Institute of Oceanology, Chinese Academy of Sciences
Article Information
- XIA Dandan(夏丹丹), MA Aijun(马爱军), HUANG Zhihui(黄智慧), SHANG Xiaomei(商晓梅), CUI Wenxiao(崔文晓), YANG Zhi(杨志), QU Jiangbo(曲江波)
- Molecular characterization and expression analysis of Lilytype lectin (SmLTL) in turbot Scophthalmus maximus, and its response to Vibrio anguillarum
- Chinese Journal of Oceanology and Limnology, 36(2): 508-518
- http://dx.doi.org/10.1007/s00343-017-6268-1
Article History
- Received Oct. 3, 2016
- accepted in principle Oct. 31, 2016
- accepted for publication Nov. 30, 2016
2 Ocean University of Shanghai, Shanghai 201306, China;
3 Laboratory for Marine Biology and Biotechnology, Qingdao National Laboratory for Marine Science and Technology, Qingdao 266071, China;
4 Yantai Tianyuan Aquatic Limited Corporation, Yantai 264006, China
The skin of fishes is covered with mucus (Suzuki et al., 2003). The mucosal layer is the first line of defense against the invasion of foreign substances and pathogenic microorganisms, and serves as a main organ of defense (Suzuki et al., 2003). The main constituent of this barrier is a mucous gel that forms a layer which covers the epithelial cells (Van der Marel et al., 2010). The mucous layer is secreted by various epidermal or epithelial mucus cells, such as goblet cells (Shephard, 1994; Spitzer and Koch, 1998). The skin mucus layer is mainly composed of water and mucins, which are glycoproteins that contain high molecular weight oligosaccharides. Skin mucus is involved in fish respiration, osmoregulation, reproduction, locomotion, defense against microbial infection, disease resistance, excretion, and communication (Shephard, 1994; Khong et al., 2009). One of the most interesting functions of fish mucus is related to its role in the immune response and disease resistance (Guardiola et al., 2014); however, further characterization of this role is needed.
Lectins are not enzymes but able to bind to carbohydrate, are present in the mucus (Ingram, 1980; Alexander and Ingram, 1992), and are capable of defending against pathogens (Suzuki et al., 2003). The biological effects of lectins primarily occur via the binding of the active site of the lectin chain to carbohydrates (Ke et al., 2005). Fish lectins are mediators of non-self recognition in a variety of biological processes. Specifically, fish lectins are involved in the identification and stimulation of pathogen uptake by phagocytes, the facilitation of innate complement-mediated cell lysis, and the enhancement of natural killer cell activity (Hoffmann et al., 1999; Sharon and Lis, 2004; Kim et al., 2011). There are already several researches of lectin in different fishes, including Conger myriaster (Kamiya et al., 1988; Shiomi et al., 1989), Repomucenus richardsonii (Shiomi et al., 1990), Misgurnus anguillicaudatus (Goto-Nance et al., 1995), Genypterus capensis (Toda et al., 1996), Anguilla japonica (Tasumi et al., 2002), and Channa striata (Arasu et al., 2013).
Based on their distinct structures and functions, lectins have been classified as galectins, C-type lectins, lily-type lectins, and rhamnose-binding lectins (Kim et al., 2011; Suzuki et al., 2003). Pufflectin, a mannosespecific lectin purified from the skin mucus of puffish by Tsutsui (2003), was named lily-type lectin (LTL). While LTL shares no significant sequence similarity with any known animal lectins, it surprisingly shares sequence homology with mannose-binding lectins of monocotyledonous plants (Suzuki et al., 2003) such as Galanthus nivalis (Van Damme et al., 1991), Clivia miniata (Van Damme et al., 1994), Allium prorrum (Van Damme et al., 1993), and Allium sativum (Smeets et al., 1997). The lectins of these monocotyledonous plants all contain a specific mannose-binding domain (QxDxNxVxY). Interestingly, the amino acid sequence of pufflectin contains two of these characteristic motifs (Tsutsui et al., 2003). Channa striata lily-type lectin (CsLTL-1) was demonstrated to contain two QxDxNxVxY mannose binding sites (Abirami Arasu, 2013). Other studies have also reported the existence of QxDxNxVxY mannose binding sites in fish lectins (Chandra, 1999; Tsutsui, 2003; Tsutsui et al., 2006). These previous reports provide an important basis for the study of Scophthalmus maximus lily-type lectin (SmLTL).
Turbot (Scophthalmus maximus) is an important farmed fish species with high commercial value in northern China and Europe. Since turbot was introduced to China in 1992, breeding scope has been constantly expanding (Huang et al., 2011). One of the most important factors influencing turbot growth is the variety of pathogens (fungi, bacteria, viruses, and parasites) which cause adverse developmental effects and high rates of mortality in this species. Vibriosis is one of the most disturbing bacterial diseases in turbot aquaculture, which have caused great damage and economic loss in aquaculture production among the world (Saulnier, 2000). Vibrio anguillarum is one of the major pathogens causing vibriosis (Toranzo, 1997). The anti-V. anguillarum response deserves more attention in order to develop more effective methods of preventing V. anguillarum. In order to efficiently manage disease and provide theory support for the enhancement of aquaculture production, immune mechanism should be thoroughly studied in turbot. Previous proteomic studies in this laboratory revealed that SmLTL protein expression was significantly changed following high temperature stress in turbot (Ma et al., 2013). In this study, the corresponding proteome maps were constructed by two-dimensional gel electrophoresis (2-DE), from which the peptide mass map with matrix-assisted laser desorption / ionization tandem time-of-flight (MALDI-TOF-TOF) was obtained, and SmLTL protein was identified by database retrieval. The partial protein sequence of SmLTL was also identified by mass spectrometry analysis. The full-length sequence, expression, and structure prediction analysis of SmLTL has not been previously published. In the current study, SmLTL was cloned, spatially analyzed, and its tertiary structure predicted for the first time. Furthermore, in turbot (Scophthalmus maximus) aquaculture, vibriosis is one of the most disturbing bacterial diseases which have caused great damage and economic loss in aquaculture production among the world (Saulnier, 2000). Vibrio anguillarum is one of the major pathogens causing vibriosis (Toranzo et al., 1997). Antibiotics are used as traditional strategy for fish disease, while it could due to the development and spread of antibiotic resistant pathogens which would have negative impacts on environment and human health (Chen et al., 2016). So we need an efficient and safe method to solve this problem. Recently many innate immune actors in turbot have been characterized, such as Stomatin-like protein2 (Chi, 2016), chemokines (Meng et al., 2013; Chen, 2015), MyD88 (Lin et al., 2015) and lysozyme (Gao et al., 2016). SmLTL was one kind of innate immune actors, could be one of the most important players on the mucus for host protection.
2 MATERIAL AND METHOD 2.1 FishHealthy turbot ((90±10.2) g) were obtained from the Tianyuan Fisheries Co. Ltd. (Yantai, China). Tissue samples, including head-kidney, kidney, liver, spleen, intestine, muscle, gill, and skin, were dissected from euthanized fish and immediately frozen in RNA holder (Tiangen Biotech Co. Ltd., Beijing, China) and stored at -80℃ until use.
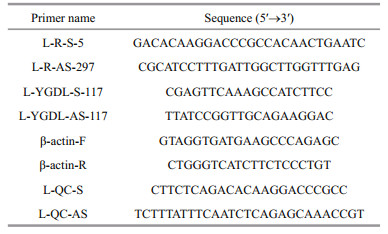
2.2 Cloning and sequencing of SmLTLAccording to the SmLTL protein (Ma et al., 2013), we designed primers (L-R-S-5 and L-R-AS-297) for rapid-amplification of cDNA ends (RACE) (Table 1). Total RNA was isolated from fish skin using an RNAprep Tissue Kit (Tiangen). The first-strand cDNA was synthesized from total skin RNA using the SMART RACE cDNA amplification kit (Clontech, Mountain View, CA, USA) for 3′-RACE with primers L-R-S-5 and L-R-AS-297. The cDNA was stored at -20℃ prior to further analysis. The polymerase chain reactions (PCR) consisted of denaturation at 94℃ for 2 min, 35 cycles of amplification (94℃ for 30s, 63℃ for 30 s, 72℃ for 30 s), and a final extension at 72℃ for 2 min. The same procedures were followed for cDNA production for 5′-RACE. All samples were analyzed in triplicate. Cloning and sequencing was performed by Sangon Biotech Co. Ltd. (Shanghai, China) after agarose gel electrophoresis separation and recovery of products by TIANgel Midi Purification Kit (Tiangen). Lasergene Seqman software (DNASTAR, Madison, WI, USA) was used for sequence assembly of the full-length sequence from the 5ʹ and 3ʹ terminals. With primer L-QC-S and L-QC-AS (Table 1) to PCR, after agarose gel electrophoresis detection and recovery agarose gel (Tiangen), cloned and sequenced by Sangon Biotech Co., Ltd. of China to confirm the full-length sequence.
The full-length SmLTL sequence was compared with other sequences available in the NCBI database (http://blast.ncbi.nlm.nih.ov/Blast) and the similarities were analyzed. The open reading frame (ORF) and amino acid sequence of SmLTL was obtained using NCBI. The hydrophilicity of the SmLTL protein was analyzed using ProtScale (http://web.expasy.org/protscale/). Sequence identity, similarity and gap percentages were calculated using the FASTA program (http://fasta.bioch.virginia.edu/fastawww2/fastawww.cgi). Signal peptide analysis was performed using SignalP (http://www.cbs.dtu.dk). The domains and motifs were analyzed using ProtScale (http://web.expasy.org/protscale/). Secondary structure was predicted and analyzed using Jpred4 (http://www.compbio.dundee.ac.uk/jpred/). The deduced amino acid sequences were submitted to multiple alignment using DNAman version 8.0 (Lynnon Biosoft, San Ramon, CA, USA). A phylogenetic tree was constructed using the Neighbor Joining method, considering 1 000 bootstrap hits in DNAman. Protein tertiary structure was predicted and inspected using PDBsum Generate (http://www.ebi.ac.uk/thorntonsrv/databases/cgi-bin/pdbsum/GetPage.pl?pdbcode=index.html). The predicted protein model was checked using PROCHECK (http://www.ebi.ac.uk/thorntonsrv/software/PROCHECK/).
2.4 Tissue-specific expression of SmLTLTotal RNA was isolated using an RNAprep Tissue Kit followed by cDNA synthesis using 0.05–5 μg of total RNA. Total RNA was mixed with random primers and RNase-free d H2O, heated to 65℃ for 5 min, placed on ice for 5 min, followed by the addition of 2x TS Reaction Mix and RI Enzyme Mix (TransGen Biotech). The mixture was then incubated at 42℃ for 30 min and then heated to 85℃ for 5 min. The cDNA was stored at -20℃ prior to further analysis. Expression analysis of SmLTL was conducted using qPCR) with the L-YGDL-S-117 / L-YGDL-AS-117 primers using a SYBR Premix Ex Taq Kit (TaKaRa, Dalian, China) according to the manufacturer's instructions. β-actin was amplified with specific F and R primers (Table 1) for use as a reference gene. The PCR consisted of denaturation at 94℃ for 2 min, 35 cycles of amplification (94℃ for 30 s, 63℃ for 30 s, 72℃ for 30 s), and a final extension at 72℃ for 2 min. The reactions were performed using an ABI 7500 Real-time Detection System (Applied Biosystems, Foster City, CA, USA). All samples were analyzed in triplicate.
2.5 Bacterial challengeV. anguillarum was conserved in our laboratory in Yellow Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences. V. anguillarum challenge was carried out as previously reported (Ma et al., 2014). V. anguillarum were inoculated on the sterilized TSB, 28℃ incubated for about 24 h and were harvested in the logarithmic phase of growth, which was monitored by the optical density assay. V. anguillarum cells were washed, resuspended, and diluted to 109 CFU/mL in sterile PBS. Fish were challenged by intraperitoneal injection with nine concentrations liveV. anguillarum(dose=experimental fish body weight (g)×1 μL/g bacteria) per fish, and PBS alone was used as a control. Each group had five fish in each of three replicates (i.e. five per tank and 15 in total). At 0, 2, 4, 6, 8, 16, 24 and 24 h post infection (hpi), the gill, intestine and skin were collected and preserved at -80℃ until subsequent use. The method of tissue-specific expression of V. anguillarum challenge reference 2.4.
2.6 Statistical analysisThe data were expressed as mean±SD and subjected to ANOVA (two-way analysis of variance) to determine differences among treatments. The differences were considered as significant at P < 0.05. All the Statistical analysis was performed using SPSS V 19.0 for windows.
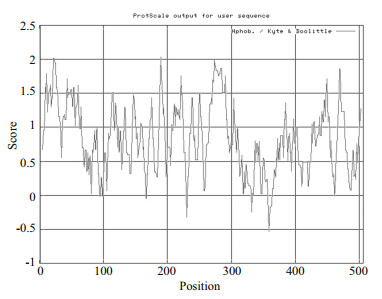
3 RESULT 3.1 Cloning and sequencing of turbot SmLTL cDNAThe full-length SmLTL cDNA was 569 bp long, included 506 bp coding sequence (CDS) (GenBank accession No. KU199003) and contained an ORF of 339 bp that encoded 112 amino acids (Fig. 1). The predicted SmLTL peptide has a theoretical molecular mass of 12.652 3 kDa, an isoelectric point (pI) of 7.86, fat factor of 71.34, and an average hydrophilicity of -0.517. (Fig. 2).

|
| Figure 1 The complete cDNA and deduced amino acid sequence of SmLTL The three specific mannose binding motifs are highlighted in grey. |
|
| Figure 2 Hydrophilicity analysis of SmLTL using the method of Kyte and Doolittle (1982) |
The secondary structure analysis of SmLTL revealed that the protein contains 5.36% alpha helices, 39.29% extended strands, 16.07% beta turns, and 39.29% random coils. The SignalP analysis of the SmLTL amino acid sequence did not reveal the presence of a signal peptide or transmembrane region. The SmLTL protein contains a bulb-type mannose binding lectin (β-lectin) domain between amino acids 3 and 112 (a total of 109 amino acids). Within the β-lectin domain, two mannose binding sites were found between amino acids 30 and 99 with the specific motif of QxDxNxVxY in a three-fold reversed internal repeat (β-prism architecture) (Fig. 1). The first repeat was located at Gln30-Asp32-Asn34-Val36-Tyr38 and the second repeat was located at Gln59-Asp61-Asn63-Val65-Tyr67. The third repeat (TxTxGxRxV) was located at Thr91-Thr93-Gly95-Arg97-Val99. Differences in the amino acid sequence between species were observed within the third repeat region (Fig. 3).

|
| Figure 3 Multiple sequence alignment of SmLTL with other homologous genes: lily-type lectin from Channa striata, Epinepheluscoioides, Esoxlucius, Larimichthys crocea, Leiognathus nuchalis, Lophiomus setigerus, Oncorhynchus mykiss, Oplegnathus fasciatus, and Platycephalus indicus The GenBank accession numbers of the homologous are given in supplementary material. The three D-mannose binding sites are highlighted. |
The sequence identity of SmLTL was compared with other lectin superfamily members, including lily-type lectin, skin mucus lectin, and mannose binding lectin, from different fishes. The SmLTL was found to have highest identity with the lily-type lectin from C. striata (60%), Lophiomus setigerus (53%), Platycephalus indicus (52%), and Larimichthys crocea (50%). The length of the SmLTL sequence was similar to other species, and conserved motifs were observed among the sequences used in the analysis, thus confirming the identity of the gene as SmLTL (Fig. 3).
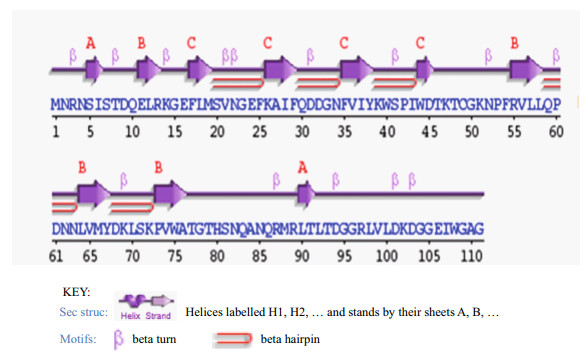
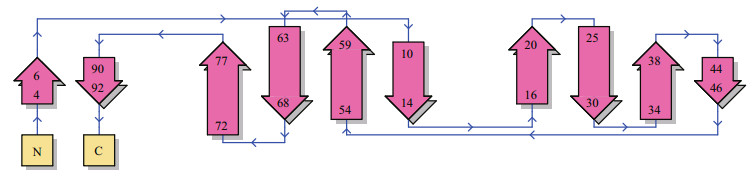
3.2.2 Tertiary structureProtein tertiary structure was predicted and inspected by PDBsum Generate (Fig. 4). The overall folding of SmLTL consisted of three anti-folding β-sheets, comprised of 10 β-strands, 14 β-turns, and 5 β-hairpins (Figs. 5, 6), and contained four protein binding sites (Fig. 7). The mannose-binding sites were located in the clefts formed by the three bundles of β-sheets.

|
| Figure 4 The three-dimensional structure of SmLTL β-sheets are indicated by patches. Turns and loops are indicated by lines. |
|
| Figure 5 Secondary structure of SmLTL contained 10 β-strands, 14 β-turns, and 5 β-hairpins |
|
| Figure 6 Secondary topology of SmLTL The five beta hairpins are located at amino acids 20-25, 30-34, 38-40, 59-63, and 68-72. |

|
| Figure 7 Schematic of the four predicted carbohydrate binding sites of SmLTL Only three of the binding sites are predicted to be functional. |
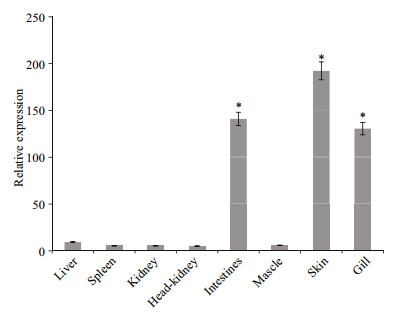
Transcripts for SmLTL were abundant (P < 0.01) in the skin, intestine, and gill. Lower levels of SmLTL transcripts were observed in the liver, head-kidney, spleen, and muscle (Fig. 9).
|
| Figure 9 Tissue distribution of SmLTL mRNA detected by quantitative real time reverse transcriptase PCR SmLTL expression levels in liver, spleen, kidney, head-kidney, intestines, mascle, skin and gill are normalized to that kidney. Vertical bars represent means+SE (n=5). |
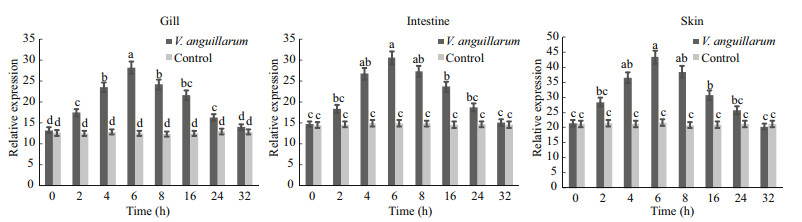
Choosing the gill, intestine, and skin, which was abundant (P < 0.01) in tissue distribution of SmLTL mRNA to challenge V. anguillarum. As shown in Fig. 10 the mRNA expression of SmLTL were upregulated after V. anguillarum challenge, and reached the highest level at 6 h.
|
| Figure 10 SmLTL gene expression in turbot gill, intestine and skin at 0–32 h after V. anguillarum challenge The relative SmLTL expression level as exhibited by 2-ΔΔCt was determined for each group and the values were shown as means±S.D (n=3). Means with different letters indicate statistical significance (P<0.05) between groups. |
In this study, a novel lectin was isolated from turbot (S. maximus) using RACE techniques and was classified as S. maximus lily-type lectin (SmLTL) based on its structural and functional characteristics. Lilytype lectin is able to bind to specific carbohydrates and plays key roles in non-self-recognition and clearance of pathogen (Dodd and Drickamer, 2001; Vasta et al., 2004). The study of SmLTL could be valuable for further identification of mucus lecin in fish.
The full-length SmLTL cDNA that was obtained in this study has a length of 569 bp and contains a 336-bp ORF that encodes a 112-residue protein that lacks a signal peptide. Sequence alignment and phylogenetic analysis (Figs. 3, 8) revealed that SmLTL had high similarity with lily-type lectin-1 from C. striata (60%), L. setigerus (53%), P. indicus (52%), Esox lucius (52%), L. crocea (50%), Oncorhynchus mykiss (42%), and other known lectin sequences from bacteria and plants. These data, together with the structural features of SmLTL, indicate that it belongs to the lily-type lectin family. The tertiary structure model of the SmLTL protein also indicated a high similarity with a variety of β-prism lectins from other species, including Galanthus nivalis (30%), Narcissus pseudonarcissus (27%), Galanthus nivalis (26%), and other monocot plant lectin proteins. The similarities between SmLTL and monocot lectins suggest new areas of research regarding the structure and function of SmLTL.

|
| Figure 8 Phylogenetic tree of lily-type lectins constructed using the Neighbor-Joining method SmLTL is indicated by a black box. The tree is based on an alignment constructed using full-length amino acid sequences using DNAman. The numbers at the branches denote bootsrap majority consensus values of 1 000 replicates. The GenBank accession numbers are provided in the supplementary material. |
The alignment of SmLTL with other lily-type lectins revealed that the mannose-binding motifs of some lectins differ slightly from the standard form (QDNVY); however, these amino acid differences are expected to affect their ability to bind mannose (Afolabi-Balogun et al., 2012; Arasu et al., 2013). In particular, repeat three was found to exhibit slight changes in fish based on current reports. For example, repeat three was reported as TxNxDxQxV in C. striata (Arasu et al., 2013, Genbank: CCQ25776), TxTxDxHxV in E. lucius (Leong et al., 2010, GenBank: ACO14169), and YxRxDxNxV in O. mykiss (Berthelot et al., 2014, GenBank: CDQ78238). However, the other two repeats (one and two) exist in all LTL. These results suggest that these lectin protein motifs are evolutionarily conserved and play an important role in their biological function (Kai et al., 2004). The modification of Arg, Lys, and Ser residues did not modify binding activity, whereas the loss of function following changes in Trp (W) or Asp/ Glu (D/E) and Tyr (Y) residues indicated their crucial role in the binding activity of Colocasia esculenta lectin (Pereira, 2014). The reason for the presence of amino acid residues that a lack of interaction of lectin with mannose may be due to the substitution, deletion, or insertion of key amino acid residues during evolution (Luo et al., 2007). It was speculated that repeat three (TxTxGxRxV) may lead to changes in binding activity based on species-specific and the standard form (QDNVY) determined them combined with mannose. Further studies are required to investigate the binding of carbohydrates by SmLTL.
Three mannose recognition sites were identified in SmLTL, in addition to four identical subunits and three anti-folding β-sheets, which were comprised of 10 β-strands, 14 β-turns, and 5 β-hairpins. The secondary structure analysis of SmLTL revealed that the protein contains 5.36% alpha helices, 39.29% extended strands, 16.07% beta turns, and 39.29% random coils. The whole folding of SmLTL, which typically consists of β-sheets connected by turns and loops, creates a very tight structural scaffold. This is very similar to the 3D structure of other mannosebinding lectins (Barre et al., 2001; Zhao et al., 2003). The monocot β-prism lectin structure also contains three mannose recognition sites and is a homotetrameric protein that is folded in a classic pattern (beta-prism Ⅱ fold) to form its advanced structure (Hester and Wright, 1996). Molecular characterization of SmLTL, such as mannose-binding site analysis, signal cleavage site prediction, and analyses of secondary and 3D structures, indicated that it shares many Exemplary features with monocot mannose-binding lectins. These similarities signify that SmLTL might have similar functions as many other mannose-binding lectins, such as binding to parasites, viruses, and fungi. For example, lectins have been observed to bind Meloidogyne incognita (Bhat et al., 2010), HIV (Ding et al., 2008), HSV-Ⅱ (Luo et al., 2007), and Rhizoctonia solani (Tian et al., 2008). The cloning of SmLTL performed in the current study will enable further research into its potential functions in disease resistance.
The abundance of SmLTL transcripts was highest in the skin, intestine, and gill, but was weak in the liver, head-kidney, spleen, and muscle. The gills in fish are involved in gas exchange and are in continuous contact with the aquatic environment and are, therefore, more susceptible to pathogen infection. The expression of CsLTL-1 mRNA was significantly higher in the gills, liver, intestine, and skin of C. striata (Abirami Arasu, 2013). Similarly, pufflectin mRNA was also widely expressed in the gills, followed by the oral cavity wall, esophagus, and skin of Takifugu rubripes (Suzuki, 2003). Park et al. (2016) also reported that RbLTL transcript was abundant in gill and intestinal tissue in rock bream (Oplegnathus fasciatus). In addition, fish can absorb environmental antigens into the body via the skin (Moore et al., 1998). The lactose-binding lectin in Japanese eel (AJL-2) was demonstrated to be produced only in the skin (Tasumi et al., 2002). According to Suzuki (2003), the intestinal isoform of pufflectin was identified in intestine of T. rubripes. In teleosts, the gut, skin and gill are the main mucosal surfaces and immune barriers (Goel et al., 2015). The high abundance of SmLTL transcripts in the skin, intestine, and gills may reflect the role of this protein in the immune response.
As V. anguillarum is one of the important pathogens responsible for major mortalities in turbot fish, the ability of SmLTL to inhibit it was of significant importance. Here, SmLTL showed the direct activity of facilitating the clearance of V. anguillarum in vivo in turbot. The mRNA expression of gill, skin and intestine in SmLTL were up-regulated after V. anguillarum challenge, and reached the highest level at 6 h. Recently many similar results have showed, such as, a novel C-type lectin (FcLec4) in Chinese white shrimp (Wang et al., 2009), a C-type lectin (AiCTL-3) in bay scallop (Huang et al., 2013), pathogen recognition receptors TLR2 in turbot (Liu et al., 2016). Obviously, mucosal immune stress response was produced after V. anguillarum injection in turbot, and stimulate the secretion of SmLTL. Our results suggest that lily-type lectins serve as the first line of defense against microbial infections and play a pivotal role in the innate mucosal immune system. We intend to further investigate the functions of SmLTL in the mucosal immune system through comparative pathogens studies.
5 CONCLUSIONThe SmLTL from turbot was identified and characterized in this study. The general characteristics of have been reported here, including protein and cDNA sequences, tissue expression profile, domain architectures and V. anguillarum stimulate. The most important result of the present study is that SmLTL not only shares similarity with monocotyledonous plant lectins, but also contains identical mannosebinding sites. However, the function of binding site three of SmLTL requires further study. The information reported here will be useful for the investigation into the multifaceted functions of SmLTL.
| Afolabi-Balogun N B, Inuwa H M, Ishiyaku M F, Bakareodunoola M T, Nok A J, 2012. Isolation and characterization of a mannose-binding insecticidal lectin gene from Allium sativum (garlic) and its putative role in insect resistance using bioinformatics tools. Infect. Genet.Evol., 12(7): 1 508–1 512. Doi: 10.1016/j.meegid.2012.05.008 |
| Alexander J B, Ingram G A, 1992. Noncellular nonspecific defense mechanism of fish. Annual Review of Fish Diseases, 2: 249–279. Doi: 10.1016/0959-8030(92)90066-7 |
| Arasu A, Kumaresan V, Sathyamoorthi A, Palanisamy R, Prabha N, Bhatt P, Roy A, Thirumalai M K, Gnaname A J, Pasupuleti M, Marimuthu K, Arockiaraj J, 2013. Fish lily type lectin-1 contains β-prism architecture:immunological characterization. Mol. Immunol., 56(4): 497–506. Doi: 10.1016/j.molimm.2013.06.020 |
| Barre A, Bourne Y, Van Damme E J M, Peumans W J, Rougé P, 2001. Mannose-binding plant lectins:different structural scaffolds for a common sugar-recognition process. Biochimie, 83(7): 645–651. Doi: 10.1016/S0300-9084(01)01315-3 |
| Berthelot C, Brunet F, Chalopin D, Juanchich A, Bernard M, Noël B, Bento P, Da Silva C, Labadie K, Alberti A, Aury J M, Louis A, Dehais P, Bardou P, Montfort J, Klopp C, Cabau C, Gaspin C, Thorgaard G H, Boussaha M, Quillet E, Guyomard R, Galiana D, Bobe J, Volff J N, Genêt C, Wincker P, Jaillon O, Crollius H R, Guiguen Y, 2014. The rainbow trout genome provides novel insights into evolution after whole-genome duplication in vertebrates. Nat. Commun., 5: 3 657. |
| Bhat G G, Shetty K N, Nagre N N, Neekhra V V, Lingaraju S, Bhat R S, Inamdar S R, Suguna K, Swamy B M, 2010. Purification, characterization and molecular cloning of a monocot mannose-binding lectin from Remusatia vivipara with nematicidal activity. Glycoconj. J., 27(3): 309–320. Doi: 10.1007/s10719-010-9279-0 |
| Chandra N R, Ramachandraiah G, Bachhawat K, Dam T K, Surolia A, Vijayan M, 1999. Crystal structure of a dimeric mannose-specific agglutinin from garlic:quaternary association and carbohydrate specificity. J. Mol. Biol., 285(3): 1 157–1 168. Doi: 10.1006/jmbi.1998.2353 |
| Chen S R., Tang J X, Cheng J M, Li J, Jin C, Li X Y, Deng S L, Zhang Y, Wang X X, Liu Y X, 2015. Loss of gata4 in sertoli cells impairs the spermatogonial stem cell niche and causes germ cell exhaustion by attenuating chemokine signaling. Oncotarget, 6(35): 37 027. |
| Chen Y D, Zhou S H, Jiang Z Q, Wang X L, Liu Y, 2016. Chemokine receptor CXCR3 in turbot (Scophthalmus maximus):cloning, characterization and its responses to lipopolysaccharide. Fish Physiol. Biochem., 42(2): 659–671. Doi: 10.1007/s10695-015-0167-1 |
| Chi H, Hu Y H, 2016. Stomatin-like protein 2 of turbot Scopthalmus maximus:gene cloning, expression profiling and immunoregulatory properties. Fish & Shellfish Immun., 49: 436. |
| Ding J J, Bao J K, Zhu D Y, Zhang Y, Wang D C, 2008. Crystallization and preliminary x-ray diffraction analysis of a novel mannose-binding lectin with antiretroviral properties from Polygonatum cyrtonema hua. Protein Pept. Lett., 15(4): 411–414. Doi: 10.2174/092986608784246551 |
| Dodd R B, Drickamer K, 2001. Lectin-like proteins in model organisms:implications for evolution of carbohydratebinding activity. Glycobiology, 11(5): 71R–79R. Doi: 10.1093/glycob/11.5.71R |
| Gao C B, Fu Q, Zhou S, Song L, RenY C, Dong X Y, Su B F, Li C, 2016. The mucosal expression signatures of g-type lysozyme in turbot (Scophthalmus maximus) following bacterial challenge. Fish Shellfish Immunol., 54: 612–619. Doi: 10.1016/j.fsi.2016.05.015 |
| Goel C, Barat A, Pande V, Sahoo P K, 2015. Molecular cloning and characterization of mannose binding lectin homologue from snow trout (Schizothorax richardsonii). The protein J., 34(1): 1–8. Doi: 10.1007/s10930-014-9591-9 |
| Goto-Nance R, Watanabe Y, Kamiya H, Ida H, 1995. Characterization of lectins feom the skin mucus of the Lonach Misgurnus anguillicaudatus. Fish. Sci., 61(1): 137–140. Doi: 10.2331/fishsci.61.137 |
| Guardiola F A, Cuesta A, Arizcun M, Meseguer J, Esteban M A, 2014. Comparative skin mucus and serum humoral defence mechanisms in the teleost gilthead seabream(Sparus aurata). Fish Shellfish Immunol., 36(2): 545–551. Doi: 10.1016/j.fsi.2014.01.001 |
| Hester G, Wright C S, 1996. The mannose-specific bulb lectin from Galanthus nivalis (snowdrop) binds mono-and dimannosides at distinct sites. Structure analysis of refined complexes at 2.3 Å and 3.0 Å resolution. J. Mol. Biol., 262(4): 516–531. Doi: 10.1006/jmbi.1996.0532 |
| Hoffmann J A, Kafatos F C, Janeway C A, Ezekowitz R A B, 1999. Phylogenetic perspectives in innate immunity. Science, 284(5418): 1 313–1 318. Doi: 10.1126/science.284.5418.1313 |
| Huang M M, Song X Y, Zhao J M, Mu C K, Wang L L, Zhang H, Zhou Z, Liu X L, Song L S, 2013. A C-type lectin(AiCTL-3) from bay scallop Argopecten irradians with mannose/galactose binding ability to bind various bacteria. Gene, 531(1): 31–38. Doi: 10.1016/j.gene.2013.08.042 |
| Huang Z H, Ma A J, Wang X A, 2011. The immune response of turbot, Scophthalmus maximus (L.), skin to high water temperature. J. Fish Dis., 34(8): 619–627. Doi: 10.1111/jfd.2011.34.issue-8 |
| Ingram G A, 1980. Substances involved in the natural resistance of fish to infection-a review. J. Fish Biol., 16(1): 23–60. Doi: 10.1111/jfb.1980.16.issue-1 |
| Kai G Y, Zhao L X, Zheng J G, Zhang L, Miao Z Q, Sun X F, Tang K X, 2004. Isolation and characterization of a new mannose-binding lectin gene from Taxus media. J. Biosci., 29(4): 399–407. Doi: 10.1007/BF02712111 |
| Kamiya H, Muramoto K, Goto R, 1988. Purification and properties of agglutinins from conger eel, Conger myriaster (Brevoort), skin mucus. Dev. Comp. Immunol., 12(2): 309–318. Doi: 10.1016/0145-305X(88)90007-9 |
| Ke J Y, Chen Y S, Rao X Z, 2005. Lectins and its biological function. J. Ningde Teach. Coll. Nat. Sci., 17(1): 19–22. |
| Khong H K, Kuah M K, Jaya-Ram A, Shu-Chien A C, 2009. Prolactin receptor mRNA is upregulated in discus fish(Symphysodon aequifasciata) skin during parental phase. Comp. Biochem. Physiol. B:Biochem. Mol. Biol., 153: 18–28. Doi: 10.1016/j.cbpb.2009.01.005 |
| Kim B S, Nam B H, Kim J W, Park H J, Song J H, Park C I, 2011. Molecular characterisation and expression analysis of a fish-egg lectin in rock bream, and its response to bacterial or viral infection. Fish & Shellfish Immun., 31(31): 1 201–1 207. |
| Kyte J, Doolittle R F, 1982. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol., 157(1): 105–132. Doi: 10.1016/0022-2836(82)90515-0 |
| Leong J S, Jantzen S G, von Schalburg K R, Cooper G A, Messmer A M, Liao N Y, Munro s, Moore R, Holt R A, Jones S J M, Davidson W S, Koop B F, 2010. Salmo salar and Esox lucius full-length cDNA sequences reveal changes in evolutionary pressures on a posttetraploidization genome. BMC Genomics, 11(1): 279. Doi: 10.1186/1471-2164-11-279 |
| Lin J Y, Hu G B, Yu C H, Li S, Liu Q M, Zhang S C, 2015. Molecular cloning and expression studies of the adapter molecule myeloid differentiation factor 88 (MyD88) in turbot (Scophthalmus maximus). Dev. Comp. Immunol., 52(2): 166–171. Doi: 10.1016/j.dci.2015.05.013 |
| Liu F Q, Su B F, Gao C B, Zhou S, Song L, Tan F H, Dong X Y, Ren Y C, Li C, 2016. Identification and expression analysis of TLR2 in mucosal tissues of turbot(Scophthalmus maximus L.) following bacterial challenge. Fish Shellfish Immunol., 55: 654–661. Doi: 10.1016/j.fsi.2016.06.047 |
| Luo Y, Xu X, Liu J, Li J, Sun Y, Liu Z, Liu J Z, Van Damme E, Balzarini J, Bao J, 2007. A novel mannose-binding tuber lectin from Typhonium divaricatum (L.) Decne (family Araceae) with antiviral activity against HSV-Ⅱ and antiproliferative effect on human cancer cell lines. BMB Rep., 40(3): 358–367. Doi: 10.5483/BMBRep.2007.40.3.358 |
| Ma A J, Guo J L, Wang X A, Huang Z H, Wang T, Shang X M, 2014. Family selection and estimation of disease resistance in turbot, Scophthalmus maximus. J. Fish. Sci.China, 21(3): 484–493. |
| Ma A J, Huang Z H, Wang X A, 2013. Changes in protein composition of epidermal mucus in turbot Scophthalmus maximus (L.) under high water temperature. Fish Physiol.Biochem., 39(6): 1 411–1 418. Doi: 10.1007/s10695-013-9795-5 |
| Meng Y Q, Liu X F, Liu Y, Chang X Q, Wang X L, Jiang Z Q, 2013. Chemokine receptor genes CCR3 and CCR9 in turbot (Scophthalmus maximus):cloning and tissue distribution. J. Fish. Sci. China, 20(5): 918–930. |
| Moore J D, Ototake M, Nakanishi T, 1998. Particulate antigen uptake during immersion immunisation of fish:the effectiveness of prolonged exposure and the roles of skin and gill. Fish Shellfish Immunol., 8(6): 393–408. Doi: 10.1006/fsim.1998.0143 |
| Park H J, Jeong J M, Bae J S, Kim J W, An C M, Min B H, Kim S Y, Myeong J I, Hwang H K, Park C I, 2016. Molecular cloning and expression analysis of a new lily-type lectin in the rock bream, Oplegnathus fasciatus. Dev. Comp.Immunol., 65: 25–30. Doi: 10.1016/j.dci.2016.06.014 |
| Pereira P R, Winter H C, Verícimo M A, Meagher J L, Stuckey J A., Goldstein I J, Paschoalin V M F, Silva J T, 2014. Structural analysis and binding properties of isoforms of tarin, the gna-related lectin from Colocasia esculenta. BBA-Proteins proteome., 1854(1): 20–30. |
| Sharon N, Lis H, 2004. History of lectins:from hemagglutinins to biological recognition molecules. Glycobiology, 14(11): 53R–62R. Doi: 10.1093/glycob/cwh122 |
| Shephard K L, 1994. Functions for fish mucus. Rev. Fish Biol.Fish., 4(4): 401–429. Doi: 10.1007/BF00042888 |
| Shiomi K, Uematsu H, Ito H, Yamanaka H, Kikuchi T, 1990. Purification and properties of a lectin in the skin mucus of the dragonet Repomucenus richardsonii. Nippon Suisan Gakk., 56(1): 119–123. Doi: 10.2331/suisan.56.119 |
| Shiomi K, Uematsu H, Yamanaka H, Kikuchi T, 1989. Purificatioin and characterization of a galactose-binding lectin from the skin mucus of the conger eel Conger myriaster. Comp. Biochem. Physiol. B Comp. Biochem., 92(2): 255–261. |
| Smeets K, Van Damme E J M, Verhaert P, Barre A, Rougé P, Van Leuven F, Peumans W J, 1997. Isolation, characterization and molecular cloning of the mannosebinding lectins from leaves and roots of garlic (Allium sativum L.). Plant Mol. Biol., 33(2): 223–234. Doi: 10.1023/A:1005717020021 |
| Spitzer R H, Koch E A. 1998. Hagfish skin and slime glands. In: Jørgensen J M, Lomholt J P, Weber R E, Malte H eds. The Biology of Hagfishes. Springer, Netherlands. p. 109-132. |
| Suzuki Y, Tasumi S, Tsutsui S, Okamoto M, Suetake H, 2003. Molecular diversity of skin mucus lectins in fish. Comp.Biochem. Physiol. B Biochem. Mol. Biol., 136(4): 723–730. Doi: 10.1016/S1096-4959(03)00178-7 |
| Tasumi S, Ohira T, Kawazoe I, Suetake H, Suzuki Y, Aida K, 2002. Primary structure and characteristics of a lectin from skin mucus of the Japanese eel Anguilla japonica. J.Biol. Chem., 277(30): 27 311. |
| Tian Q, Wang W, Miao C, Peng H, Liu B, Leng F, Dai L, Chen F, Bao J, 2008. Purification, characterization and molecular cloning of a novel mannose-binding lectin from rhizomes of Ophiopogon japonicus, with antiviral and antifungal activities. Plant Sci., 175(6): 877–884. Doi: 10.1016/j.plantsci.2008.09.008 |
| Toda M, Goto-Nance R, Muramoto K, Kamiya H, 1996. Characterization of the lectin from the skin mucus of the Kingklip Genypterus capensis. Fish. Sci., 62(1): 138–141. Doi: 10.2331/fishsci.62.138 |
| Toranzo A E, Santos Y, Barja J L, 1997. Immunization with bacterial antigens:vibrio infections. Dev. Biol. Stand., 90: 93–105. |
| Tsutsui S, Tasumi S, Suetake H, Suzuki Y, 2003. Lectins homologous to those of monocotyledonous plants in the skin mucus and intestine of pufferfish, Fugu rubripes. J.Biol. Chem., 278(23): 20 882–20 889. Doi: 10.1074/jbc.M301038200 |
| Tsutsui S, Tasumi S, Suetake H, Kikuchi K, Suzuki Y, 2006. Carbohydrate-binding site of a novel mannose-specific lectin from fugu (Takifugu rubripes) skin mucus. Comp.Biochem. Physiol. B Biochem. Mol. Biol., 143(4): 514–519. Doi: 10.1016/j.cbpb.2006.01.005 |
| van Damme E J M, De Clercq N, Claessens F, Hemschoote K, Peeters B, Peumans W J, 1991. Molecular cloning and characterization of multiple isoforms of the snowdrop(Galanthus nivalis L.). lectin. Planta, 186(1): 35–43. Doi: 10.1007/BF00201495 |
| van Damme E J M, Smeets K, Engelborghs I, Aelbers H, Balzarini J, Pusztai A, van Leuven F, Goldstein I J, Peumans W J, 1993. Cloning and characterization of the lectin cDNA clones from onion, shallot and leek. Plant Mol. Biol., 23(2): 365–376. Doi: 10.1007/BF00029011 |
| van Damme E J M, Smeets K, Van Leuven F, Peumans W J, 1994. Molecular cloning of mannose-binding lectins from Clivia miniata. Plant Mol. Boil., 24(5): 825–830. Doi: 10.1007/BF00029865 |
| van der Marel M, Caspari N, Neuhaus H, Meyer W, Enss M L, Steinhagen D, 2010. Changes in skin mucus of common carp, Cyprinus carpio L., after exposure to water with a high bacterial load. J. Fish Dis., 33(5): 431–439. Doi: 10.1111/jfd.2010.33.issue-5 |
| Vasta G R, Ahmed H, Odom E W, 2004. Structural and functional diversity of lectin repertoires in invertebrates, protochordates and ectothermic vertebrates. Curr. Opin.Struct. Biol., 14(5): 617–630. Doi: 10.1016/j.sbi.2004.09.008 |
| Wang X W, Zhang X W, Xu W T, Zhao X F, Wang J X, 2009. A novel C-type lectin (FcLec4) facilitates the clearance of Vibrio anguillarum in vivo in Chinese white shrimp. Dev.Comp. Immunol., 33(9): 1 039–1 047. Doi: 10.1016/j.dci.2009.05.004 |
| Zhao X Y, Yao J H, Liao Z H, Zhang H Y, Chen F, Wang L, Lu Y Q, Sun X F, Yu S Q, Tang K X, 2003. Molecular cloning of a novel mannose-binding lectin gene from Arisaema heterophyllum. Plant Sci., 165(1): 55–60. Doi: 10.1016/S0168-9452(03)00126-2 |
 2018, Vol. 36
2018, Vol. 36