Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZENG Lei(曾磊), HE Feng(贺锋), ZHANG Yi(张义), LIU Biyun(刘碧云), DAI Zhigang(代志刚), ZHOU Qiaohong(周巧红), WU Zhenbin(吴振斌)
- Size-dependent responses of zooplankton to submerged macrophyte restoration in a subtropical shallow lake
- Chinese Journal of Oceanology and Limnology, 36(2): 376-384
- http://dx.doi.org/10.1007/s00343-018-6192-z
Article History
- Received Jul. 27, 2016
- accepted in principle Sep. 6, 2016
- accepted for publication Feb. 3, 2017
2 University of Chinese Academy of Sciences, Beijing 100049, China
Shallow lakes, which generally have water columns less than 3 m in depth, are characterized by strong material exchange between the water column and sediment, slow deposition, unstable thermal stratification and increased sensitivity to pollution compared with deep lakes (Jeppesen et al., 1997; Sachse et al., 2014). Most shallow lakes are confined to low-lying areas and are vulnerable to nutrient enrichment from domestic sewage, intensive agricultural activities and industry (Kõiv et al., 2011). During the past 50 years, eutrophication has become a serious threat to shallow lakes around the world, causing deterioration of aquatic ecosystem quality and toxic algal blooms, which has resulted in water shortages for residential supplies and decreased lake recreational values. Eutrophication and the changes associated with it are especially problematic in developing countries, where they constantly endanger human health and the quality of aquatic products.
In recent decades, many efforts have been made to solve the problems associated with eutrophication, particularly in Europe and North America (Jeppesen et al., 2005a, b; Søndergaard et al., 2005). Although substantial reduction in external nutrient loading is widely regarded as a prerequisite for restoring lake ecosystems (Jeppesen et al., 2007a; Xu et al., 2010), this alone is not sufficient because of the delayed effects of internal nutrient release from sediments and biological resistance (Søndergaard et al., 2002; Gulati et al., 2008; Jeppesen et al., 2009). Accordingly, various physico-chemical and biological methods have been used and developed to overcome these problems, such as sediment removal (Zhang et al., 2010a), chemical treatment of sediment (Reitzel et al., 2005), fish manipulation (Beklioglu et al., 2008) and protection and restoration of submerged macrophytes (Lauridsen et al., 2003; Zhang et al., 2010b).
The restoration and protection of macrophytes has received increasing attention from lake managers and ecologists. Moreover, lake managers have adopted the option of increasing macrophyte abundance to restore eutrophic waters in temperate and subtropical/ tropical regions. The extensive use of macrophyte restoration to reconstruct aquatic ecosystems mainly results from its positive impacts on the formation and stabilization of a clear-water state in shallow lakes, and various mechanisms have been proposed for these impacts. One suggested mechanism is that the allelopathic substances released by macrophytes significantly suppress phytoplankton, decreasing the risk of algal blooms (Mulderij et al., 2003). Another mechanism is that macrophytes can develop water transparency by reducing wind- and fish-induced sediment resuspension (Gulati and van Donk, 2002). In addition, the direct absorption of nutrients by macrophytes can effectively decrease nutrition loading in the water column, thereby acting as a major nutrient sink. Finally, macrophytes can provide refuge for large-sized zooplankton from fish predation, resulting in increased phytoplankton grazing (Peretyatko et al., 2009).
Among the aforementioned mechanisms, the increased predation pressure of large-sized zooplankton on phytoplankton has been widely studied in many shallow lakes around the world (Brönmark and Weisner, 1992; Romo et al., 2005; Beklioglu et al., 2007), especially in European temperate lakes (Jeppesen et al., 2007b). In these lakes, zooplankton can enhance survival by migrating to habitats in which predation risk is low, such as littoral areas that are covered with submerged macrophytes (Estlander et al., 2009; Sagrario et al., 2009), after which they exert strong grazing pressure on phytoplankton (Agasild et al., 2007). However, while studies of the effects of macrophyte restoration on zooplankton in temperate, shallow lakes has provided relatively comprehensive information, the size-dependent responses of zooplankton to macrophyte restoration are less known in subtropical (Meerhoff et al., 2007) and tropical lakes (Jeppesen et al., 2007b, 2012).
Therefore, we conducted a three year investigation to explore the size-dependent responses of rotifers and crustaceans to macrophyte restoration in a subtropical shallow lake. Moreover, macrophytes and water quality were also investigated to explore the potential relevant factors responsible for the sizedependent responses of zooplankton.
2 MATERIAL AND METHOD 2.1 Study siteThe Xihu Lake (30°15′N, 120°09′E) is a typical shallow lake located in Hangzhou City, Zhejiang Province, eastern China that attracts a great number of tourists from all over the world. The lake was officially added to the World Heritage List in 2011. The lake occupies an area of 6.5 km2 and has a mean depth of 2.27 m, giving a water volume of 1.49×107 m3.
This study investigated Maojiabu Lake (30°13′N, 120°07′E), which has an area of 0.27 km2 and a mean depth of 1.3 m and is located in the western portion of the Xihu Lake (Fig. 1). Before macrophyte restoration, this lake was in a turbid state with a high chlorophyll a (Chl-a) concentration (mean: 25±6 μg/L) and low transparency (Secchi depth: 0.6±0.12 m). Additionally, the lake was characterized by high total nitrogen (TN) (2.6±0.29 mg/L) and chemical oxygen demand (COD) (2.2±0.34 mg/L) and low total phosphorus (TP) (0.03±0.01 mg/L) levels. At this time, almost no submerged macrophytes were present in the lake (Zeng, unpublished data).

|
| Figure 1 Location and enlarged view of Maojiabu Lake in the Xihu Lake in Hangzhou, China The 14 sites and three black filled circles were designed to collect samples of submerged macrophyte and water quality from 2010 to 2012, respectively. |
To restore the aquatic ecosystem in Maojiabu Lake, an attempt was first made to reconstruct the submerged macrophyte community in autumn 2010 to spring 2011. In November 2010, macrophyte restoration started and turions of Potamogeton crispus were planted in most parts of the lake (ca. 80% of the overall area). In February 2011, seeds of Vallisneria spiralis were planted in zones less than 0.5 m deep. One month later, adult V. spiralis, Ceratophyllum demersum and Myriophyllum verticillatum were planted, mostly in zones that were greater than 0.5 m in depth. The V. spiralis seedlings in the shallow zones grew poorly from March to October in 2011, but adult macrophytes exhibited exuberant growth in the zones deeper than 0.5 m.
In November 2011, a second attempt at macrophyte restoration was made, mainly in zones less than 0.5 m. This time, adult V. spiralis instead of their seeds were planted in the shallow zones, and successful macrophyte restoration was achieved in 2012.
2.3 Sampling and treatmentDuring the restoration, continuous tracking surveys of the macrophyte community were conducted in spring (April), summer (July) and autumn (October) from 2010 to 2012. Fourteen sampling sites were selected across the lake to measure the biomass and percentage coverage of macrophytes (Fig. 1). At each sampling site, we used a grass sickle to collect triplicate macrophyte samples, with each sample being collected from an area of about 0.18 m2. In addition, species and coverage were recorded simultaneously during field sampling. The fresh weight was obtained after washing the plants with tap water and weighing them in a PuChun electronic scale (6 kg/0.2 g) in the laboratory.
Water quality was also monitored seasonally at the same frequency as the macrophytes, but only three sampling sites (6, 12 and 14) were selected to collect the water samples (Fig. 1). COD, TN, nitrate nitrogen (NN), ammonium nitrogen (AN), TP and Chl-a were analyzed according to the standard methods (Editorial Board of Monitoring and Determination Methods for Water and Wastewater, State Environmental Protection Administration of China, 2002).
Triplicate zooplankton samples were also collected seasonally in the same frequency from the same sampling sites used to evaluate water quality (Fig. 1). Crustaceans (cladoceran and copepod) were collected by filtering 10 L of water through a 64-μm plankton net into a 30-mL plastic bottle, after which they were preserved by adding 3 mL 5% formalin. Rotifer samples were obtained by injecting 1 L of water into a 1.5-L plastic bottle, then fixed with 9 mL Lugol's solution. In the laboratory, crustacean samples were identified directly in a dissecting stereoscope at 40× magnification. Rotifer samples were first concentrated to 30 mL sub-samples, after which 1 mL sub-samples were absorbed with a graduated pipette into a countframe and counted using an inverted microscope at 160× magnification. Zooplankton species were identified to the genus/species level with reference to Wang (1961), Tai and Chen (1979) and Chiang and Du (1979).
2.4 Statistical analysisThe Shapiro-Wilks test and Levene's test were used to assess the normality and equality of variance, respectively. To analyze the size-dependent responses of zooplankton to macrophyte restoration, zooplankton was divided into two subgroups (crustacean and rotifer), and rotifers were divided into three groups based on their sizes (G1: < 200 μm; G2: 200 μm– 400 μm; G3: > 400 μm) (Wang et al., 1961). Moreover, one-way ANOVA and Duncan's test were conducted to identify significant differences in the density and biomass of rotifers, crustaceans and zooplankton, as well as the crustacean-to-rotifer ratio with changes in macrophyte cover. Significant changes in rotifers with different sizes were also evaluated to analyze the effects of reconstructed macrophytes on rotifers. All analyses were completed using the statistical program SPSS 21.0 for windows.
Multivariate analysis using redundancy analysis (RDA) was conducted with the CANOCO 4.5 software to study the correlations between environmental factors and zooplankton during macrophytes restoration. The Monte Carlo permutation test was conducted to test the significance of eigenvalues of the first and all ordination axes. Eight environmental variables were included in this analysis: macrophyte biomass and coverage, COD, TN, TP, AN, NN and Chl-a. The six zooplankton parameters included in this analysis were the densities of total zooplankton, total rotifer, crustaceans and rotifers at size categories G1, G2 and G3.
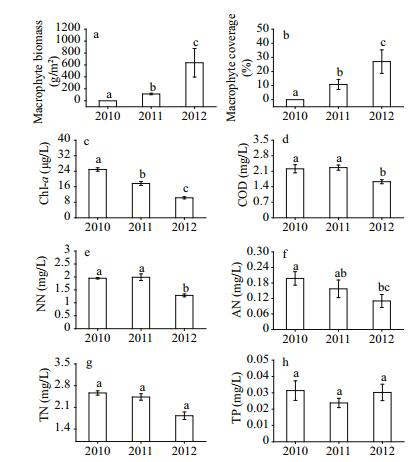
3 RESULT 3.1 Interannual variations in macrophyte and water qualityThe three surveys conducted in 2010 prior to the restoration showed that almost no submerged macrophytes were present in the lake. After the first restoration trial in winter of 2010 and February of 2011, the annual mean biomass and coverage of macrophytes in 2011 had increased significantly from 0 to 113±12 g/m2 and 0 to 11%±4% compared to those in 2010, respectively (P < 0.05, Fig. 2a–b). The dominant species (V. spiralis, P. crispus, C. demersum and M. verticillatum) were mainly distributed in the deep zone. However, the germination rate of V. spiralis seeds in shallow areas was very low, and the seedlings showed poor growth.
|
| Figure 2 Interannual mean variations (±SE) in submerged macrophyte (MB (a) and MC (b)) (n=126) and water quality parameters (Chl-a (c), COD (d), NN (e), AN (f), TN (g), and TP (h)) (n=27) from 2010 to 2012 The abbreviations of the environmental variables are defined in Table 2. Bars with identical lowercase letters indicate no significant differences (P > 0.05), while bars with different letters indicate significant differences (P < 0.05). |
Surveys conducted in 2012 showed that the annual mean biomass and coverage of macrophytes increased significantly compared to those in 2011 (P < 0.05), and were 637±239 g/m2 and 27%±8%, respectively. The dominant species were V. spiralis, Najas marina and M. verticillatum. The N. marina was likely brought into the lake with other macrophytes during two restoration attempts.
All water quality parameters except for TP presented significant differences during the restoration (P < 0.05, Fig. 2c–g). Specifically, the concentrations of TN, NN and COD did not differ significantly between 2010 and 2011, but decreased in 2012 (P < 0.05). The Chl-a concentration also gradually decreased significantly every year (P < 0.05), while significant differences in AN only occurred between 2010 and 2012. The TP concentration was in a stable state throughout the restoration (Fig. 2h).
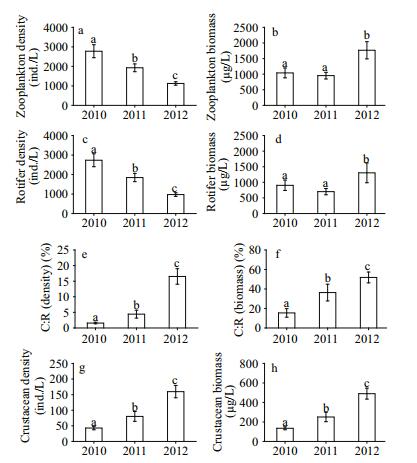
3.2 Interannual variations in zooplankton related to body sizeDuring the restoration, similar changes in the density and biomass of zooplankton and rotifers were observed (Fig. 3a–d). Specifically, their density decreased significantly every year (P < 0.05), but their biomass increased significantly in 2012 (P < 0.05) after undergoing a stable period from 2010 to 2011. Moreover, the change trends in the crustacean-to-rotifer ratios and in crustaceans were similar (Fig. 3e–h), with both gradually increasing significantly every year (P < 0.05).
|
| Figure 3 Interannual mean variations (±SE) in zooplankton density (a) and biomass (b), rotifer density (c) and biomass (d), the density (e) and biomass (f) ratio of crustaceans to rotifers (C:R), and crustacean density (g) and biomass (h) from 2010 to 2012 (n=27) Bars with identical lowercase letters indicate no significant differences (P > 0.05), while bars with different letters indicate significant differences (P < 0.05). |
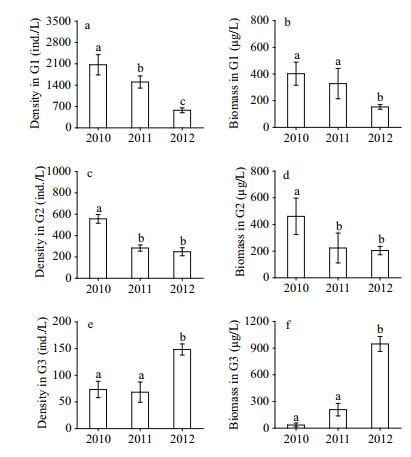
Rotifers of different sizes also exhibited different responses to macrophyte restoration (Fig. 4). Specially, the rotifer density in G1 (representative species: Polyarthra trigla, Keratella cochlearis, Anuraeopsis fissa) (Table 1) decreased significantly every year (P < 0.05, Fig. 4a), as did the biomass in 2012 compared to 2010 and 2011 (P < 0.05, Fig. 4b). The density of rotifers in G2 (representative species: Synchaeta stylata, S. bologna.) showed a similar change trend as biomass, with significant decreases occurring in 2011 and 2012 compared to 2010 (P < 0.05, Fig. 4c–d). However, the density of rotifers in G3 (representative species: S. pectinata, Eosphora najas, Enteroplea lacustris, Asplanchna priodonta) increased significantly in 2012 compared to 2010 and 2011, and the biomass increased significantly every year (P < 0.05, Fig. 4e–f).
|
| Figure 4 Interannual mean variations (±SE) in rotifer density and biomass in G1 (a–b), G2 (c–d) and G3 (e–f) from 2010 to 2012 (n=27) RD: rotifer density; RB: rotifer biomass. Bars with identical lowercase letters indicate no significant differences (P > 0.05), while bars with different letters indicate significant differences (P < 0.05). |
In the ordination diagram, strong correlations existed between zooplankton and environmental factors (water quality and macrophyte), with zooplankton-environment correlations of 0.81 on the first axis and 0.927 on the second axis. The cumulative percentage variance of the zooplankton-environment relationship on the first axis was 53.2%, whereas that on the second axis was 42.5%. The cumulative percentage variance of the zooplankton data explained by the first four axes of the RDA was 62.2%, with 33.1% on first axis and 26.5% on the second axis (Fig. 5). The Monte Carlo permutation test was significant on the first axis (F-ratio=8.903, Pvalue=0.01) and on all axes (F-ratio=3.707, Pvalue=0.002). According to the permutation test of all environmental factors, six variables (macrophyte biomass and coverage, TN, NN Chl-a and TP) were the best explanatory variables for zooplankton variations, explaining 0.503 of total zooplankton variations (0.622).
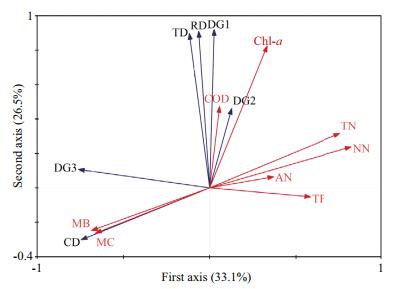
|
| Figure 5 The RDA ordination plots with zooplankton and environment variables and samples The red arrows represent environment variables (water quality and macrophytes), and the blue arrows represent zooplankton variables. Abbreviations of environmental variables are presented in Table 1. TD, CD and RD represent densities of total zooplankton, crustaceans and rotifers, respectively. DG1, DG2 and DG3 represent rotifer density in G1, G2 and G3, respectively. |
According to the centroid principle and distance rule implied in RDA, the crustacean density was positively correlated with macrophyte biomass and coverage, but negatively correlated with Chl-a, COD, TN and NN. Significant positive correlations also existed between total zooplankton, total rotifer, rotifer in G1 and G2, COD and Chl-a. In addition, rotifer in G3 was only negatively correlated with TN and NN (Fig. 5).
The correlations between macrophytes and water quality obtained through RDA (Table 2) showed that macrophyte biomass and coverage were negatively correlated with COD, TN, NN and Chl-a. However, COD showed significantly positive correlations with TN and Chl-a, and significantly positive correlations also existed between TP and TN, TN and NN or Chl-a.
The results of this study revealed that a successful macrophyte restoration in 2012 led to significant increases in the density and biomass of crustaceans compared to 2010 and 2011. When combined with the positive correlations between macrophytes and crustaceans, these findings suggest that macrophytes enhanced the survival of crustaceans by providing refuge effects against fish predation. This conclusion is consistent with the findings of studies conducted in temperate lakes (Špoljar et al., 2011, 2016). For example, Cazzanelli et al. (2008) and Špoljar et al. (2012) stated that dense macrophytes in the littoral zone with a low predation risk might enhance crustacean survival. Burks et al. (2002) also suggested that crustaceans could take full advantage of the barrier function of macrophytes in the littoral zone to escape predator predations when a high risk of predation existed in the open water during the daytime.
However, some studies in tropical and subtropical lakes have suggested that the areas of refuge provided for large-sized zooplankton by submerged macrophytes were very limited (Jeppesen et al., 2005b; Castro et al., 2007; Meerhoff et al., 2007). This conclusion is primarily based on the fact that the number and diversity of fish in macrophytes in subtropical/tropical lakes is greater than in temperate lakes, thus producing greater predation pressures on large zooplankton (Teixeira-de Mello et al., 2009). Indeed, fish communities in warm tropical/subtropical lakes are characterized by short lifespan, early maturity, vigorous growth and frequent reproduction (Blanck and Lamouroux, 2007; van Leeuwen et al., 2007), and can exhibit stronger predation pressures on large-sized zooplankton than temperate lakes. However, the fact that the predation efficiency of fish can also be significantly influenced by the complex structure of macrophytes should not be ignored. Theoretically, fish predation of large zooplankton will be weakened if macrophyte coverage or biomass is sufficient. Whether large-sized zooplankton select macrophytes or not largely depends on the trade-off analysis of refuge and predation among macrophytes.
In this study, when macrophyte mean coverage and biomass in 2011 reached 11% and 113 g/m2, respectively, the crustaceans increased significantly compared with those in 2010, suggesting that the protection of crustaceans by macrophytes already existed in 2011. Moreover, the protection of crustaceans from fish predation was enhanced by the increasing vegetation coverage and biomass in 2012. These results were consistent with those of enclosure experiments conducted by Schriver et al. (1995), who found that some crustaceans could be effectively protected against fish predation when macrophyte coverage exceed 15%–20%, but that the protection would disappear when macrophyte coverage was lower than 10%.
4.2 Responses of rotifersUnlike the protection provided by macrophytes to crustaceans, the effects of macrophyte restoration on rotifers mainly depended on their sizes. Specifically, the abundance of large sized rotifers increased, while moderate and small-sized rotifers were suppressed during the restoration. Moreover, these size-dependent differences led to decreased total rotifer density, but increased biomass.
However, the positive correlations between small and moderate sized rotifers and COD or Chl-a might indicate that their growth inhibition resulted from a shortage of food resources. Based on the negative correlations between macrophytes and COD or Chl-a, restored macrophytes might indirectly suppress the growth of rotifers in G1 and G2 by decreasing their food concentrations.
In shallow lakes, COD is most likely to be affected by suspension of sediments in response to waves caused by wind and boats (Miranda, 2008). The Xihu Lake is affected by typhoons from the East China Sea every year, and patrol and cruise boats frequently cross the lake, all of which results in large waves, and therefore increased COD levels. However, reconstructed macrophytes have been shown to effectively reduce wave energies, protecting the sediment from erosion and resuspension and promoting sedimentation (Kufel and Kufel, 2002; Pluntke and Kozerski, 2003; James et al., 2004; Li et al., 2008). These changes ultimately lead to decreased concentrations of organic matter. Furthermore, the negative correlation between macrophytes and Chl-a might indicate that phytoplankton biomass was also suppressed by restored macrophytes. Accordingly, two mechanisms might contribute to this inhibition. Specifically, macrophytes may directly suppress phytoplankton and periphyton by producing allelopathic substances (Chang et al., 2012; EspinosaRodríguez et al., 2016) and competing for limited nutrients. Conversely, they may indirectly decrease phytoplankton levels by strengthening the predation of large-sized zooplankton on phytoplankton (Lacerot et al., 2013). Therefore, reconstructed macrophytes could significantly decrease the food resources (COD and Chl-a) for rotifers in G1 and G2, and thus indirectly suppress their growth.
4.3 Zooplankton community variationsIn this study, the overall density of total zooplankton decreased significantly every year. Within this group, rotifer density decreased significantly, but that of crustaceans increased significantly every year, as did the crustacean-to-rotifer-density ratio. However, the crustacean-to-rotifer-biomass ratio and the biomass of total zooplankton, rotifers and crustaceans in 2012 were significantly higher than those in 2010 and 2011.
In shallow, subtropical lakes, fish predation is an important factor controlling large-sized zooplankton, such as crustaceans and large rotifers (Fernandes et al., 2009; Teixeira-de Mello et al., 2009). As discussed above, dense macrophytes could effectively decrease fish predation of large zooplankton, thereby promoting their growth. Moreover, once predation by fish predators was no longer the main factor limiting large-sized zooplankton growth, the inherent competitive advantages (e.g., large body size) of large-sized zooplankton compared to small rotifers (e.g., stronger starvation tolerance, higher potential fecundity and broader food spectrum) contributed to their significant increases (Cyr and Curtis, 1999).
During the restoration, the main food resources (phytoplankton and organic detritus) for zooplankton were significantly decreased by macrophytes. Thus, limited resources made large-sized zooplankton more competitive while suppressing the growth of small rotifers because of a lack of available food. Based on the above analysis, it was not difficult to infer that submerged macrophyte restoration could encourage large-sized zooplankton and suppress small rotifers, leading to a significant increase in crustacean-torotifer ratio in the zooplankton community.
5 CONCLUSIONThis study showed that submerged macrophyte restoration increased the ratio of large-sized zooplankton in the zooplankton community in a subtropical shallow lake. Specifically, crustaceans and large-sized rotifers exhibited vigorous growth, while small-sized rotifers were significantly suppressed. However, the main mechanisms responsible for these effects might be different. Macrophytes primarily accelerated the growth of large-sized zooplankton by providing effective refuge effects against predator predation. Conversely, the growth inhibition of small-sized rotifers in response to restored macrophytes was likely a result of bottom-up control of nutrients. Overall, these findings indicated that an important mechanism by which macrophyte restoration leads to remarkable improvements in aquatic ecosystems is via increased predation of phytoplankton by large-sized zooplankton in subtropical shallow lakes.
6 ACKNOWLEDGEMENTWe thank Prof. QIU Dongru and Dr. WANG Yafen for their valuable comments and advice. Thanks are also given to other laboratory colleagues for field and laboratory work assistance.
| A Y, Chen G X. 1979. Cyclopoida. Fauna Sinica, Crustacea, Freshwater Copepoda. Science Press, Beijing, China. 450p. (in Chinese) |
| Agasild H, Zingel P, Tõnno I, Haberman J, Nõges T, 2007. Contribution of different zooplankton groups in grazing on phytoplankton in shallow eutrophic Lake Võrtsjärv(Estonia). Hydrobiologia, 584(1): 167–177. Doi: 10.1007/s10750-007-0575-z |
| Beklioglu M, Gozen A G, Yıldırım F, Zorlu P, Onde S, 2008. Impact of food concentration on diel vertical migration behaviour of Daphnia pulex under fish predation risk. Hydrobiologia, 614(1): 321–327. Doi: 10.1007/s10750-008-9516-8 |
| Beklioglu M, Romo S, Kagalou I, Quintana X, Bécares E, 2007. State of the art in the functioning of shallow Mediterranean lakes:workshop conclusions. Hydrobiologia, 584(1): 317–326. Doi: 10.1007/s10750-007-0577-x |
| Blanck A, Lamouroux N, 2007. Large-scale intraspecific variation in life-history traits of European freshwater fish. J. Biogeogr., 34(5): 862–875. |
| Brönmark C, Weisner S E B, 1992. Indirect effects of fish community structure on submerged vegetation in shallow, eutrophic lakes:an alternative mechanism. Hydrobiologia, 243-244: 293–301. Doi: 10.1007/BF00007045 |
| Burks R L, Lodge D M, Jeppesen E, Lauridsen T L, 2002. Diel horizontal migration of zooplankton:costs and benefits of inhabiting the littoral. Freshwater Biol., 47(3): 343–365. Doi: 10.1046/j.1365-2427.2002.00824.x |
| Castro B B, Marques S M, Gonçalves F, 2007. Habitat selection and diel distribution of the crustacean zooplankton from a shallow Mediterranean lake during the turbid and clear water phases. Freshwater Biol., 52(3): 421–433. Doi: 10.1111/fwb.2007.52.issue-3 |
| Cazzanelli M, Warming T P, Christoffersen K S, 2008. Emergent and floating-leaved macrophytes as refuge for zooplankton in a eutrophic temperate lake without submerged vegetation. Hydrobiologia, 605(1): 113–122. Doi: 10.1007/s10750-008-9324-1 |
| Chang X X, Eigemann F, Hilt S, 2012. Do macrophytes support harmful cyanobacteria? Interactions with a green alga reverse the inhibiting effects of macrophyte allelochemicals on Microcystis aeruginosa. Harmful Algae, 19: 76–84. Doi: 10.1016/j.hal.2012.06.002 |
| Cyr H, Curtis J M, 1999. Zooplankton community size structure and taxonomic composition affects size-selective grazing in natural communities. Oecologia, 118(3): 306–315. Doi: 10.1007/s004420050731 |
| Editorial Board of Monitoring and Determination Methods for Water and Wastewater, State Environmental Protection Administration of China. 2002. Monitoring and Determination Methods for Water and Wastewater. 4th edn. China Environmental Science Press, Beijing, China. 836p. (in Chinese) |
| Espinosa-Rodríguez C A, Valencia-del Toro G, Sarma S S S, Nandini S, 2016. Allelopathic activity and chemical analysis of crude extracts from the macrophyte egeria densa on selected phytoplankton species. Allelopathy J., 39(1): 147–160. |
| Estlander S, Nurminen L, Olin M, Vinni M, Horppila J, 2009. Seasonal fluctuations in macrophyte cover and water transparency of four brown-water lakes:implications for crustacean zooplankton in littoral and pelagic habitats. Hydrobiologia, 620(1): 109–120. Doi: 10.1007/s10750-008-9621-8 |
| Fernandes R, Gomes L C, Pelicice F M, Agostinho A A, 2009. Temporal organization of fish assemblages in floodplain lagoons:the role of hydrological connectivity. Environ.Biol. Fish., 85(2): 99–108. Doi: 10.1007/s10641-009-9466-7 |
| Gulati R D, Pires L M D, van Donk E, 2008. Lake restoration studies:failures, bottlenecks and prospects of new ecotechnological measures. Limnologica, 38(3-4): 233–247. Doi: 10.1016/j.limno.2008.05.008 |
| Gulati R D, van Donk E, 2002. Lakes in the Netherlands, their origin, eutrophication and restoration:state-of-the-art review. Hydrobiologia, 478(1-3): 73–106. |
| James W F, Barko J W, Butler M G, 2004. Shear stress and sediment resuspension in relation to submersed macrophyte biomass. Hydrobiologia, 515(1-3): 181–191. Doi: 10.1023/B:HYDR.0000027329.67391.c6 |
| Jeppesen E, Jensen J P, Søndergaard M, Lauridsen T, Pedersen L J, Jensen L, 1997. Top-down control in freshwater lakes:the role of nutrient state, submerged macrophytes and water depth. Hydrobiologia, 342-343: 151–164. Doi: 10.1023/A:1017046130329 |
| Jeppesen E, Jensen J P, Søndergaard M, Lauridsen T L, 2005b. Response of fish and plankton to nutrient loading reduction in Eight shallow Danish lakes with special emphasis on seasonal dynamics. Freshwater Biol., 50(10): 1 616–1 627. Doi: 10.1111/fwb.2005.50.issue-10 |
| Jeppesen E, Meerhoff M, Jacobsen B A, Hansen R S, Søndergaard M, Jensen J P, Lauridsen T L, Mazzeo N, Branco C W C, 2007b. Restoration of shallow lakes by nutrient control and biomanipulation-the successful strategy varies with lake size and climate. Hydrobiologia, 581(1): 269–285. Doi: 10.1007/s10750-006-0507-3 |
| Jeppesen E, Søndergaard M, Jensen H S, Ventäla A M. 2009. Lake and reservoir Management. In: Likens G E ed. Encyclopedia of Inland Waters. Elsevier, Oxford. |
| Jeppesen E, Søndergaard M, Lauridsen T L, Hansen R S, Søndergaard M, Jensen J P, Lauridsen T L, Davidson T A, Liu Z W, Mazzeo N, Trochine C, Özkan K, Jensen H S, Trolle D, Starling F, Lazzaro X, Johansson L S, Bjerring R, Liboriussen L, Larsen S E, Landkildehus F, Egemose S, Meerhoff M, 2012. Chapter 6-biomanipulation as a restoration tool to combat eutrophication:recent advances and future challenges. Adv. Ecol. Res., 47: 411–488. Doi: 10.1016/B978-0-12-398315-2.00006-5 |
| Jeppesen E, Søndergaard M, Mazzeo N, Meerhoff M, Branco C W C, Huszar V L M, Scasso F. 2005a. Lake restoration and biomanipulation in temperate lakes: relevance for subtropical and tropical lakes. In: Reddy V ed. Tropical Eutrophic Lakes: Their Restoration and Management. Oxford Publishing, I. B. H Publishing, New Hampshire. p. 331-359. |
| Jeppesen E, Søndergaard M, Meerhoff M, Lauridsen T L, Jensen J P, 2007a. Shallow lake restoration by nutrient loading reduction-some recent findings and challenges ahead. Hydrobiologia, 584(1): 239–252. Doi: 10.1007/s10750-007-0596-7 |
| Jiang S, Du N. 1979. Fauna Sinica, Crustacea, Freshwater Cladocera. Science Press, Academia Sinica, Beijing, China. 297p. (in Chinese) |
| Kõiv T, Nõges T, Laas A, 2011. Phosphorus retention as a function of external loading, hydraulic turnover time, area and relative depth in 54 lakes and reservoirs. Hydrobiologia, 660(1): 105–115. Doi: 10.1007/s10750-010-0411-8 |
| Kufel L, Kufel I, 2002. Chara beds acting as nutrient sinks in shallow lakes-a review. Aquat. Bot., 72(3-4): 249–260. Doi: 10.1016/S0304-3770(01)00204-2 |
| Lacerot G, Kruk C, Lürling M, Scheffer M, 2013. The role of subtropical zooplankton as grazers of phytoplankton under different predation levels. Freshwater Biol., 58(3): 494–503. Doi: 10.1111/fwb.2013.58.issue-3 |
| Lauridsen T L, Jensen J P, Jeppesen E, Søndergaard M, 2003. Response of submerged macrophytes in Danish lakes to nutrient loading reductions and biomanipulation. Hydrobiologia, 506-509(1-3): 641–649. Doi: 10.1023/B:HYDR.0000008633.17385.70 |
| Li E H, Li W, Liu G H, Yuan L Y, 2008. The effect of different submerged macrophyte species and biomass on sediment resuspension in a shallow freshwater lake. Aquat. Bot., 88(2): 121–126. Doi: 10.1016/j.aquabot.2007.09.001 |
| Meerhoff M, Clemente J M, de Mello F T, Iglesias C, Pedersen A R, Jeppesen E, 2007. Can warm climate-related structure of littoral predator assemblies weaken the clear water state in shallow lakes?. Global Change Biol., 13(9): 1 888–1 897. Doi: 10.1111/gcb.2007.13.issue-9 |
| Miranda L E. 2008. Extending the scale of reservoir management. In: Allen M S, Sammons S, Maceina M J, eds. Balancing Fisheries Management and Water Uses for Impounded River Systems. American Fisheries Society, Bethesda. |
| Mulderij G, van Donk E, Roelofs J G M, 2003. Differential sensitivity of green algae to allelopathic substances from Chara. Hydrobiologia, 491(1-3): 261–271. Doi: 10.1023/A:1024483704903 |
| Peretyatko A, Teissier S, De Backer S, Triest L, 2009. Restoration potential of biomanipulation for eutrophic peri-urban ponds:the role of zooplankton size and submerged macrophyte cover. Hydrobiologia, 634(1): 125–135. Doi: 10.1007/s10750-009-9888-4 |
| Pluntke T, Kozerski H P, 2003. Particle trapping on leaves and on the bottom in simulated submerged plant stands. Hydrobiologia, 506(1-3): 575–581. |
| Reitzel K, Hansen J, Andersen F Ø, Hansen K S, Jensen H S, 2005. Lake restoration by dosing aluminum relative to mobile phosphorus in the sediment. Environ. Sci. Technol., 39(11): 4 134–4 140. Doi: 10.1021/es0485964 |
| Romo S, Villena MJ, Sahuquillo M, Soria J M, Gimenez M, Alfonso T, Vicente E, Miracle M R, 2005. Response of a shallow Mediterranean lake to nutrient diversion:does it follow similar patterns as in northern shallow lakes?. Freshwater Biol, 50(10): 1 706–1 717. Doi: 10.1111/fwb.2005.50.issue-10 |
| Sachse R, Petzoldt T, Blumstock M, Moreira S, Pätzig M, Rücker J, Janse J H, Mooij W M, Hilt S, 2014. Extending one-dimensional models for deep lakes to simulate the impact of submerged macrophytes on water quality. Environ. Modell. Sof., 61: 410–423. Doi: 10.1016/j.envsoft.2014.05.023 |
| Sagrario G, De Losángeles M, Balseiro E, Ituarte R, Spivak E, 2009. Macrophytes as refuge or risky area for zooplankton:a balance set by littoral predacious macroinvertebrates. Freshwater Biol., 54(5): 1 042–1 053. Doi: 10.1111/fwb.2009.54.issue-5 |
| Schriver P, Bøgestrand J, Jeppesen E, Søndergaard M, 1995. Impact of submerged macrophytes on fish-zooplanlphytoplankton interactions:large-scale enclosure experiments in a shallow eutrophic lake. Freshwater Biol., 33(2): 255–270. Doi: 10.1111/fwb.1995.33.issue-2 |
| Søndergaard M, Jensen J P, Jeppesen E, Møller P H, 2002. Seasonal dynamics in the concentrations and retention of phosphorus in shallow Danish lakes after reduced loading. Aquat. Ecosyst. Health Manag., 5(1): 19–29. Doi: 10.1080/14634980260199936 |
| Søndergaard M, Jeppesen E, Jensen J P, Amsinck S L, 2005. Water framework directive:ecological classification of Danish lakes. J. Appl. Ecol., 42(4): 616–629. Doi: 10.1111/jpe.2005.42.issue-4 |
| Špoljar M, Dražina T, Habdija I, Meseljević M, Grčić Z, 2011. Contrasting zooplankton assemblages in two oxbow lakes with low transparencies and narrow emergent macrophyte belts (Krapina River, Croatia). Int. Rev. Hydrobiol., 96(2): 175–190. Doi: 10.1002/iroh.v96.2 |
| Špoljar M, Dražina T, Šargač J, Kralj Borojević K, Žutinić P, 2012. Submerged macrophytes as a habitat for zooplankton development in two reservoirs of a flow-through system(Papuk Nature Park, Croatia). Ann. Limnol. Int. J. Lim., 48(2): 161–175. Doi: 10.1051/limn/2012005 |
| Špoljar M, Tomljanović T, Dražina T, Lajtner J, Štulec H, Matulić D, Jelena F, 2016. Zooplankton structure in two interconnected ponds:similarities and differences. Croat.J. Fish., 74(1): 6–13. |
| Teixeira-de Mello F, Meerhoff M, Pekcan-Hekim Z, Jeppesen E, 2009. Substantial differences in littoral fish community structure and dynamics in subtropical and temperate shallow lakes. Freshwater Biol., 54(6): 1 202–1 215. Doi: 10.1111/fwb.2009.54.issue-6 |
| van Leeuwen E, Lacerot G, van Nes E H, Hemerik L, Scheffer M, 2007. Reduced top-down control of phytoplankton in warmer climates can be explained by continuous fish reproduction. Ecol. Model., 206(1-2): 205–212. Doi: 10.1016/j.ecolmodel.2007.03.039 |
| Wang J J, 1961. Freshwater Rotifer Fauna in China. Science Press, Beijing, China288p. |
| Xu H, Paerl H W, Qin B Q, Zhu G W, Gao G, 2010. Nitrogen and phosphorus inputs control phytoplankton growth in eutrophic Lake Taihu, China. Limnol. Oceanogr., 55(1): 420–432. Doi: 10.4319/lo.2010.55.1.0420 |
| Zhang S Y, Liu A F, Ma J M, Zhou Q H, Xu D, Chen S P, Zhao Q, Wu Z B, 2010b. Changes in physicochemical and biological factors during regime shifts in a restoration demonstration of macrophytes in a small hypereutrophic Chinese lake. Ecol. Eng., 36(12): 1 611–1 619. Doi: 10.1016/j.ecoleng.2010.05.006 |
| Zhang S Y, Zhou Q H, Xu D, Lin J D, Chen S P, Wu Z B, 2010a. Effects of sediment dredging on water quality and zooplankton community structure in a shallow of eutrophic lake. J. Environ. Sci., 22(2): 218–224. Doi: 10.1016/S1001-0742(09)60096-6 |
 2018, Vol. 36
2018, Vol. 36