Institute of Oceanology, Chinese Academy of Sciences
Article Information
- LI Yalei(李亚雷), LIU Qigen(刘其根), CHEN Liping(陈丽平), ZHAO Liangjie(赵良杰), WU Hao(吴昊), CHEN Liqiao(陈立侨), HU Zhongjun(胡忠军)
- A comparison between benthic gillnet and bottom trawl for assessing fish assemblages in a shallow eutrophic lake near the Changjiang River estuary
- Chinese Journal of Oceanology and Limnology, 36(2): 572-586
- http://dx.doi.org/10.1007/s00343-018-6219-5
Article History
- Received Sep. 4, 2016
- accepted in principle Sep. 17, 2016
- accepted for publication Jan. 17, 2017
2 Xinyang College of Agriculture and Forestry, Xinyang 464000, China;
3 Nanjing Institute of Environmental Science, Ministry of Environmental Protection, Nanjing 210042, China;
4 School of Life Science, East China Normal University, Shanghai 200062, China
Abbreviation: catch per unit effort (CPUE); station-month trawling collections (SMTC); number per unit effort (NPUE); biomass per unit effort (BPUE); contingency table analysis (CTA); cluster analysis (CA); non-metric multidimensional scaling (NMDS); average silhouette width (ASW); normalized gamma (NG); Analysis of Similarity (ANOSIM); Multi-response Permutation Procedure (MRPP); Wilcoxon signed-rank test (WSRT); indicator value (INDVAL); analysis of variance (ANOVA); coefficient of variation (CV); monthly mean air temperatures (MMAT); month group with similar air temperatures (MGSAT)
1 INTRODUCTIONThe attributes (e.g. species richness and composition, relative abundances and biomass, size structure) of fish assemblages are essential to and have been extensively utilized in the theoretical research and application practice, such as environmental assessment, ecological restoration, fishery management, ecological modelling, and food web study. It is a challenging task to unbiasedly estimate the attributes of a fish community in standing waters (Kubečka et al., 2009; Olin et al., 2009)because all sampling methods display more or less selectivity (Bethke et al., 1999). Comparison of the attributes between fishing gears, especially between passive and active one, is most often recommended to gain a 'true picture' of lake fish stocks (Olin and Malinen, 2003; Lapointe et al., 2006; Erős et al., 2009; Kubečka et al., 2009; Olin et al., 2009; Rotherham et al., 2012; Clement et al., 2014).
Trawl and gillnet are both extensively used as fishing tools throughout the world. Gillnet is a passive gear and its catching effectiveness is contingent on fish activity, size and morphology. The size distribution estimates are skewed because small individuals move less and when encounter the net are caught less effectively due to slower speed (Olin et al., 2009). However, catchability of active trawl can be relatively low for large individuals, which may avoid the trawl but not gillnets (Olin et al., 2009). To date, very few studies have dealt with inter-comparison of the two gears in freshwaters and none of them come from the Asian region (Olin and Malinen, 2003; Olin et al., 2009; Rotherham et al., 2012).
In present study, we examine differences in species composition and size structure of fish assemblages sampled monthly with gillnets and bottom trawls in Dianshan Lake, a subtropical and estuarine shallow lake in China. Based on results of previous studies done elsewhere (Olin and Malinen, 2003; Olin et al., 2009; Rotherham et al., 2012), we predicted that: (ⅰ) gillnets and bottom trawls would give different pictures of species composition of fish assemblages; (ⅱ) they would display different pictures of size distribution of fish assemblages, in which large-and small-sized fishes would be underestimated by trawls and gillnets, respectively. Furthermore, we also aimed to compare the effectiveness of trawl and gillnet in sampling species richness and to explore the relationship between catch per unit effort (CPUE) of trawl and gillnet.
2 MATERIAL AND METHOD 2.1 Study areaDianshan Lake (31°04'N–31°12'N, 120°54'E–121°01'E) in the lower reaches of Taihu Lake watershed is a tidal freshwater lake and located in the eastern part of Shanghai City, covering a surface water area of roughly 63 km2 and having a mean water depth of 2.1 m (maximum water depth 3.6 m). It is connected with Changjiang River estuary through Huangpu River with a length of 113 km. Jishui Port and Dazhushe are the two main feeding tributaries of the lake, together contributing about 68% of the total inflow. Lanlu Port is the main drainage outlet, accounting for approximately 71% of the total outflow (Fig. 1). Dianshan Lake has a subtropical monsoon climate, with an annual average air temperature of 15.5℃ and mean rainfall amount of 1 037.7 mm.
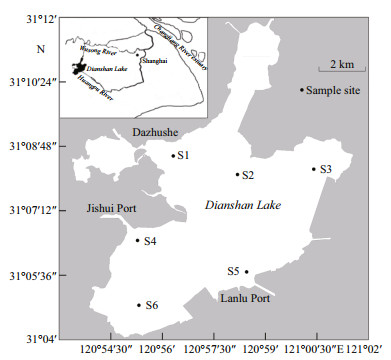
|
| Figure 1 Map of Lake Dianshan showing sampling stations |
Trawling and gillnetting were carried out monthly from August 2009 to July 2010 excluding February 2010 in 6 stations of Dianshan Lake (Fig. 1). At each station, one 150-m benthic multi-panel gill net was set at 03:30–05:30 a.m., and lifted 2 hours later to avoid excessive accumulation of fish in the nets (Erős et al., 2009). Each gill net consisted of six 25-m panels with height of 1–1.5 m and stretch mesh sizes ranging (at 20-mm intervals) from 20 to 120 mm. The trawl was a benthic electric pair-trawl (24 V-Storage Battery, Eagles Ⅲ TZH-2008 Inverter: 20 kW, DC 100 V/220 V/300 V, 300 V was adopted in the study) with a theoretical opening of 1.8 m× 1.4 m and stretch mesh size of 18 mm. Just after set-down of the gill net, the trawl was towed with a speed of 0.8 to 0.9 m/s nearby the gillnetting stations. And the distance between trawl and gillnet sites was about 600 m, which is believed to be safe relating the fish disturbance. Each of 53 station-month trawling collections (SMTC) had 3 replicate hauls, which of 11 SMTC had only 2 replicate hauls because of malfunction of the device of diesel engine or loss of the caught fish samples. Length of each replicate transect towed was approximately 270 m. The towed distance of the third replicate haul of the remaining 2 SMTC deviated greatly from the preseted length of 270 m.
The catches of gillnet and trawl were sorted and counted by species. The total length (TL) and weight of each intact fish was measured to the nearest centimeter and gram, respectively. Fishes were collected at approximately the same time and location of Dianshan Lake using trawl and gillnet. Therefore, it could be assumed that the fish assemblage attributes were obtained by sampling the same fish populations (Bonar et al., 2009).
2.3 Data analysesSimilar to data reporting form of Prchalová et al. (2012), abundance and biomass are expressed as number per unit effort (NPUE) and biomass per unit effort (BPUE), respectively, reported as the number of individuals or grams per 1 000 m2 of gillnets or 1 000 m2 of open water sampled by trawl. According to habitat preference described by Chen (1998) and East China Sea Fisheries Research Institute, et al. (1990), all the fish species collected in Dianshan Lake were divided into two habitat guilds: benthic and nonbenthic (Erős et al., 2009).
Although it is difficult to standardize samples caught with an active and a passive fishing technique, the least-biased evaluation of differences in the number of species collected with the two techniques can be obtained through rarefaction analyses (Erős et al., 2009). In present study, sample-based and individual-based rarefaction analysis is employed to make comparisons of the estimated species richness between gears (Erős et al., 2009, and the references therein), which was represented by the average of three nonparametric statistical estimators: ACE, Chao 1 and Jack 1 (Colwell, 2013). The 90% of the asymptote of sample-based rarefaction curve (i.e., species accumulation curve) was adopted as the desired minimum level of completeness of fish biodiversity (species richness) in Dianshan Lake (Moreno and Halffter, 2000). The difference in species richness between trawling and gillnetting was tested using function c2cv() of R package "rich" with 1 000 randomized runs.
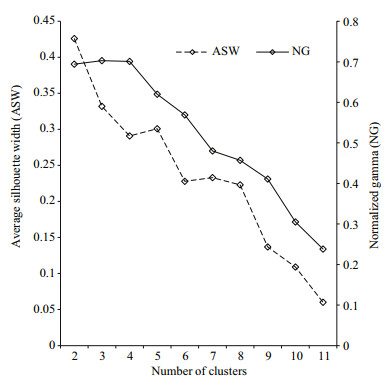
The homogeneity of whole-species distribution in the gears was tested using contingency table analysis (CTA, species × gear) for the caught number of all species (Olin et al., 2009). Based on square-rootarcsine transformed relative abundance, we also used hierarchical agglomerative cluster analysis (CA) and non-metric multidimensional scaling (NMDS), performed with the function hclust() and metaMDS() of R package "vegan", respectively, to identify the difference in fish assemblage composition between the two gears (Erős et al., 2009). The information from the CA was added to the NMDS ordination plot using the function metaMDS(). Average linkage technique in combination with Bray-Curtis distance was used in the CA and the distance was adopted in the NMDS as well. To produce robust results, samples from a certain station were pooled according to the collection technique utilized, and fish species with an overall relative abundance below 1% were combined into a single "rare species" group to prevent the number of variables (i. e., species) from highly exceeding the number of objects (stations) (Erős et al., 2009). Using the function cluster.stats() of R package "fpc", two cluster-validation estimates such as the average silhouette width (ASW) and normalized gamma (NG) were calculated to determine the optimal number of clusters. The greatest values of ASW and NG indicate the most validated clustering (Halkidi et al., 2001; Aho et al., 2008). Bray-Curtis distance metric was further analyzed by ANOSIM (Analysis of Similarity) and MRPP (Multi-response Permutation Procedure) of R package "vegan" to determine if clustering solutions determined by the clustervalidation estimates were statistically significant. ANOSIM produces the R statistic, indicating the degree of cluster separation; R generally ranges between 0, representing no separation and 1, indicating complete separation. Concretely, values of R > 0.75, ≈0.5, and < 0.25 represent fish assemblage groupings either being completely separated, overlapping but noticeably different, or barely separable, respectively (Clarke and Gorley, 2001). MRPP generates the agreement statistic (A), which describes within-group homogeneity. Values of A larger than 0.1 can be used to reject the null hypothesis of no differences among groups (Zweig and Kitchens, 2008). The species-specific differences between the gears were tested using Wilcoxon signed-rank test (WSRT) and indicator species analysis (ISA, Dufrêne and Legendre, 1997). ISA conducted using the function indval() of R package "labdsv" produces an indicator value (INDVAL), ranging from 0 (no indication) to 1 (maximum indication) for each species. The significance of INDVAL was assessed with 1 000 permutations. Only those species with significant (P < 0.05) INDVAL higher than 0.5 were perceived as indicators.
Relative length distributions (percentage value of the NPUE of each 1-cm total length class to the total NPUE) were calculated for each gear. CTA (5-cm total-length class × gear) or WSRT was used to detect for the differences in size structure of 5-cm totallength class, relative frequencies of each of 5-cm total-length classes, and habitat guild between the two gears (Olin et al., 2009). The differences in means of total length and body weight of the whole assemblage and the main fish species between the two gears were detected using two independent samples t-test or Mann-Whitney U test.
Using WSRT, we compared variability of CPUE estimates across sampling stations between gears by monthly calculating the sample coefficient of variation (CV=SD/mean) among stations because there was no replication within stations for gillnetting (Van Den Avyle et al., 1995). A gear that yields high values of CV provided relatively imprecise data. Based on the dataset of 53 SMTC, a two-way analysis of variance (ANOVA) was used to test for significant differences among the seasons and stations in the precision of trawling CPUE estimates computed separately for each station (spring=Mar., Apr., May; summer=Jun., Jul., Aug.; autumn=Sep., Oct., Nov.; winter=Dec., Jan.), and non-parametric Friedman test (multiple paired samples analysis) was conducted to examine differences in the CPUE among the three hauls. Bonar et al. (2009) recommended that comparison of samples collected with passive and active methods should be restricted to larger system-wide (e.g., whole lake) averages. Therefore, we used the stationaveraged NPUE to establish the relationship between trawling and gillnetting NPUE with curve estimation models, such as linear function, logarithmic function, exponential function, inverse function, power law function, quadratic polynomial function, and cubic polynomial function. Correlation analysis between the station-averaged NPUE of trawl and gillnet was also conducted to reveal their relationship. To remove the effect of temperature on their relationship as possible, 11 months were classified into 5 month groups, each with similar monthly mean air temperature. Subsequently, correlation analyses of the NUPE of the whole assemblages and the assemblages excluding the fishes with total length of smaller than 6 cm and larger than 20 cm were conducted between gears for each of the 5 month groups with different air temperatures. The change patterns of trawling and gillnetting NPUE with monthly mean air temperature (MMAT) were modelled with the above functions. The extraordinarily high trawling NPUE in November (c. 274.4 ind./1 000 m2) was identified as outlier (Dixon's test and Grubbs' test, P < 0.05 and < 0.01, respectively) using the function dixon.test() and grubbs.test() of R package "outliers" and this outlier was not included in the regressions (Mehner and Schulz, 2002). The Akaike Information Criterion (AIC) and Schwarz Bayesian Information Criterion (BIC) were used to determine the best regression models. For both AIC and BIC, smallest values indicate better, more parsimonious, models (Quinn and Keough, 2002). According to the values of AIC and BIC of different regression models (Appendix Table A1), quadratic polynomial regression model was most suitable for describing the relationships between trawling and gillnetting NPUE and between gillnetting NPUE and MMAT. Likewise, a cubic polynomial function was most suitable for representing the relationship between the trawling NPUE and MMAT.
All the CA, NMDS, ANOSIM, MRPP, clustervalidation estimates, and CTA indicated that the fish assemblage composition (CTA: χ2=13.081, df=9, P > 0.05, the other results see Appendix Fig.A1, A2, A3 & Table A2) and relative length distribution (CTA: χ2 =3.419, df=6, P > 0.05) of trawling catch did not differ significantly between the first two haul data and the whole data with three hauls from the 53 SMTC. Moreover, no obvious differences were found in the expected species richness obtained with sample-based and individual-based rarefaction analyses between the 53 and 66 SMTC (Fig. 4a, b). Furthermore, Friedman test indicated no differences in the trawling CPUE between different hauls (Section 3.5). Therefore, it is reasonable to use the whole dataset of 66 SMTC to participate in the analyses mentioned in the above paragraphs.
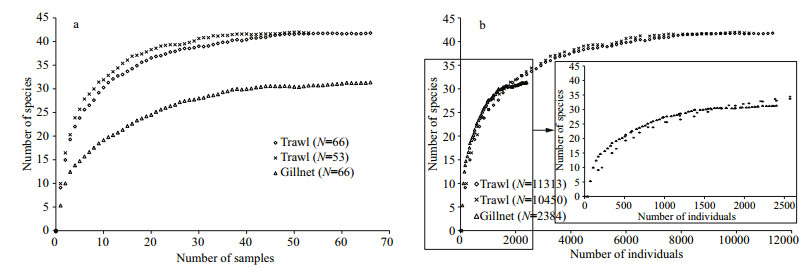
|
| Figure 4 Estimated species richness as function of (a) number of samples and (b) number of individuals caught with gillnetting (open triangle) and trawling (open diamond and fork represent 66 and 53 station-month collections, respectively) The magnified portion in the inserted oblong represents saturation obtained by the individual-based rarefaction curve of gillnet. |
Prior to the above parametric analyses, data were transformed using log, square root, or cube root functions to meet the assumptions of normality and homogeneity of variances. If not yet met, the nonparametric aligned rank transform procedure was used in the two-way ANOVA. Meanwhile, other nonparametric methods such as WSRT and Mann-Whitney U test were applied when the sample size was small or the above assumptions were untenable.
3 RESULT 3.1 Species compositionA total of 40 fish species were caught by the two gears in Dianshan Lake, with 28 and 38 species harvested by gillnets and trawls, respectively. Of which, 26 fish species were shared by the two gears. There were several fish species that were caught exclusively in either gear. Hemiculter leucisculus and Megalobrama amblycephala were only collected by gillnet, whereas 12 species were only caught by trawl. Collectively, trawl gave a more diversified picture of the fish community in this lake. The fish numerical composition of the whole survey year differed significantly between the gears (CTA: χ2=1 891.628, df=10, P < 0.001, Table 1).
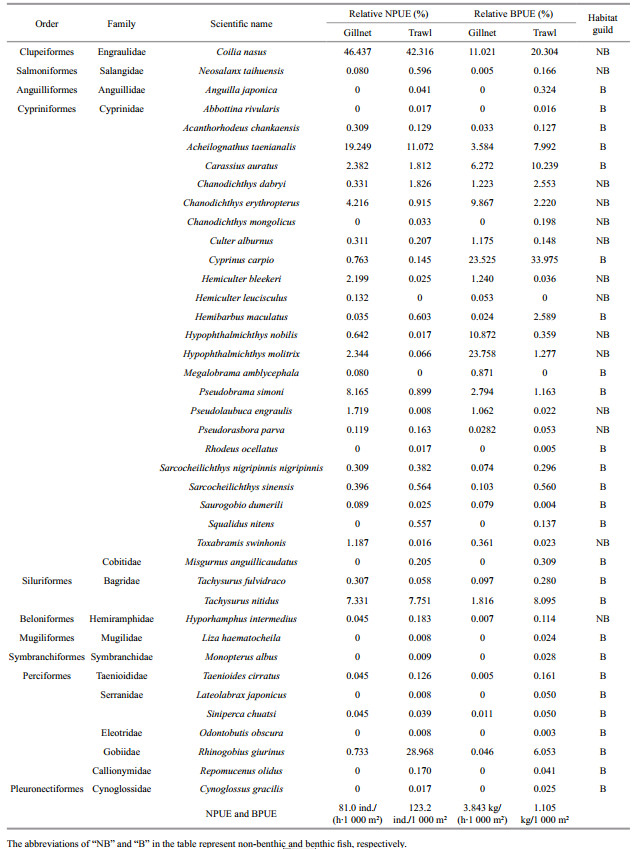
|
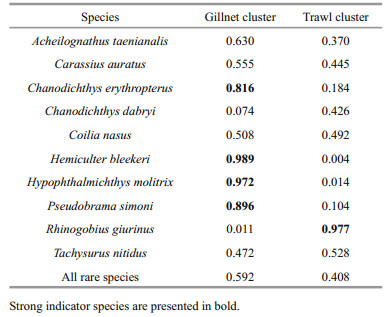
Coilia nasus was the most abundant species both in gillnet and trawl catch. Its proportion by number and biomass did not significantly differ between gears, respectively, and the same was true for Carassius auratus (WSRT, Table 2). Similarly, no difference in the proportion by number of Acheilognathus taenianalis was found between the two gears, whereas its proportion by biomass in trawl catch was significantly larger than in gillnet catch. Correspondingly, the proportions by number and biomass of Tachysurus nitidus and R. giurinus in trawl catch, however, were significantly higher than in gillnet catch. By number of fish caught, gillnets had significantly higher proportion of C. erythropterus, P. simoni, H. molitrix, Hypophthalmichthys nobilis, H. bleekeri, and Cyprinus carpio compared with trawls. The analogous patterns were detected by biomass proportion of the above fish species except for C. carpio.
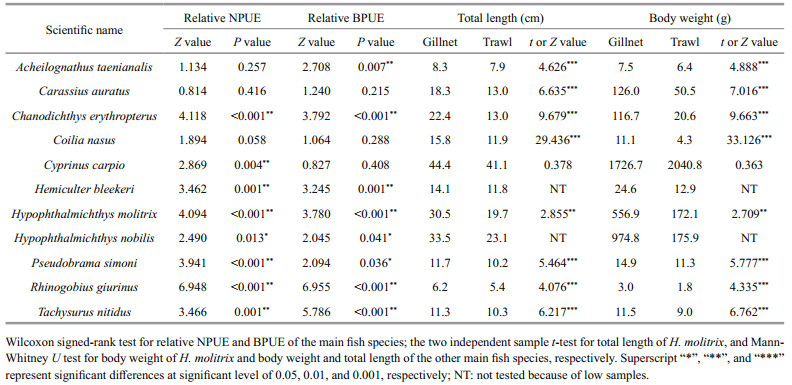
|
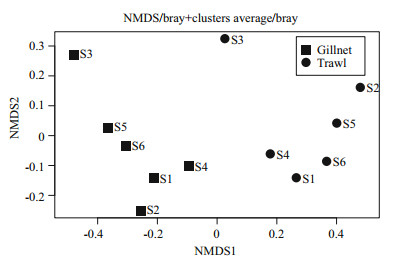
The NMDS solution based on relative NPUE (stress=0.040; non-metric fit: R2=0.998; Fig. 2) was significant. The NMDS result mirrored that of hierarchical clustering (Fig. 2 and Appendix Fig.A4). Combined results of the ASW and NG suggested that two clusters were the best clustering solution for the dataset of relative NPUE (Fig. 3). The MRPP (A=0.301, δ=0.196, P=0.006) and ANOSIM (R=0.828, P=0.003) both revealed significant and complete separation between the two clusters. The strong indicator species (Table 3) for the first division of the dendrogram based on relative NPUE at height of 0.274 (Appendix Fig.A4) were C. erythropterus, H. molitrix, P. simoni and H. bleekeri (gillnet cluster) and R. giurinus (trawl cluster), suggesting that this division separated a gillnet-based fish community (gillnet cluster) from a trawl-based fish community (trawl cluster).
|
| Figure 2 Non-metric multidimensional scaling (NMDS) scatterplot obtained on the basis of relative abundance, with fish groups identified through cluster analysis superimposed Ordination stress=0.040. Results of cluster analysis see Fig.A4 of Appendix. |
|
| Figure 3 The values of a selection of cluster-validation measures inspected in order to determine the most suitable number of clusters in the datasets |
|
The significantly higher species richness (P < 0.001) was observed with trawling (38) than with gillnetting (28). Trawling was consistently more effective in detecting new species than gillnetting, although expected species richness increased with the number of samples for both gears (Fig. 4a). However, gillnetting displayed its effectiveness in detecting new species comparable to trawling when number of individuals caught is lower than about 1 625 and the samples were standardized according to the number. Over that approximate threshold of the number of individuals, the effectiveness of gillnetting became lower than that of trawling (Fig. 4b).
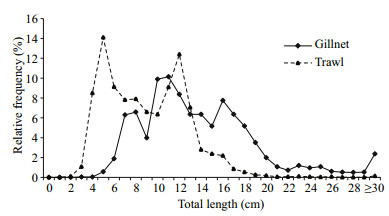
3.3 Size distributionThe overall range in fish length was 3.5–80.0 cm for the gillnet catch and 0.8–73.0 cm for the trawl catch. The total-length distributions differed clearly between the two gears (Fig. 5, CTA: χ2=2348.6, df=6, P < 0.001), whereas the mean total length and body weight for the whole assemblage in gillnet catch were significantly larger than in trawl catch (Mann-Whitney U test: 14.6±0.2 cm vs. 9.6±0.1 cm, Z=38.292, P < 0.001; 56.7±5.6 g vs. 9.0±1.0 g, Z=50.142, P < 0.001; data expressed as mean±SE), respectively. The relative frequencies of the two 5 cm size classes smaller than 10.0 cm in trawl catch were significantly higher than in gillnet catch (WSRT: Z=6.174, P < 0.001 for 0–4.9 cm class; Z=6.427, P < 0.001 for 5.0–9.9 cm class). The reverse was detected for the three 5 cm size classes ranging from 15.0 to 29.9 cm and the class larger than 29.9 cm (WSRT: Z=6.313, 5.681, 5.268 and 4.603 for the four increasing size classes, respectively, all P < 0.001). The proportion of length class of 10.0–14.9 cm did not differ significantly between the gears (WSRT: Z=1.378, P=0.168).
|
| Figure 5 Size distributions of gillnet and trawl catches in Dianshan Lake from August 2009 to July 2010 |
Gillnets rarely caught the small-sized individuals < 6 cm only comprising 0.65% by number of the gillnet catch, whereas individuals < 6 cm comprised 23.82% of the trawl catch by number. More largesized individuals ≥20.0 cm were taken by gillnet than by trawl; they contributed 11.54% of the gillnet catch and 0.80% of the trawl catch by number, respectively.
Gillnet caught H. bleekeri and H. nobilis with larger mean total length and body weight than trawl, but the significance of the differences between the gears was not tested because of the extremely low sample size in trawl catch. For the other 9 main fish species, the species-specific total length and body weight in gillnet catch were significantly larger than in trawl catch except for C. carpio. The body weight of C. carpio in trawl catch, though not significant, was larger than in gillnet catch (two independent sample t-test or Mann-Whitney U test, Table 2).
3.4 Benthic and non-benthic guildsOf the 40 fish species caught by the two gears, 26 belonged to benthic species and 14 non-benthic fish (Table 1). Twenty-five and 15 from the 26 benthic species were captured by trawl and gillnet, respectively, and 13 of the 14 non-benthic species were both captured by the two gears. Non-benthic species accounted for more than half in gillnet catch by number and biomass and there were significant differences in the proportions between non-benthic and benthic species (WSRT: 60.5% vs. 39.5%, Z=2.947, df=65, P < 0.001 for the proportion by number; 61.0% vs. 39.0%, Z=3.282, df=65, P < 0.001 for the proportion by biomass). In trawl catch, however, the proportions by number and biomass of benthic species were significantly higher than those of non-benthic species (WSRT: 52.9% vs. 47.1%, Z=3.469, df=65, P < 0.001 for the proportion by number; 71.9% vs. 28.1%, Z=4.232, df=65, P < 0.001 for the proportion by biomass).
3.5 The relationships between CPUE of the two gears and monthly mean air temperatureThe NPUE and BPUE both did not differed between three hauls (Friedman test, NPUE: χ2=1.895, df=2, P=0.388 > 0.05; BPUE: χ2=0.604, df=2, P=0.739 > 0.05) based on the dataset of 53 SMTC. The trawl catches had CV of 0.446±0.034 (SE) and 0.593±0.044 for NPUE and BPUE of the above dataset, respectively (Fig. 6). The CVs did not differ between stations and seasons for both NPUE (season effect: F=0.847, P=0.480; station effect: F=2.040, P=0.102; season×station interaction: F=0.455, P=0.945) and BPUE (season effect: F=1.212, P=0.323; station effect: F=1.004, P=0.433; season×station interaction: F=0.723, P=0.743) of trawl catch. When the data of stations in each month were pooled according to gears, the CVs of NPUE and BPUE for gillnet were 0.739±0.057 (SE) and 0.812±0.090, and those for trawl were 0.714±0.103 and 0.980±0.143, respectively (Fig. 6). No significant differences in CV of NPUE (WSRT: Z=0.533, df=10, P=0.594) and BPUE (Z=1.156, df=10, P=0.248) were detected between gears.
|
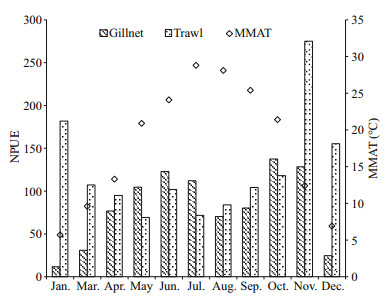
| Figure 6 The monthly changes in gillnet and trawl NPUE expressed as the number of individuals per 1 000 m2 of gillnets or 1 000 m2 of open water sampled by trawl MMAT: monthly mean air temperature. |
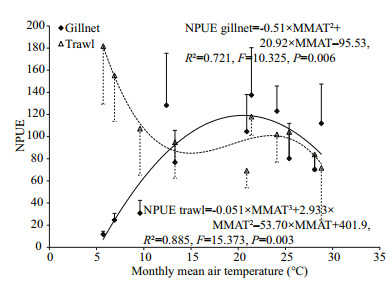
There were strong seasonalities in NPUE of gillnet and trawl. Generally, the relatively higher gillnet catch and lower trawl catch were recorded in warm months and the reverse patterns were found in cold months (Fig. 6). Significantly negative correlation between trawl NPUE and MMAT was detected (r=-0.762, P=0.010, n=10). However, gillnet NPUE was significantly and positively correlated with MMAT (r=0.638, P=0.035, n=11). Regression models showed that trawl NPUE changed as a cubic polynomial function and gillnet NPUE varied as a quadratic polynomial function of MMAT (Fig. 7). With increasing air temperature, gillnet NPUE increased gradually at temperatures below about 20.5℃ and decreased monotonically at temperatures above about 20.5℃, and trawl NPUE declined greatly when temperatures were lower than about 15.1℃ or larger than about 23.2℃ and fluctuated largely at temperatures in-between (Fig. 7).
|
| Figure 7 Change patterns of NPUE of gillnet and trawl with monthly mean air temperature (MMAT) The outlier of trawl NPUE in November is not included in the regression of trawl NPUE. The bars represent standard error. Units of NPUE of gillnet and trawl see Fig. 6. |
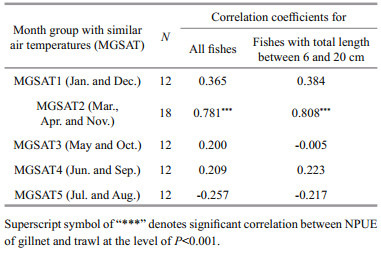
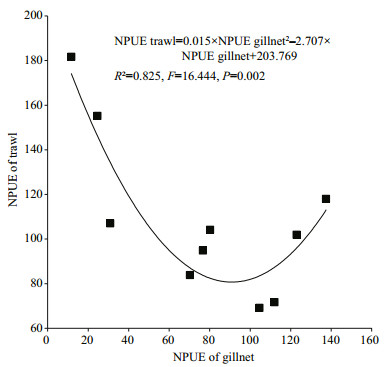
Significant correlation between trawl and gillnet NPUE was detected only for the second month group (MGSAT2) among the five month groups, each with similar air temperature (MGSAT), and their relationship was positive (Table 4). No significant changes in their relationships were found when the fishes with total length of less than 6 cm and more than 20 cm were not included (Table 4). Totally, trawl NPUE was significantly and negatively correlated with gillnet NPUE (r=-0.656, P=0.040, n=10) when both of them in the 6 stations were averaged monthly and the pairwise NPUE data in November were excluded for the outlier of trawl NPUE in this month. The trawl NPUE varied as a quadratic polynomial function of gillnet NPUE, with minimum trawl NPUE at 90.2 ind./1 000 m2/h of gillnet NPUE. With increasing gillnet NPUE, trawl NPUE decreased when gillnet NPUE was lower than 90.2 ind./1 000 m2/h and increased when gillnet NPUE was larger than 90.2 ind./1 000 m2/h (Fig. 8).
|
|
| Figure 8 Regression relationship between trawl and gillnet NPUE The outlier of trawl NPUE in November is not included in the regression. Units of NPUE of gillnet and trawl see Fig. 6. |
Our results showed that the assemblages in Dianshan Lake significantly differed in most of the structural features studied between trawling and gillnetting, including species richness, species numerical composition and size structure. Different pictures of those characteristics between active and passive gear have often been reported by previous studies, such as gillnetting vs. electric fishing (Erős et al., 2009), gillnetting vs. trawling (Olin and Malinen, 2003; Olin et al., 2009; Rotherham et al., 2012), gillnetting vs. seining (Prchalová et al., 2008), and other combining use of passive and active gears (Clement et al., 2014). Results of present study also displayed that bottom trawl was more effective in detecting new species than sink gillnet, gillnet catches could serve as a proxy for density estimated from trawling under a monthly survey procedure or when surveys were conducted in months of March, April and November, and a hump-shaped relationship existed between gillnet catches and MMAT but trawl catches decreased with increasing MMAT both at low and high temperatures.
In Dianshan Lake, the effectiveness of trawling and gillnetting in detecting new species depended largely on sampling effort (i.e., number of samples). Nevertheless, the species accumulation rates levelled off and the observed species richness was very close to or somewhat over 90% of the expected one for both gears, which indicate that the sample size (66 monthstation combinations) is appropriate (Moreno and Halffter, 2000). The results also showed that active trawling produced much more species richness than passive gillnetting (Fig. 4a), which is similar to other comparative studies of active vs. passive gears, such as electric fishing vs. gill netting (Growns et al., 1996; Erős et al., 2009), but this pattern is not always consistent. Although active seine net and passive Windermere trap proved to be the most and least effective among four gears, active boat electrofishing was surpassed by passive hoop net in effectiveness of detecting new species (Lapointe et al., 2006). In Siitinselkä Lake, passive gillnets gave a more diversified picture of fish community than active seines (Jurvelius et al., 2011). There are few comparative studies on the effectiveness of detecting species richness between passive gillnetting and active trawling. Olin and Malinen (2003) and Olin et al. (2009) found that similar species numbers were collected by the two gears, which are inconsistent with our result. This disagreement may be due to the difference in trawl types. Pelagic trawls were used by Olin et al. (2009), whereas bottom ones were employed in our studied lake. Strict benthic species are not caught efficiently since their pelagic trawls were towed slightly above the bottom (Olin et al., 2009). Difference between studies in Finland (Olin and Malinen, 2003; Olin et al., 2009) and results of this study is also in number of species present in sampled lakes. There is low fish species diversity in Finnish lakes, therefore both gears can successfully detect most of them. Species richness tends to increase with increasing collections (Lapointe et al., 2006, and the reference therein), which did so in Dianshan Lake. The species richness expected from individual-based rarefaction analysis was similar for trawling and gillnetting when the number of sampled individuals is lower than approximately 1 625, over which the effectiveness of trawling in detecting new species was higher than that of gillnetting; and finally the rarefaction curves of trawl and gillnet catches both gave saturation (Fig. 4b). Those results from rarefaction curves indicate that trawl is a more effective gear than gillnet in assessing fish species richness in the lake.
Body form and morphology can influence fish capture efficiencies (Bonar et al., 2009). For example, the probability of a fish to encounter and retain in a gillnet increases with discontinuities of body outline (Olin and Malinen, 2003). In Dianshan Lake, many rare species (total relative abundance < 1%) were sampled only by trawl, including Anguilla japonica, Cynogossus gracilis, Misgurnus anguillicaudatus, Monopterus albus, and Repomucenus olidus. They are strictly benthic inhabitants, with flattened-or rodshaped, or eel-like body form and smooth body due to the presence of large amounts of skin mucus, both making them more difficult to be caught by gillnets. Similarly, European eel (A. anguilla) is also not easy to catch by gillnets because of its smooth body and admirable motor abilities (Prchalová et al., 2013). Catchability of deep-bodied species like bream (Abramis brama) is relatively poor for gillnets compared with that for trawls (Hamley, 1975; Olin et al., 2009; Šmejkal et al., 2015). A. taenianalis is also a deep-bodied species. However, it was the second and third dominant species by number in the gillnet and trawl catches of Dianshan Lake, respectively, and no difference in its numerical proportion was detected between gears, which might be due to the relatively smaller body size of this species compared with that of bream.
Selectivity of gears depends also on fish activity, which makes its catchabilities different between gears. For example, predatory fishes have more probabilities of encountering gillnets due to necessarily travelling more distance to seek prey and of escaping from catching by trawl because of having high swimming speed. Therefore, catchability of predatory species is more effective by gillnets than by trawls (Olin and Malinen, 2003), which can be supported by the strong indicator predatory species C. erythropterus in gillnet catch of Dianshan Lake (Table 2). The relative NPUE of this species in gillnet catch was significantly larger than in trawl catch (Table 1).
The efficiency of most sampling methods is influenced by fish size and its associated activity. Particularly, gillnet and trawl produce high selectivity over fish size. Olin et al. (2009) and Jurvelius et al. (2011) found that gillnet underestimated small-sized fish species such as smelt and the density of < 6 cm small-sized individuals compared with trawl. In Dianshan Lake, gillnet also underestimated smallsized fish compared with trawl, which was demonstrated by the significantly less relative frequencies of 5-cm total length classes smaller than 10 cm and relative NPUE of R. giurinus in gillnet catch than in trawl catch. This unique strong indicator species of trawl catches has the smallest total length among the 40 caught species in the lake. Moreover, the percent of < 6 cm individuals in gillnet catch was very low (0.65%), whereas the proportion in trawl catch was quite high (23.82%). Three factors might be ascribed to the underestimation of gillnet. First, small-sized individuals would swim through gillnet when their maximum perimeters were less than the perimeters of the mesh (Olin et al., 2009), which may do better because the mesh size (20 mm) of gillnet pane with minimum mesh size adopted by us is larger than that (5 mm, from knot to knot) used by Olin et al. (2009). Second, small-sized individuals have poor swimming ability and thus travel short distance, which may reduce their probabilities of encountering gillnets and thus their catchability by gillnets. However, active trawl has an advantage in catching small-sized species or individuals due to their poor activity and low swimming speed. Mechanic parameters of gillnet of different mesh sizes and thread are also important and often affect the catchability of gillnets (Prchalová et al., 2009). Furthermore, mesh obstruction may also be responsible for the difference in the numerical composition of small-sized individuals between gears. According to our observations, trawl meshes were usually clogged by detritus, submerged macrophyte, and dead or alive mollusks etc. during the process of trawling. As a result, even < 1 cm individuals also could be caught by trawl. Thus, the subtle difference in mesh size between trawl (18 mm) and gillnet pane of minimum mesh (20 mm) is probable not the main reason for the difference in size structure between gears.
Conversely, gillnet is prone to catch big-sized individuals or species. In Dianshan Lake, the effectiveness in catching fishes ≥20 cm was very low for trawl (0.80%) but relatively high for gillnet (11.54%). Meanwhile, the total length and/or body weight of the whole assemblages and most of the main species, and the relative frequencies of 5-cm total length classes larger than 15 cm in gillnet catch were significantly larger than in trawl catch. In addition, gillnet strong indicator H. molitrix, a largesized species, was caught more effectively by gillnet than by trawl. Huse et al. (2000) showed that the mean length of cod in gillnet catch was bigger than in trawl catch. Big-sized individuals have a great chance to escape from the trawl and would be underestimated by trawl because they tend to have high swimming speed and strong activity, while they are easily caught by gillnet due to the high speed and thus large travelling distance (Bethke et al., 1999; Olin and Malinen, 2003; Olin et al., 2009; Prchalová et al., 2009). Besides, the relatively small opening size of our trawls could increase the chance of escape of large specimens. It could also be considered that benthic species are generally less active than pelagic species and thus are underestimated in gillnet catches.
It is not easy to obtain reliable fish stock estimates even though different sampling methods are used (Dahm et al., 1992). Nevertheless, comparative studies still have been made largely since then. Most of these studies have focused on comparisons between gillnet catches and hydroacoustic estimates of a single, several fish species or fish assemblage, and only a few studies have correlated gillnet and trawl catches (Prchalová et al., 2012). On the whole, their correlation has not always been confirmed (Peltonen et al., 1999; Deceliere-Vergès and Guillard, 2008). No or weak correlations can be found in most comparative studies (Van Den Avyle et al., 1995; Deceliere-Vergès and Guillard, 2008, Jurvelius et al., 2011; Achleitner et al., 2012; Dennerline et al., 2012; Prchalová et al., 2012). In Dianshan Lake, a significant correlation between gillnet and trawl density estimates was only found for one of the five month groups with different temperatures (Table 4). Good correlations in some studies can be detected when the individuals with the smallest and/or largest body length were excluded (e.g. Van Den Avyle et al., 1995; Elliott and Fletcher, 2001; Mehner and Schulz, 2002; Olin and Malinen, 2003; Olin et al., 2009). However, correlation coefficients between gillnet and trawl catches in Dianshan Lake did not change significantly when we removed the smallest and largest fishes compared with no removal, as done by Olin et al. (2009) (Table 4). First, although it is generally accepted that the gillnet catch is dependent on a given fish density, enhanced gillnet catches are more related to increased fish activity than to the density recorded by trawling (Prchalová et al., 2012). Second, many factors such as net saturation, fish escapement, and avoidance of/attraction to a gillnet with already enmeshed fish play a significant role during every gillnet sampling (Olin et al., 2004; Prchalová et al., 2011), which do not cause gillnet catchability to inevitably vary linearly with fish density (Prchalová et al., 2012).
Furthermore, sampling precision of density estimates may also be responsible for correlations of abundance estimates between the two gears used by us. Density estimates by trawl with mean CV of 0.446 in Dianshan Lake, similar to that (0.433) of trawl catches of threadfin in tropical reservoirs (Prchalová et al., 2012) and less than those of threadfin and gizzard shad in Texoma Lake ranging from 0.4 to 1.7 (Van Den Avyle et al., 1995), can be considered to have adequate precision (Casado and Cutillas, 2011). Nevertheless, the CVs for trawl samples in Dianshan Lake ranged from 0.039 to 1.105, with 18 of the 53 stations towed three replicates having CVs of more than 0.5, which can be regarded as poor precision (Casado and Cutillas, 2011). The length of each mesh pane of a set of gillnet used by us is about ten times longer than that of European standardized multi-mesh gillnets (EN 14757). The CVs could not be calculated because no replicates for gillnetting at each station in each month were sampled. This shortcoming makes it impossible to reveal the accuracy of gillnet catches and compare it with that of trawl catches. Prchalová et al. (2012) found that sampling precision for gillnet catches of threadfin shad was significantly larger than that for trawl catches. Substantial variability of gillnet catches is thought to be caused by predators attacking threadfin shad enmeshed in gillnets and by uneven spatial distribution due to the schooling of shad, but trawl has the potential to reduce the natural variability caused by uneven distribution (Prchalová et al., 2012). Engraulid fishes including C. nasus, a predominant fish in Dianshan Lake, have schooling behavior (Young et al., 1994). It is speculated that the accuracy of trawl catches would be larger than that of gillnet catches when sampling replicates were set and enough for both gears, even though the two gears had comparable mean CV when CVs were monthly calculated among stations in this study. Therefore, no correlations between gillnet and trawl catches for the four MGSAT (Table 4) could partially be explained by catch variability and adequate replicates for both gears in Dianshan Lake are needed to increase the accuracy of each sampling method. The CPUE often provides an index to fish density (Pope and Willis, 1996). Our results showed that the correlation between NPUE data of fish populations caught by trawls and gill nets in Dianshan Lake was significant when data from stations were pooled. A relatively high goodness of fit was obtained when a quadratic polynomial function was used to describe their relationship. Those results indicate that the gillnet NPUE in the future investigation with the same sampling procedure as that in 2009–2010 has the potential to be transformed to absolute abundance of trawl and to provide an index to fish density if accuracies of the two gears would increase largely after adequate replicates were set up at each station. It is true especially for catching fish in the months with similar temperatures of March, April and November because of high correlation detected between NPUE of gillnet and trawl (Table 4).
Gillnet NPUE of fish assemblage was significantly and positively correlated with MMAT in Dianshan Lake, which is consistent with the findings of previous studies on fish assemblage or a single fish species (Dennerline et al., 2012; Olin et al., 2016). However, in other reports no significant relationships were detected (Hansson and Rudstam, 1995; Tremain and Adams, 1995). Minimum gillnet NPUE was most often recorded in cold month, whereas maximum NPUE was rarely found in "hot month" but mostly observed in months with temperatures in-between (Neumann and Willis, 1995; Tremain and Adams, 1995; Pope and Willis, 1996; Bobori and Salvarina, 2010). In Dianshan Lake, minimum gillnet NPUE occurred in the coldest month (January); high temperatures did not always represent high gillnet NPUE and the opposite is true, which conform to the results of most of the above studies. The relationship between gillnet NPUE and MMAT was strengthened when a quadratic polynomial function was used to describe their relationship; gillnet NPUE rose first and then fell with increasing MMAT. The similar "hump-shaped" patterns were also observed for European perch (Perca fluviatilis) and roach (Rutilus rutilus) in six lakes of south-east Norway (Linløkken and Haugen, 2006). Gillnet NPUE is a function of both fish abundance and catchability at the time of fishing (Hamley, 1975); water temperature can increase fish activity and thus its catchability in gillnets (Hansson and Rudstam, 1995; Tang and Boisclair, 1995; Olin et al., 2016). Therefore, positive correlation between gillnet NPUE and water temperature may be expected and has been found in previous investigations (Linløkken and Haugen, 2006; Olin et al., 2016). However, water temperature not only increases fish activity but also decreases it. For example, swimming speed rate of brook trout (Salvelinus fontinalis) tended to increase with water temperature up to 18℃ and decrease at 20.7℃ (Tang and Boisclair, 1995). Thus, high water temperatures may decrease the distance travelled by fishes during sampling period and thus reduce their catchability, which may be used to explain the second part of "hump-shaped" pattern between gillnet NPUE and temperature obtained by present study and Linløkken and Haugen (2006). Some ecological factors such as water color, oxygen conditions, water clarity and real fish density also have effects on fish catchability in gillnets (Linløkken and Haugen, 2006; Prchalová et al., 2010; Olin et al., 2016). They change seasonally like water temperature, which may complicate the interpretation of the relationship between gillnet NPUE and temperature found by us.
A few relationships between trawl NPUE and water temperature have been reported; Tremain and Adams (1995) found a significantly positive association between trawl catches per haul and water temperature in their monthly sampling procedure. By contrast, significantly negative relationships between them were found in Dianshan Lake, which may be ascribed to the increased escape activity of fishes in months with higher water temperature.
5 CONCLUSIONThe pictures of fish community including species richness, species numerical composition and size structure in Dianshan Lake shown by gillnet and trawl, respectively, are significantly different. Active trawling produces higher species richness than passive gillnetting when sampling size is enough. Bottom trawl is more effective for capturing benthic species than benthic gillnet in the shallow lake due to the fact that body form and morphology could influence fish capture efficiencies of different gears.
The efficiencies of most sampling methods are influenced by fish size and its associated activity. Gillnet captures relatively less small-sized fish but overestimates large-sized fish compared with trawl. Moreover, gillnet NPUE of fish assemblage is significantly and positively correlated with monthly mean air temperatures (MMAT), but significantly negative relationship for trawl is found in Dianshan Lake.
Actually single-gear-based survey (for instance, only gillnet or trawl is adopt) is often misleading in assessments of attributes of fish assemblages. Therefore, several sampling gears are recommended to be adopted jointly to assess the real picture of fish community scientifically and reasonably. According to our results, multi-mesh monofilament gillnets and benthic trawls may be a workable combination to reveal the attributes of fish assemblages in Dianshan Lake and similar lakes in Taihu Lake Basin such as shallow eutrophic lakes. Nevertheless, there is a lot of research work to do to standardize the procedures of each fishing gear.
| Achleitner D, Gassner H, Luger M, 2012. Comparison of three standardised fish sampling methods in 14 alpine lakes in Austria. Fish. Manage. Ecol., 19(4): 352–361. |
| Aho K, Roberts D W, Weaver T, 2008. Using geometric and non-geometric internal evaluators to compare eight vegetation classification methods. J. Veg. Sci., 19(4): 549–562. Doi: 10.3170/2008-8-18406 |
| Bethke E, Arrhenius F, Cardinale M, Håkansson N, 1999. Comparison of the selectivity of three pelagic sampling trawls in a hydroacoustic survey. Fish. Res., 44(1): 15–23. Doi: 10.1016/S0165-7836(99)00054-5 |
| Bobori D C, Salvarina I, 2010. Seasonal variation of fish abundance and biomass in gillnet catches of an East Mediterranean lake:Lake Doirani. J. Environ. Biol., 31(6): 995–1. |
| Bonar S A, Hubert W A, Willis D W, 2009. Standard Methods for Sampling North American Freshwater Fishes. American Fisheries Society, Bethesda, Maryland. |
| Casado P, Cutillas P R, 2011. A self-validating quantitative mass spectrometry method for assessing the accuracy of high-content phosphoproteomic experiments. Mol. Cell.Proteomics., 10(1): M110.003079. Doi: 10.1074/mcp.M110.003079 |
| Chen Y Y, 1998. Fauna Sinica, Osteichthyes, Cypriniformes Ⅱ. Science Press, Beijing, China. |
| Clarke K R, Gorley R N, 2001. PRIMER Version 5.0:User Manual/Tutorial. PRIMER-E Ltd, Plymouth, UK. |
| Clement T A, Pangle K, Uzarski D G, Murry B A, 2014. Effectiveness of fishing gears to assess fish assemblage size structure in small lake ecosystems. Fish. Manage.Ecol., 21(3): 211–219. Doi: 10.1111/fme.2014.21.issue-3 |
| Colwell R K. 2013. EstimateS: statistical estimation of species richness and shared species from samples. Version 9. User's Guide and Application. Persistent URL<purl.oclc.org./estimates>. |
| Dahm E, Hartman J, Jurvelius J, Löffler H, Völzke V, 1992. Review of the European Inland Fisheries Advisory Commission (EIFAC) experiments on stock assessment in lakes. J. Appl. Ichthyol., 8(1-4): 1–9. Doi: 10.1111/jai.1992.8.issue-1-4 |
| Deceliere-Vergès C, Guillard J, 2008. Assessment of the pelagic fish populations using CEN multi-mesh gillnets:consequences for the characterization of the fish communities. Knowl. Managt. Aquatic Ecosyst., 389(4): 1–16. |
| Dennerline D E, Jennings C A, Degan D J, 2012. Relationships between hydroacoustic derived density and gill net catch:implications for fish assessments. Fish. Res., 123-124: 78–89. Doi: 10.1016/j.fishres.2011.11.012 |
| Dufrêne M, Legendre P, 1997. Species assemblages and indicator species:the need for a flexible asymmetrical approach. Ecol. Monogr., 67(3): 345–366. |
| East China Sea Fisheries Research Institute, Chinese Academy of Fisheries Science, Shanghai Fisheries Research Institute, 1990. The Fishes of Shanghai Area. Shanghai Scientific and Technical Publishers, Shanghai. |
| Elliott J M, Fletcher J M, 2001. A comparison of three methods for assessing the abundance of Arctic charr, Salvelinus alpinus, in Windermere (northwest England). Fish. Res., 53(1): 39–46. Doi: 10.1016/S0165-7836(00)00270-8 |
| Erős T, Specziár A, Bíró P, 2009. Assessing fish assemblages in reed habitats of a large shallow lake-a comparison between gillnetting and electric fishing. Fish. Res., 96(1): 70–76. Doi: 10.1016/j.fishres.2008.09.009 |
| Growns I O, Pollard D A, Harris J H, 1996. A comparison of electric fishing and gillnetting to examine the effects of anthropogenic disturbance on riverine fish communities. Fish. Manage. Ecol., 3(1): 13–24. Doi: 10.1111/fme.1996.3.issue-1 |
| Halkidi M, Batistakis Y, Vazirgiannis M, 2001. On clustering validation techniques. J. Intell. Inf. Syst., 17(2-3): 107–145. |
| Hamley J M, 1975. Review of gillnet selectivity. Journal of the Fisheries Research Board of Canada, 32(11): 1 943–1 969. Doi: 10.1139/f75-233 |
| Hansson S, Rudstam L G, 1995. Gillnet catches as an estimate of fish abundance:a comparison between vertical gillnet catches and hydroacoustic abundances of Baltic Sea herring (Clupea harengus) and sprat (Sptattus sptattus). Can. J. Fish. Aquat. Sci., 52(1): 75–83. Doi: 10.1139/f95-007 |
| Huse I, Løkkeborg S, Soldal A V, 2000. Relative selectivity in trawl, longline and gillnet fisheries for cod and haddock. ICES. J. Mar. Sci., 57(4): 1 271–1 282. Doi: 10.1006/jmsc.2000.0813 |
| Jurvelius J, Kolari I, Leskelä A, 2011. Quality and status of fish stocks in lakes:gillnetting, seining, trawling and hydroacoustics as sampling methods. Hydrobiologia, 660(1): 29–36. Doi: 10.1007/s10750-010-0385-6 |
| Kubečka J, Hohausová E, Matěna J, Peterka J, Amarasinghe U S, Bonar S A, Hateley J, Hickley P, Suuronen P, Tereschenko V, Welcomme R, Winfield I J, 2009. The true picture of a lake or reservoir fish stock:a review of needs and progress. Fish. Res., 96(1): 1–5. Doi: 10.1016/j.fishres.2008.09.021 |
| Lapointe N W R, Corkum L D, Mandrak N E, 2006. A comparison of methods for sampling fish diversity in shallow offshore waters of large rivers. North Am. J. Fish.Manage., 26(3): 503–513. Doi: 10.1577/M05-091.1 |
| Linløkken A, Haugen T O, 2006. Density and temperature dependence of gill net catch per unit effort for perch, Perca fluviatilis, and roach, Rutilus rutilus. Fish. Manage.Ecol., 13(4): 261–269. Doi: 10.1111/fme.2006.13.issue-4 |
| Mehner T, Schulz M, 2002. Monthly variability of hydroacoustic fish stock estimates in a deep lake and its correlation to gillnet catches. J. Fish. Biol., 61(5): 1 109–1 121. Doi: 10.1111/jfb.2002.61.issue-5 |
| Moreno C E, Halffter G, 2000. Assessing the completeness of bat biodiversity inventories using species accumulation curves. J. Appl. Ecol., 37(1): 149–158. Doi: 10.1046/j.1365-2664.2000.00483.x |
| Neumann R M, Willis D W, 1995. Seasonal variation in gillnet sample indexes for northern pike collected from a glacial prairie lake. North Am. J. Fish. Manage., 15(4): 838–844. Doi: 10.1577/1548-8675(1995)015<0838:SVIGSI>2.3.CO;2 |
| Olin M, Kurkilahti M, Peitola P, Ruuhijärvi J, 2004. The effects of fish accumulation on the catchability of multimesh gillnet. Fish. Res., 68(1-3): 135–147. Doi: 10.1016/j.fishres.2004.01.005 |
| Olin M, Malinen T, 2003. Comparison of gillnet and trawl in diurnal fish community sampling. Hydrobiologia, 506(1-3): 443–449. |
| Olin M, Malinen T, Ruuhijärvi J, 2009. Gillnet catch in estimating the density and structure of fish community-comparison of gillnet and trawl samples in a eutrophic lake. Fish. Res., 96(1): 88–94. Doi: 10.1016/j.fishres.2008.09.007 |
| Olin M, Tiainen J, Kurkilahti M, Rask M, Lehtonen H, 2016. An evaluation of gillnet CPUE as an index of perch density in small forest lakes. Fish. Res., 173: 20–25. Doi: 10.1016/j.fishres.2015.05.018 |
| Peltonen H, Ruuhijärvi J, Malinen T, Horppila J, 1999. Estimation of roach (Rutilus rutilus (L.)) and smelt(Osmerus eperlanus (L.)) stocks with virtual population analysis, hydroacoustics and gillnet CPUE. Fish. Res., 44(1): 25–36. Doi: 10.1016/S0165-7836(99)00057-0 |
| Pope K L, Willis D W, 1996. Seasonal influences on freshwater fisheries sampling data. Rev. Fish. Sci., 4(1): 57–73. Doi: 10.1080/10641269609388578 |
| Prchalová M, Kubečka J, Říha M, Litvín R, Čech M, Frouzová J, Hladík M, Hohausová E, Peterka J, Vašek M, 2008. Overestimation of percid fishes (Percidae) in gillnet sampling. Fish. Res., 91(1): 79–87. Doi: 10.1016/j.fishres.2007.11.009 |
| Prchalová M, Kubečka J, Říha M, Mrkvička T, Vašek M, Jůza T, Kratochvíl M, Peterka J, Draštík V, Křížek J, 2009. Size selectivity of standardized multimesh gillnets in sampling coarse European species. Fish. Res., 96(1): 51–57. Doi: 10.1016/j.fishres.2008.09.017 |
| Prchalová M, Mrkvička T, Kubečka J, Peterka J, Čech M, Muška M, Kratochvíl M, Vašek M, 2010. Fish activity as determined by gillnet catch:a comparison of two reservoirs of different turbidity. Fish. Res., 102(3): 291–296. Doi: 10.1016/j.fishres.2009.12.011 |
| Prchalová M, Mrkvička T, Peterka J, Čech M, Berec L, Kubečka J, 2011. A model of gillnet catch in relation to the catchable biomass, saturation, soak time and sampling period. Fish. Res., 107(1-3): 201–209. Doi: 10.1016/j.fishres.2010.10.021 |
| Prchalová M, Neal J W, Muñoz-Hincapié M, Jůza T, Říha M, Peterka J, Kubečka J, 2012. Comparison of gill nets and fixed-frame trawls for sampling threadfin shad in tropical reservoirs. Trans. Am. Fish. Soc., 141(4): 1 151–1 160. Doi: 10.1080/00028487.2012.675922 |
| Prchalová M, Kubečka J, Říha M, Čech M, Jůza T, Ketelaars H A, Kratochvíl M, Mrkvička T, Peterkaa J, Vašeka M, Wagenvoort A J, 2013. Eel attacks-a new tool for assessing European eel (Anguilla anguilla) abundance and distribution patterns with gillnet sampling. Limnologica, 43(3): 194–202. Doi: 10.1016/j.limno.2012.09.003 |
| Quinn G P, Keough M J, 2002. Experimental Design and Data Analysis for Biologists. Cambridge University Press, Cambridge. |
| Rotherham D, Johnson D D, Kesby C L, Gray C A, 2012. Sampling estuarine fish and invertebrates with a beam trawl provides a different picture of populations and assemblages than multi-mesh gillnets. Fish. Res., 123-124: 49–55. Doi: 10.1016/j.fishres.2011.11.019 |
| Šmejkal M, Ricard D, Prchalová M, Říha M, Muška M, Blabolil P, Čech M, Vašek M, Jůza T, Monteoliva Herreras A, Encina L, Peterka J, Kubečka J, 2015. Biomass and abundance biases in European standard gillnet sampling. PLoS One, 10(3): e0122437. Doi: 10.1371/journal.pone.0122437 |
| Tang M, Boisclair D, 1995. Relationship between respiration rate of juvenile brook trout (Salvelinus fontinalis), water temperature, and swimming characteristics. Can. J. Fish.Aquat. Sci., 52(10): 2 138–2 145. Doi: 10.1139/f95-806 |
| Tremain D M, Adams D H, 1995. Seasonal variations in species diversity, abundance, and composition of fish communities in the northern Indian River Lagoon, Florida. Bull. Mar. Sci., 57(1): 171–192. |
| Van Den Avyle M J, Boxrucker J, Michaletz P, Vondracek B, Ploskey G R, 1995. Comparison of catch rate, length distribution, and precision of six gears used to sample reservoir shad populations. North Am. J. Fish. Manage., 15(4): 940–955. Doi: 10.1577/1548-8675(1995)015<0940:COCRLD>2.3.CO;2 |
| Young S S, Chiu T S, Shen S C, 1994. A revision of the family Engraulidae (Pisces) from Taiwan. Zool. Stud., 33(3): 217–227. |
| Zweig C L, Kitchens W M, 2008. Effects of landscape gradients on wetland vegetation communities:information for large-scale restoration. Wetlands, 28(4): 1 086–1 096. Doi: 10.1672/08-96.1 |
 2018, Vol. 36
2018, Vol. 36


