1038/nbt1313 |
| Dumetz F, Duchaud E, Claverol S, Orieux N, Papillon S, Lapaillerie D, Le Hénaff M, 2008. Analysis of the Flavobacterium psychrophilum outer-membrane subproteome and identification of new antigenic targets for vaccine by immunomics. Microbiology, 154(6): 1793–1801. Doi: 10.1099/mic.0.2008/016600-0 |
| Edwards R A, Keller L H, Schifferli D M, 1998. Improved allelic exchange vectors and their use to analyze 987P fimbria gene expression. Gene, 207(2): 149–157. Doi: 10.1016/S0378-1119(97)00619-7 |
| Fujita K, Shimomura K, Yamamoto K, Yamashita T, Suzuki K, 2006. A chitinase structurally related to the glycoside hydrolase family 48 is indispensable for the hormonally induced diapause termination in a beetle. Biochem.Biophys. Res. Commun., 345(1): 502–507. Doi: 10.1016/j.bbrc.2006.04.126 |
| Gay P, Le Coq D, Steinmetz M, Ferrari E, Hoch J A, 1983. Cloning structural gene sacB, which codes for exoenzyme levansucrase of Bacillus subtilis:expression of the gene in Escherichia coli. J. Bacteriol., 153(3): 1424–1431. |
| Gooday G W. 1990. Physiology of microbial degradation of chitin and chitosan. In: Ratledge C ed. Biochemistry of Microbial Degradation. Springer, Dordrecht, Netherlands. p. 279-312. |
| Hackman R H, 1962. Studies on chitin V. The action of mineral acids on chitin. Aust. J. Biol. Sci., 15(3): 526–537. |
| Hoell I A, Vaaje-Kolstad G, Eijsink V G H, 2010. Structure and function of enzymes acting on chitin and chitosan. Biotechnol. Genet. Eng. Rev., 27(1): 331–366. Doi: 10.1080/02648725.2010.10648156 |
| Kesari P, Patil D N, Kumar P, Tomar S, Sharma A K, Kumar P, 2015. Structural and functional evolution of chitinase-like proteins from plants. Proteomics, 15(10): 1693–1705. Doi: 10.1002/pmic.v15.10 |
| Kharade S S, McBride M J, 2014. Flavobacterium johnsoniae chitinase ChiA is required for chitin utilization and is secreted by the type IX secretion system. J. Bacteriol., 196(5): 961–970. Doi: 10.1128/JB.01170-13 |
| Koskiniemi S, Lamoureux J G, Nikolakakis K C, t'Kint de Roodenbeke C, Kaplan M D, Low D A, Hayes C S, 2013. Rhs proteins from diverse bacteria mediate intercellular competition. Proc. Natl. Acad. Sci. U. S. A., 110(17): 7032–7037. Doi: 10.1073/pnas.1300627110 |
| Kubota T, Miyamoto K, Yasuda M, Inamori Y, Tsujibo H, 2004. Molecular characterization of an intracellular β-N-acetylglucosaminidase involved in the chitin degradation system of Streptomyces thermoviolaceus OPC-520. Biosci. Biotechnol. Biochem., 68(6): 1306–1314. Doi: 10.1271/bbb.68.1306 |
| Kumari S, Rath P K, 2014. Extraction and characterization of chitin and chitosan from (Labeo rohit) fish scales. Procedia Mater. Sci., 6: 482–489. Doi: 10.1016/j.mspro.2014.07.062 |
| Kunttu H M T, Jokinen E I, Valtonen E T, Sundberg L R, 2011. Virulent and nonvirulent Flavobacterium columnare colony morphologies:characterization of chondroitin AC lyase activity and adhesion to polystyrene. J. Appl.Microbiol., 111(6): 1319–1326. Doi: 10.1111/j.1365-2672.2011.05149.x |
| Kunttu H M T, Suomalainen L R, Jokinen E I, Valtonen E T, 2009. Flavobacterium columnare colony types:connection to adhesion and virulence? Microb. Pathog., 46(1): 21–27. |
| Laanto E, Penttinen R K, Bamford J K H, Sundberg L R, 2014. Comparing the different morphotypes of a fish pathogen-implications for key virulence factors in Flavobacterium columnare. BMC Microbiol., 14: 170. Doi: 10.1186/1471-2180-14-170 |
| Li N, Qin T, Zhang X L, Huang B, Liu Z X, Xie H X, Zhang J, McBride M J, Nie P, 2015. Gene deletion strategy to examine the involvement of the two chondroitin Lyases in Flavobacterium columnare virulence. Appl. Environ.Microbiol., 81(21): 7394–7402. Doi: 10.1128/AEM.01586-15 |
| Lu Q Z, Ni D S, Ge R F, 1975. Studies on the gill diseases of the grass carp (Ctenopharyngodon idelluls) Ⅰ. Isolation of a myxobacterial pathogen. Acta Hydrobiol. Sin., 5(3): 315–334. |
| McBride M J, Kempf M J, 1996. Development of techniques for the genetic manipulation of the gliding bacterium Cytophaga johnsonae. J. Bacteriol., 178(3): 583–590. Doi: 10.1128/jb.178.3.583-590.1996 |
| McBride M J, Nakane D, 2015. Flavobacterium gliding motility and the type IX secretion system. Curr. Opin.Microbiol., 28: 72–77. Doi: 10.1016/j.mib.2015.07.016 |
| McBride M J, Zhu Y T, 2013. Gliding motility and Por secretion system genes are widespread among members of the phylum Bacteroidetes. J. Bacteriol., 195(2): 270–278. Doi: 10.1128/JB.01962-12 |
| McBride M J, 2001. Bacterial gliding motility:multiple mechanisms for cell movement over surfaces. Annu. Rev.Microbiol., 55: 49–75. Doi: 10.1146/annurev.micro.55.1.49 |
| Michel E, Reich K A, Favier R, Berche P, Cossart P, 1990. Attenuated mutants of the intracellular bacterium Listeria monocytogenes obtained by single amino acid substitutions in listeriolysin O. Mol. Microbiol., 4(12): 2167–2178. Doi: 10.1111/mmi.1990.4.issue-12 |
| Mortazavi A, Williams B A, McCue K, Schaeffer L, Wold B, 2008. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods., 5(7): 621–628. Doi: 10.1038/nmeth.1226 |
| Murthy N, Bleakley B, 2012. Simplified method of preparing colloidal chitin used for screening of chitinase-producing microorganisms. Internet. J. Microbiol., 10(2): 14186. |
| Pauer H, Cavalcanti S N V, Teixeira F L, Santos-Filho J, Vommaro R C, Oliveira A C S C, Ferreira E O, Domingues R R M C P, 2013. Inactivation of a fibronectin-binding TonB-dependent protein increases adhesion properties of Bacteroides fragilis. J. Med. Microbiol., 62(10): 1524–1530. |
| Pukatzki S, Ma A T, Revel A T, Sturtevant D, Mekalanos J J, 2007. Type VI secretion system translocates a phage tail spike-like protein into target cells where it cross-links actin. Proc. Natl. Acad. Sci. U. S. A., 104(39): 15580–15513. |
| Reed L J, Muench H, 1938. A simple method of estimating fifty per cent endpoints. Am. J. Hyg., 27(3): 493–497. |
| Rhodes R G, Nelson S S, Pochiraju S, McBride M J, 2011. Flavobacterium johnsoniae sprB is part of an operon spanning the additional gliding motility genes sprC, sprD, and sprF. J. Bacteriol., 193(3): 599–610. Doi: 10.1128/JB.01203-10 |
| Rinaudo M, 2006. Chitin and chitosan:properties and applications. Prog. Polym. Sci., 31(7): 603–632. Doi: 10.1016/j.progpolymsci.2006.06.001 |
| Shieh H S, 1980. Studies on the nutrition of a fish pathogen, Flexibacter columnaris. Microbios Lett., 13: 129–133. |
| Smith S G J, Mahon V, Lambert M A, Fagan R P, 2007. A molecular Swiss army knife:OmpA structure, function and expression. FEMS Microbiol. Lett., 273(1): 1–11. Doi: 10.1111/fml.2007.273.issue-1 |
| Staats C C, Kmetzsch L, Lubeck I, Junges A, Vainstein M H, Schrank A, 2013. Metarhizium anisopliae chitinase CHIT30 is involved in heat-shock stress and contributes to virulence against Dysdercus peruvianus. Fungal Biol., 117(2): 137–144. Doi: 10.1016/j.funbio.2012.12.006 |
| Stringer-Roth K M, Yunghans W, Caslake L F, 2002. Differences in chondroitin AC lyase activity of Flavobacterium columnare isolates. J. Fish Dis., 25(11): 687–691. Doi: 10.1046/j.1365-2761.2002.00421.x |
| Štrojsová M, Vrba J, 2005. Direct detection of digestive enzymes in planktonic rotifers using enzyme-labelled fluorescence (ELF). Mar. Freshwater Res., 56(2): 189–195. Doi: 10.1071/MF04280 |
| Suomalainen L R, Tiirola M, Valtonen E T, 2006. Chondroitin AC lyase activity is related to virulence of fish pathogenic Flavobacterium columnare. J. Fish Dis., 29(12): 757–763. Doi: 10.1111/jfd.2006.29.issue-12 |
| Tekedar H C, Karsi A, Gillaspy A F, Dyer D W, Benton N R, Zaitshik J, Vamenta S, Banes M M, Gülsoy N, AbokoCole M, Waldbieser G C, Lawrence M L, 2012. Genome sequence of the fish pathogen Flavobacterium columnare ATCC 49512. J. Bacteriol., 194(10): 2763–2764. Doi: 10.1128/JB.00281-12 |
| Vrba J, Šimek K, Pernthaler J, Psenner R, 1996. Evaluation of extracellular, high-affinity β-N-acetylglucosaminidase measurements from freshwater lakes:an enzyme assay to estimate protistan grazing on bacteria and picocyanobacteria. Microb. Ecol., 32(1): 81–97. Doi: 10.1007/BF00170109 |
| Xie H X, Nie P, Chang M X, Liu Y, Yao W J, 2005. Gene cloning and functional analysis of glycosaminoglycan-degrading enzyme chondroitin AC lyase from Flavobacterium columnare G4. Arch. Microbiol., 184(1): 49–55. Doi: 10.1007/s00203-005-0009-0 |
| Xie H X, Nie P, Sun B J, 2004. Characterization of two membrane-associated protease genes obtained from screening out-membrane protein genes of Flavobacterium columnare G4. J. Fish Dis., 27(12): 719–729. Doi: 10.1111/jfd.2004.27.issue-12 |
| Youderian P, Hartzell P L, 2007. Triple mutants uncover three new genes required for social motility in Myxococcus xanthus. Genetics, 177(1): 557–566. Doi: 10.1534/genetics.107.076182 |
 2017, Vol. 35
2017, Vol. 35