Institute of Oceanology, Chinese Academy of Sciences
Article Information
- WEI Jie(魏杰), ZHAO Wen(赵文), WANG Shan(王珊), WANG Meiru(王美儒), WANG Xiaoliang(王小亮), JI Shichen(季世琛), AN Hao(安浩)
- Effect of temperature, salinity, and body length on the energy budget of Daphniopsis tibetana Sars (Cladocera: Daphniidae)
- Chinese Journal of Oceanology and Limnology, 36(5): 1812-1824
- http://dx.doi.org/10.1007/s00343-018-7192-8
Article History
- Received Jul. 11, 2017
- accepted in principle Aug. 26, 2017
- accepted for publication Sep. 28, 2017
Daphniopsis tibetana Sars (1903) (Family Daphniidae) is a cladoceran that inhabits lowtemperature brackish water lakes. Daphniopsis tibetana is resistant to high salinity (2.7–35) and is mainly distributed in high altitude, cold (-2–8℃), oligotrophic waters of Tibet, Qinghai, and Xinjiang, China, with a pH of 8.7–10.4 (Shen and Song, 1964; Chiang and Du, 1979; Jiang, 1983; Zhao, 1991; Zhao et al., 1996, 2006, 2010, 2016). Daphniopsis tibetana has an advantage of resistance to low temperature, which can make up for the lack of Moina mongolica in the north by low temperature limit. Daphniopsis tibetana is being acclimated to live in brackish water (salinity, 25) for used as a live food for juvenile marine fish and shrimp.
Many studies have been conducted on shrimp and crab bioenergetics, such as the effects of salinity on the energy budgets of Penaeus chinensis, Litopenaeus vannamei, and Macrobrachium nipponense (Shi et al., 1994; Zhang and Dong, 2002; Wang et al., 2004; Tian et al., 2005). However, only a few studies are available on marine cladoceran bioenergetics, and those were aimed at feeding, assimilation, and metabolic indices (Smirnov, 2017). Systemic studies on energy budgets have been conducted by Richman (1958), Slobodkin (1959), Kryutchkova (1967), Schindler (1968), Lei and Armitage (1980), Lynch et al. (1986), and Yang and He (1997). The biological characteristics (Zhao et al., 2002), morphology (Zhao et al., 2005), habit (Manca et al., 1994), natural distribution (Jiang, 1983), karyotype (Zhao et al., 2004), oxygen consumption rate (Zhao et al., 2005b), and ammonia excretion rate (Zhao et al., 2008b) of D. tibetana have been investigated, but variations in the energy budget have not been reported. Studies on D. tibetana bioenergetics would determine the energy budget and provide an understanding of the effects and mechanisms of environmental factors on the energy budget, as well as improve culture methods, enhance food utilization rate, and promote the complete growth potential of D. tibetana. In this study, we investigated cladoceran bioenergetics and provide a scientific basis for D. tibetana mass culture by studying the effects of water temperature, salinity, and body length on the D. tibetana energy budget. Additionally, we deduced the energy budget equilibrium equation by determining the optimal environmental conditions for D. tibetana growth.
2 MATERIAL AND METHODDaphniopsis tibetana were collected from Namukaco (northern Tibet). They were acclimated and cultured in 20 seawater and fed Chlorella pyrenoidosa, Chaetoceros muelleri, Isochrysis zhanjiangensis, and Dunaliella salina. The four species of algae are mixed by specific volume ratio, 2 (I. zhanjiangensis): 1 (C. muelleri): 1 (C. pyrenoidosa): 1 (D. salina). Diluted mixed feed to salinity test required after feeding by aeration tap water, feed density is (8.5–9.5)×106 cells/mL. The water for the experiment was obtained offshore of Heishi Reef, Dalian. Debris and sediment in the water was precipitated, and the water was filtered with 300-mesh silk cloth (aperture, 37.5 μm). Salinity was adjusted just prior to use. Daphniopsis tibetana were maintained under a 12-h light:12-h dark photoperiod with a light intensity of 700–900 lx.
2.1 Effects of temperature, salinity, and body length on D. tibetana feeding rate 2.1.1 Effect of temperature on D. tibetana feeding rateThree temperatures (14±0.5, 20±0.5, and 22±0.5℃ were tested. Larvae (body length, 0.77±0.09 mm; body weight, 0.05±0.02 mg/ind.) cultured at 14℃ and 15 salinity were slowly adapted to the water at each trial temperature and starved for 24 h before the experiment. The larvae were collected from the culture beaker using a wide-mouthed burette, cleaned twice with filtered seawater for 10 s each to avoid polluting the original medium, and placed in a 15-mL experimental test tube. Each test tube in the experimental group had 10 mL of medium and 10 larva, which were fed C. muelleri at an initial food density of 4.7×105 ind. /mL. The tubes were placed in constant temperature lighting incubators set to 14, 20, and 22℃ (type 3744; Thermo Scientific, Rockford, IL, USA). We divided day and night into four intervals. Larvae were fed for 3 h during each interval, removed in 5 mL culture medium, and 5% formaldehyde was added for fixation. We collected exuviated shells and counted them. When the test larvae were cleaned with a wide-mouthed burette during intermission, they were moved temporarily to other similarly prepared containers. The culture media was replaced, and C. muelleri density was adjusted. After the continuous day and night observations, we summed up the four intervals of data, determined the proportions, and calculated the daily amount fed and the daily exuviate. The body lengths (L) of D. tibetana in all groups were measured independently before and after the experiment under a microscope. The control and experimental groups were tested in triplicate.
2.1.2 Effect of salinity on D. tibetana feeding rateFive salinities (5, 10, 15, 20, and 25) were tested. Larvae (body length, 0.79±0.08 mm; body weight, 0.05±0.02 mg/ind.) cultured at 14℃ and 15 were slowly adapted to the test salinities. The experiment was conducted in a constant 14℃ incubator as described above for the temperature trials. The control and experimental groups were tested in triplicate.
2.1.3 Effect of body length on D. tibetana feeding rateFive body lengths (0.83±0.04, 1.17±0.03, 1.49±0.04, 1.83±0.06, and 2.33±0.02 mm) were tested. The associated body weights were 0.06±0.01, 0.15±0.01, 0.29±0.02, 0.51±0.05, and 0.98±0.02 mg/ ind. Larvae cultured at 14℃ and 15 were used, and the experiment was conducted in a constant 14℃ incubator. The control and experimental groups were tested in triplicate.
2.2 Effects of temperature, salinity, and body length on D. tibetana oxygen consumptionAccording to Zhao et al. (2005b):
The regression equation for temperature and specific oxygen (SO) consumption rate is: SO=0.0678e0.1605T (μg/(mg·h)).
The regression equation for salinity and the SO consumption rate is: SO=0.115S0.5612 (μg/(mg·h)).
The regression equation for body length and the SO consumption rate is: SO=-0.2059L+0.7908 (μg/ (mg·h)).
The D. tibetana SO consumption rates under the trial temperatures, salinities, and body lengths were calculated separately according to the regression equations.
2.3 Effects of temperature, salinity, and body length on D. tibetana ammonia excretionAccording to Zhao et al. (2008b):
The regression equation for temperature and the ammonia-N excretion rate (NR) is:
NR=0.0359ln(T)−0.0185 (mg/(g·h)).
The regression equation for salinity and the NR is:
NR=0.0535e0.0213S (mg/(g·h)).
The regression equation for body length and the NR is:
NR=0.0568L-1.4154 (mg/(g·h)).
The D. tibetana NR under the trial temperatures, salinities, and body lengths were calculated separately according to the regression equations.
2.4 Energy measurements and calculation of the energy budgetEnergy values of D. tibetana and C. muelleri
Phytoplankton and zooplankton energy is typically measured directly by micro-bomb calorimetery or dichromate oxidation (wet combustion process). It can also be calculated indirectly using an elemental analyzer after analyzing phytoplankton and zooplankton biochemical compositions. We did not measure the energy of D. tibetana and C. muelleri owing to limitations of the experimental conditions, which were estimated according to the model and data from references.
Chaetoceros muelleri energy was measured by calculating the average diameter of 30 random C. muelleri cells under a microscope (mean, 2.9 μm) and calculating cell volume (57.38 μm3, equivalent to biomass of 57.38×10-9 mg/ind.), according to the following equations:
lgCa1=0.76lgV−0.29 (Mitchell and Williams, 1982)
or
lgCa2=0.866lgV−0.460 (Strathmann, 1967),
where Ca1 is carbon content (×10-9 mg C/ind.) of each cell according to the model of Mullin et al. (1966), Ca2 is carbon content (×10-9 mg C/ind.) of each cell according the model of Strathmann (1967), and V is cell volume (57.38 μm3). The Ca1 and Ca2 values were calculated to be:Ca=1/2(Ca1+Ca2)=11.35×10-9 mgC/ind. as C. muelleri carbon content. If lgC=104 cal is used, the energy value is 11.35×10-8 cal/ind., which is the same as 4.75×10-7 J/ind.
Daphniopsis tibetana energy was determined using information from Zhang and He (1991). The energy of cladoceran dry weight per unit is 4.6 kcal/g (Zhang and He, 1991), which equals 19.24 kJ/g. Using 19.24 kJ/g as the mean energy value for dry weight per unit, the energy of D. tibetana wet weight per unit was 0.962 J/mg, based on the D. tibetana dry-wet weight ratio of 0.05.
2.5 Food energy (C)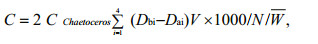
where C is daily larval feed energy (J/(g·d)), CChaetoceros is C. muelleri (J/ind.) energy, Dbi and Dai are C. muelleri densities of the control and experimental groups after each interval of the feeding test (×104 ind./mL), V is the experimental medium volume (mL), N is the number of larvae in the experimental test tube, and W is individual average wet weight.
2.6 Growth energy (P)(1) Energy for growth (Pg)

where Pg is the energy of D. tibetana wet weight per unit for daily growth (J/(g·d)), CDaphniopsis is the energy of D. tibetana (J/mg) wet weight per unit, Vw is the growth rate of an individual based on wet weight (mg/d), and W is average individual wet weight (mg). Vw was calculated using a regression equation for D. tibetana body length (L) and body weight (W): W=0.0993L2.7024 (Zhao et al., 2008b).
(2) Energy for exuviation (Pex)
Daphniopsis tibetana exuviate during growth. Because this is an important component of cladoceran growth energy, it should be considered when estimating the energy budget. The loss of energy from exuviation could not be measured owing to limitations of the experimental conditions. Lynch et al. (1986) reported a relationship between the carbon value of an exuviated Daphnia pulex shell and the carbon value of an individual as: M=0.016B1.472. Wissing and Hasler (1968) found that chitin shell energy of Daphnia spp. accounts for 15% of total body energy.
We adopted the Daphnia estimate of Wissing and Hasler (1968) based on the similar shape and size of Daphnia and Daphniopsis. Exuviation energy was calculated as:
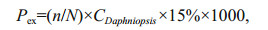
where Pex is the energy lost owing to daily D. tibetana exuviation (J/(g·d)), CDaphniopsis is the energy of D. tibetana (J/mg) wet weight per unit, n is the daily number of exuviated shells in the experimental group, and N is the number of larvae in the test tube.
2.7 Respiration (R) and excretion energy (U)Ammonia is the main product excreted by crustaceans, accounting for 40%–90% of nitrogencontaining excreta, which is continuously discharged into the water (Chen and Nan, 1995). Thus, we used the amount of NR excreted by D. tibetana and did not include carbamide because of its minimal contribution.
R=SO×14.24×24 and
U=NR×24.83×24,
where R is metabolic energy (J/(g·d)), U is excreted energy (J/(g·d)), SO is the larval specific oxygen consumption rate (μg/(mg·h)), NR is the ammonia-N excretion rate (mg/(g·h)), 14.24 is the amount of energy (J/mg) lost by larva consuming 1 mg O2 (Bayne et al., 1987), and 24.83 is the amount of energy (J/mg) lost by larva excreting 1 mg NH3-N (Cui, 1989).
2.8 D. tibetana energy budget equationsAccording to the formula proposed by Richman (1958) and Petrusewicz and Macfadyen (1970), the cladoceran energy budget equation is: C=P+R+U+F, and the assimilation energy equation is A=P+R. Thus, it was deduced that fecal energy=C–P–R–U, assimilation efficiency (AE)=A/C=(P+R)/C×100%, gross growth efficiency (K1)=(P/C)×100%, and net growth efficiency (K2)=(P/A)×100%.
The data were subjected to analysis of variance (ANOVA) using EXCEL 2003 (Microsoft Inc., Redmond, WA, USA) and SPSS 16.0 (SPSS Inc., Chicago, IL, USA) statistical software. Duncan's multiple comparison test was used to detect differences at P < 0.05.
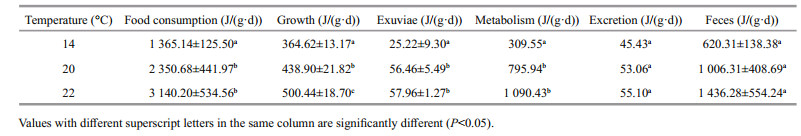
3 RESULT 3.1 Effect of temperature on the D. tibetana energy budgetThe D. tibetana energy budget values altered by temperature (Table 1). The increase of temperature seems to induce the food consumption, growth, exuviae, metabolism and excretion, although for excretion and feces significant differences were observed between treatments.
However, the trend was different at different temperatures (Table 2). The percentage of energy used for growth and excretion within food consumption was negatively associated with water temperature and was significantly higher at 14℃ than at 20 or 22℃ (P < 0.05). The percentage of energy used for metabolism was positively associated with water temperature and was clearly higher at 20 and 22℃ than that at 14℃ (P < 0.05). The percentage of energy used for exuviation and feces was not different among the trial water temperatures. Based on the D. tibetana energy expenditure at 14℃, the order was feces > metabolism > excretion > exuviation, whereas the order was feces > metabolism > exuviation > excretion at 20 and 22℃. Temperature had a significant effect on the proportion of energy used for D. tibetana excretion (P < 0.01).

|
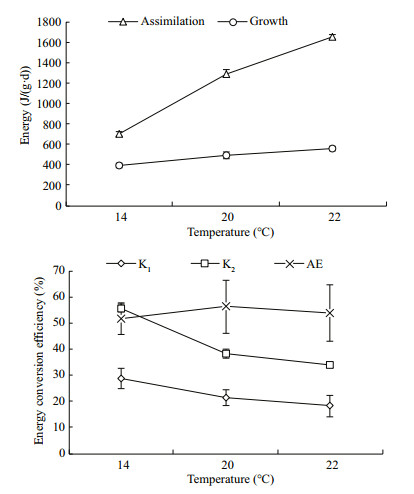
Assimilation (A) and growth (P), as well as energy conversion efficiency at the different water temperatures are shown in Fig. 1. ANOVA showed that temperature had a significant influence on assimilation and growth, as well as net growth efficiency (P < 0.01). Assimilation and growth increased noticeably with an increase in water temperature (P < 0.05), whereas AE (51.39%–56.22%) was not different among the different temperatures. Gross growth efficiency and net growth efficiency were 28.78% and 55.71%, respectively, and were significantly higher at 14℃ than those at the other temperatures (P < 0.05).
|
| Figure 1 Assimilation, growth and energy efficiency (K1, K2, and AE) of Daphniopsis tibetana at different temperatures |
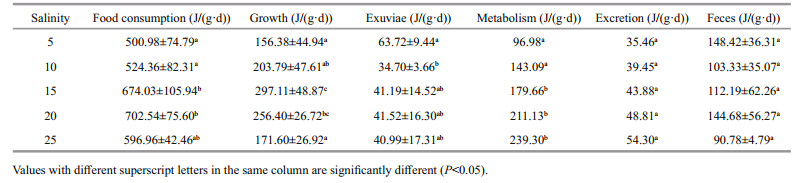
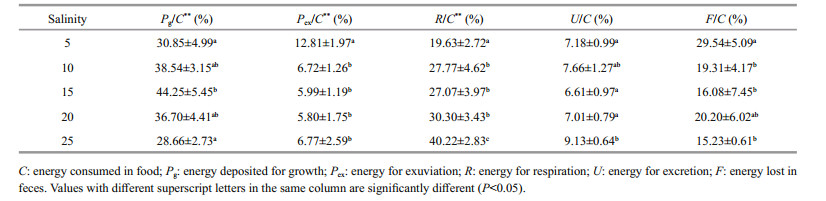
The D. tibetana energy budget based on the trial salinities is shown in Table 3. The D. tibetana energy budget values altered by salinity. The increase of salinity seems to reduce the food consumption, growth, metabolism and excretion, and reduce the the feces exuviae and reduce. The regression equation for growth and salinity was Pg=-1.138 4S2+ 35.812S−7.072 7 (n=5, R=0.931 0), and the regression equation for metabolism and salinity was R=7.0536S+68.228 (n=5, R=0.995 0). The energy used for food consumption and growth rose initially and then decreased with increasing salinity. Food consumption and growth energy used by the 15 and 20 groups was notably higher than that in the other groups. Exuviation energy was significantly different, with the maximum observed in the 5 group and the minimum in the 10 group (P < 0.05). Metabolic energy use was positively associated with salinity, and energy used at 15, 20, and 25 was significantly higher than that used at 5 and 10. The excretion and feces energy values were not different among the test salinities.
The D. tibetana energy budget within the trial salinities is shown in Table 4. ANOVA demonstrated that the percentage of energy used for growth (Pg), exuviation, and metabolism was significantly affected by salinity (P < 0.01). The regression equation for growth and salinity was Pg/C (%)=-0.1278S2+ 3.708 7S+15.306 (n=5, R=0.972 3). The regression equation for exuviation and salinity was Pex/C (%)= 0.041 8S2−1.514 5S+18.83 (n=5, R=0.967 8). The regression relation for metabolism and salinity was R/C (%)=0.021 6S2+0.227 6S+19.63 (n=5, R=0.941 6). The proportion of energy for growth rose initially and then decreased with increasing salinity. The maximum value occurred at 15, and the proportion of energy used for exuviation was higher at 5 than that at the other salinities (P < 0.05). The proportion of energy used for metabolism increased significantly with increasing salinity (P < 0.05), whereas that for excretion and feces was not different among the trial salinities. Based on the D. tibetana energy expenditure at 5, the order was feces > metabolism > exuviation > excretion, whereas the order was metabolism > feces > excretion > exuviation at salinities of 10–25.
|
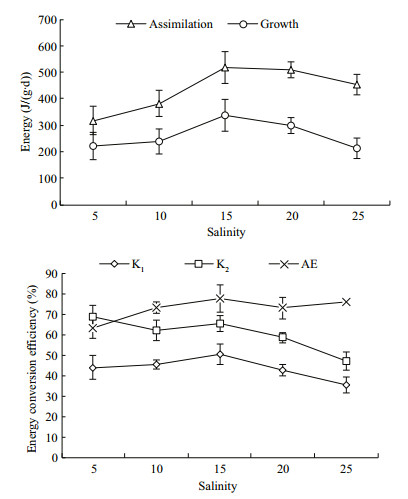
Assimilation (A), growth (P), and energy conversion efficiency at the different salinities is shown in Fig. 2. ANOVA showed that assimilation and net growth efficiency were significantly affected by salinity (P < 0.01). The regression equation for assimilation energy and salinity was A=-1.110 3S2+ 41.249S+122.09 (n=5, R=0.950 3). The regression equation for net growth efficiency and salinity was K2=-0.055 1S2+0.699 6S+64.902 (n=5, R=0.946 3). Energy for both assimilation and growth rose initially and then decreased with increasing salinity. The highest values were observed at 15 and were significantly higher than values at 5 and 10 (P < 0.05). No difference was detected at 20. Minimum assimilation energy was found at 5 but no differences were detected with the other groups. Minimum gross growth efficiency and net growth efficiency occurred at 25 and was not different from the other groups. In summary, D. tibetana energy conversion efficiency (AE, K1, and K2) was higher at 10–20, with values of 72.80%–77.31%, 42.50%–50.42%, and 58.43%– 65.02%, respectively.
|
| Figure 2 Assimilation, growth and energy efficiency (K1, K2, and AE) of Daphniopsis tibetana at different salinities |
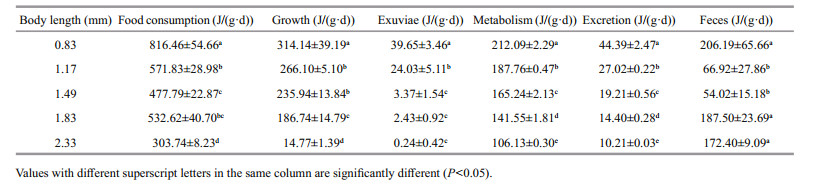
The D. tibetana energy budget within the trial body lengths is shown in Table 5. The D. tibetana energy budget values altered by body lengths. The increase of body length seems to reduce the food consumption, growth, exuviae, metabolism and excretion, although for the feces decrease first and then rise. The regression equations were: C=-434.18ln(L)+698.5 (n=5, R=0.935 8), Pg=-107.67L2+150.94L+253.73 (n=5, R=0.993 2), Pex=867.21e-3.436 3L (n=5, R=0.980 8), R= -70.529L+270.46 (n=5, R=1), and U=33.94L-1.421 8 (n=5, R=1). The proportion of energy used for food consumption, growth, exuviation, metabolism, and excretion decreased significantly with increasing body length (P < 0.05). The energy lost to feces for a body length of 1.17–1.49 mm was significantly higher than that of the other groups (P < 0.05).
The D. tibetana energy budget for the trial body lengths is shown in Table 6. ANOVA demonstrated that the energy proportion allocated to D. tibetana food consumption was significantly affected by body length (P < 0.01). The regression equations were: Pg/C (%)=-42.434L2+111.37L−24.704 (n=5, R=0.995 9), Pex/C (%)=71.709e-2.881L (n=5, R=0.976 6), R/C (%)= 37.712L3−180.37L2+272.4L−97.735 (n=5, R=0.940 3), U/C (%)=1.525 9L2−6.450 5L+9.942 3 (n=5, R=0.944 8), and F/C (%)=40.204L2−102.48L+79.75 (n=5, R=0.957 9). The proportion of growth energy for food consumption rose initially and then decreased with increasing body length. The group with a body length of 1.17–1.19 mm had a significantly higher proportion of growth energy for food consumption than that of the other groups (P < 0.05). The proportion of energy for exuviation decreased with increasing body length, and the group with a body length 0.83– 1.17 mm had significantly higher energy for exuviation than that of the other groups (P < 0.05). The proportion of energy used for metabolism tended to increase initially, then decreased, and rose again with increasing body length. The groups with body lengths of 1.17–1.19 mm and 2.33 mm used significantly more energy for metabolism than that of the other groups (P < 0.05). The proportion of energy lost to excretion decreased initially and then rose with increasing body length. The minimum energy used for excretion occurred when body length was 1.83 mm and was significantly different from that of the other groups (P < 0.05). The proportion of energy lost to feces also decreased initially and then rose with increasing body length. Energy used for feces in the 1.17–1.19 mm body length group was significantly lower than that in the other groups (P < 0.05). The D. tibetana energy expenditure budget showed an order of metabolism > feces > excretion > exuviation when body length was 0.83–1.49 mm, whereas the order was feces > metabolism > excretion > exuviation when body length was 1.83–2.33 mm.
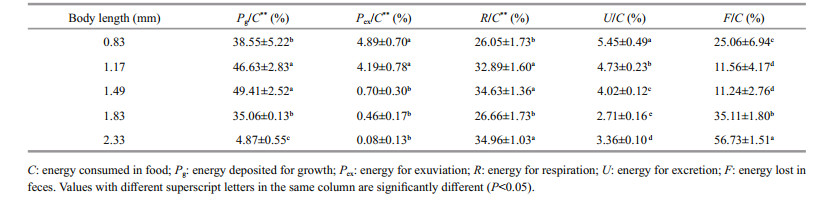
|
Assimilation and growth (P) energy as well as energy conversion efficiency of D. tibetana by body length are shown in Fig. 3. ANOVA demonstrated that assimilation and growth (P) energy as well as energy conversion efficiency of D. tibetana were significantly affected by body length (P < 0.01). The regression equations were: A=-287.31L+819.61 (n=5, R=0.988 4), P=-216.78L+549.15 (n=5, R=0.979 9), AE=-41.725L2+108.91L+10.32 (n=5, R=0.961 0), K1=-39.753L2+99.325L−11.203 (n=5, R=0.997 7), and K2=-40.662L2+98.677L+5.555 2 (n=5, R=0.972 8. The proportions of energy used for assimilation and growth were significantly negatively related to body length (P < 0.01). Energy conversion efficiency increased initially and then decreased with increasing body length. Assimilation and gross growth efficiency of the group with a body length of 1.17–1.19 mm were significantly higher than those in the other groups (P < 0.05). Net growth efficiency of the group with a body length of 2.33 mm was significantly lower than that of the other groups (P < 0.05). No differences were observed in the group with a body length of 0.83–1.83 mm. In summary, D. tibetana energy conversion efficiency (AE, K1, and K2) was highest at a body length of 1.17–1.49 mm, with values of 83.70%–84.73%, 50.11%–50.81%, and 59.13%– 60.71%, respectively.

|
| Figure 3 Assimilation, growth, and energy efficiency (K1, K2, and AE) of Daphniopsis tibetana at different body lengths |
Temperature is one of the most important environmental factors influencing growth of an organism. A change in temperature can influence metabolism directly or indirectly and, thus, affect growth and other life activities. Many studies have been conducted on the effects of temperature on physiological activities of marine invertebrates. Our results show that the energy used for food consumption and metabolism increased with increasing temperature, as the proportion used for growth decreased gradually. This phenomenon is common in clams and other shellfish when temperature rises above the optimum. Clams reduce filtration rate and increase respiratory rate sharply with an increase in temperature, which leads to less energy for growth (Newell, 1979; Newell and Branch, 1980; Dong et al., 2003).
Environmental temperature has a profound effect on respiratory metabolism of an organism. Growth is accelerated and oxygen consumption rate increases with rising temperature below the optimum temperature. The oxygen consumption rate at temperatures higher than optimal does not increase during growth and development and may even decrease (Dong and Xue, 2000). The optimal temperature for D. tibetana is 13–16℃ (Zhao et al., 2005a, b). The energy used for D. tibetana metabolism increased with increasing temperature during the temperature experiments, and the values were 0.015 2–0.053 0 J/d. The oxygen consumption rates of M. mongolica increases significantly within the same body length when temperature increases between 20 and 30℃ (Wang, 1999). The oxygen consumption rate of Calanus sinicus follows the same rule as indicated above (Zhang et al., 2000). This may be a result of increased metabolic activity when water temperature increases, which would increase oxygen consumption. Most fish, shrimp, and crabs have similar oxygen consumption rates (Dong et al., 1994a).
Wang and He (1997) reported that M. mongolica metabolism increases rapidly with increasing temperature. Cladocerans must increase their food consumption to maintain a high metabolic level to prevent losing energy owing to temperature changes. Metabolic rate rise with increasing water temperature within a suitable temperature; Daphniopsis required more energy for survival. As enzyme activity is enhanced, the rate of digestion accelerates at the same time. However, food consumption rate decreases sharply when water temperature surpasses the upper maximum (Brett, 1979). In our study, energy utilization efficiency was significantly higher at 14℃ than that at 20 or 22℃. Food energy and assimilation energy were enhanced continuously with increasing temperature, indicating that 20–22℃ was not the maximum temperature limit for Daphniopsis. Daphniopsis uses more energy to adapt to an environmental change.
The proportion of energy used for growth decreased with increasing temperature. In the temperature trial, energy for growth and metabolism at 14℃ was not different from that at 20 or 22℃, and the P/R value at 14℃ was significantly higher than that at 20 and 22℃. This result indicates that energy used for growth increased with temperature within the suitable temperature range, but that more energy would be used for metabolism and excretion at a temperature outside of the preferred range. Growth efficiency decreased and the proportion of energy used for growth decreased. The proportion of growth energy of the total food consumption energy is called growth efficiency (P/R) in ecology or food conversion efficiency. Food conversion efficiency is an important index to determine energy utilization efficiency and has a close relationship with the economics crustacean aquaculture.
4.2 Effect of salinity on the D. tibetana energy budgetChanges in salinity have clear effects on marine animal metabolism. Daphniopsis tibetana is euryhaline and can survive in hypertonic and hypotonic environments. The optimal salinity for osmoregulation is typically close to the isotonic point of body fluid (15–20). Our results show that the optimal salinity for D. tibetana growth and metabolism was 20, which is within the optimal salinity range for osmoregulation. The D. tibetana oxygen consumption rate increased gradually as salinity was changed from 5 to 25. The hypertonic regulation that occurred at 5–15 was a sign of increased metabolism, whereas hypotonic regulation from 15 to 25 was a sign of decreased metabolism, and indicates the need for enhanced metabolism to maintain homeostasis in vivo. Death ensued if salinity continued to rise owing to an inability of D. tibetana to maintain osmoregulation with a sharp decrease in the oxygen consumption rate.
The energy used for metabolism in D. tibetana maintained at 5–10 was significantly different from that of the groups held at 15–25, and energy conversion efficiency and assimilation energy of the group held at 20 were both higher than those of the other groups. The oxygen consumption rates of cladocerans are related to food density and dissolved oxygen (Heisey and Porter, 1977; Porter et al., 1982). Many crustaceans live in isotonic environments, where osmotic pressure is low, and any extra metabolic energy expenditure is small owing to the low salinity gradient. Thus, less energy is needed to maintain homeostasis in vivo. Therefore, basic metabolism is low, and energy conversion efficiency is highest at this time (Dalla Via, 1986, 1987; Chen and Lai, 1993; Shi et al., 1994; Chen and Nan, 1995; Pillai and Diwan, 2002). Kutty et al. (1974) showed that longterm acclimation to salinity by Fenneropenaeus indicus eliminates the differences in metabolism rates at different salinities. That study showed that feeding, assimilation, and absorption are closely related. In our study, the energy used for food consumption reached its lowest value at 5, yet D. tibetana survived by changing assimilation and absorption rates and reducing basic metabolic energy needs.
Salinity is another factor that affects growth and exuviation in Cladocera. Some studies have reported no effect of salinity on exuviation (Dall et al., 1990), whereas others have found that reducing salinity promotes prawn exuviation and that unsuitably high salinity limits exuviation (Hu and Lu, 1990; Wang, 1991; Allan and Maguire, 1992; Chen and Bian, 1994). The number of larvae surviving to exuviate and their body weights affect the energy used for exuviation. Our results show that salinity had no significant effect on D. tibetana exuviation. The highest exuviation energy was observed at 20. The energy used for growth was significantly higher at 15–20 but significantly lower at 25 than those in the other groups because optimal salinity for D. tibetana was 15–20. Thus, larva increased respiratory metabolism to adapt beyond their optimum salinity, and energy used for growth decreased correspondingly.
Salinity had no significant effect on D. tibetana energy used for food consumption. The maximum amount of energy used for food consumption by D. tibetana occurred at 15–20. Respiration and feeding in cladocerans are both accomplished by the thoracic limb. Feeding intensity increased with age. Food consumption rate is mainly related to dissolved oxygen status; thereby, food consumption increased correspondingly when D. tibetana was maintained at a suitable salinity with good dissolved oxygen. More data are available on the effect of salinity on feeding in fish than on the effect of salinity on D. tibetana feeding intensity. Marine fish metabolism is usually lower in low salinity water, whereas enzyme activities and appetite increase in fish with increasing salinity. Fish become stressed, their appetite decreases, and death may ensue if salinity is too high (Zhou et al., 2006). Our observations indicate that D. tibetana has similar responses.
The D. tibetana NR increased with increasing salinity, but no significant effect of salinity on D. tibetana energy lost to excretion was observed. Maximum energy was lost to excretion at 20, indicating that metabolic activity was strongest at this time.
4.3 Effect of body length on the D. tibetana energy budgetThe regression equation of body length and body weight (W=0.099 3L2.702 4) indicated that they had a similar trend. Our results show that the energy used for growth, food consumption, and metabolism increased with increased body length. Energy utilization efficiency of the group with a body length of 1.83 mm was significantly higher than that of the other groups, whereas energy utilization efficiency of the group with a body length of 2.33 mm was significantly lower than that of the other groups. The regression equation showed that energy is mainly used for growth at the larval stage, whereas energy was mainly used for reproduction in adults. Thus, less energy was used for growth, body length increased slowly, and food consumption and metabolic energy were higher owing to the larger adult body. The energy expenditure for metabolism is usually the highest (Brown et al., 1989; Dong et al., 1994b; Zhang et al., 1998) or only less than energy used for growth (Villarreal, 1991; Rosas et al., 1993) in most cladocerans; thus, changes in growth and metabolism have important effects on the energy budget. Body weight is one of the most important factors affecting oxygen consumption. The oxygen consumption rates of adult shrimp, Macrobrachium rosenbergii (Lin et al., 1999), juvenile Macrobrachium acanthurus (Gasca-Leyva et al., 1991), juvenile Macropetasma africanus (Cockcroft and Wooldridge, 1985), juvenile marine shrimp, Palaemon pacificus, and adult P. chinensis increase with increasing body weight, whereas SO consumption rates decrease with increases in body weight. The D. tibetana SO consumption rate increased with increased body length, which was similar to that of other aquatic animals. Zhao et al. (2005b) reported that the reason of SO consumption rate decreased with individual increased had connect with proportion of all kinds of tissue and organs. The oxygen consumption rates per unit weight of the liver, gills, and digestive organs are higher, as they are directly related to life processes, whereas oxygen consumption rates of other tissues, such as muscle and fat are lower. The mass percentage oxygen consumed by the organs directly related to life processes is higher in growing animals, which leads to small individuals consuming more than that of larger individuals. Body length and weight are the main factors affecting NR. NR increases as body length increases under the same temperature and salinity conditions. Because the metabolic energy expenditure of an individual is higher, energy lost to excretion and feces increases correspondingly. In our study, D. tibetana food consumption rate increased gradually with increasing body length and was significantly different at the different body lengths. The proportion of energy used for growth and growth efficiency in large yellow croaker decrease gradually as body weight increases, whereas food consumption rate increases (Zhou et al., 2008). This result was similar to D. tibetana growth and metabolism based on the regression equations and energy metabolism.
4.4 Application prospects for D. tibetanaA series of cladoceran studies have been conducted to provide new live food for cultured marine animals. Studies on freshwater Cladocera indicate that their upper salinity limit is 14, and that they are difficult to adapt to seawater in the short term. An observational study by Zhao et al. (2008b) showed that the marine cladoceran M. mongolica is adaptable to seawater and can be used as food in juvenile marine fish production. It not only has an advantage of the proper size, but reproduces and grows rapidly at 25–28℃ in 5–40 with a high nutrition value, and is suitable for high intensity culture. Compared with M. mongolica, D. tibetana is a brackish water cladoceran that is dominant in most brackish lakes of Tibet and Xinjiang with considerable biomass. Our experimentally adapted D. tibetana population had its highest energy conversion efficiency at 14℃ and 20, which agrees with the optimal temperature of 13–16℃ and salinity of 15–20 reported by Zhao et al. (2008a). Therefore, D. tibetana can acclimate to the water temperature conditions during northern marine fish and shrimp breeding, and overcome culture application limitations of M. mongolica need to warming in the northern low temperature period. The isoleucine, threonine, methionine, and histidine contents of D. tibetana were all significantly higher than those of M. mongolica and Tigriopus japonicus, which are food organisms. Methionine content in D. tibetana reached 3.64%, and the ratio of unsaturated fatty acids to total fatty acids was 71.58% (Zhao et al., 2006). Thus, D. tibetana would completely satisfy the nutritional needs for fish and shrimp growth. Therefore, D. tibetana has great theoretical and practical value to become a promising new live feed for marine aquaculture seed production.
5 CONCLUSIONThe results of energy budget of D. tibetana showed that assimilation efficiency (AE) with values of 51.59%–56.22% at the trial temperatures. Gross growth efficiency (K1) and net growth efficiency (K2) were 28.78% and 55.71%, respectively, at 14℃. Daphniopsis tibetana energy conversion efficiency (AE, K1, and K2) was higher at salinities of 10–20, with values of 72.80%–77.31%, 42.50%–50.42%, and 58.43%–65.02%, respectively. Daphniopsis tibetana energy conversion efficiency was highest when body length was 1.17–1.19 mm, with values of 83.70%–84.73%, 50.11%–50.81%, and 59.13%– 60.71%, respectively.
6 DATA AVAILABILITY STATEMENTWe declare that the data supporting the findings of this study are available within the article.
Allan G L, Maguire G B. 1992. Effects of pH and salinity on survival, growth and osmoregulation in Penaeus monodon Fabricius. Aquaculture, 107(1): 33-47.
DOI:10.1016/0044-8486(92)90048-P |
Bayne B l, Hawkins A J S, Navarro E. 1987. Feeding and digestion by the mussel Mytilus edulis L. (Bivalvia: Mollusca) in mixtures of silt and algal cells at low concentrations. Journal of Experimental Marine Biology and Ecology, 111(1): 1-22.
DOI:10.1016/0022-0981(87)90017-7 |
Brett J R. 1979. Environmental factors and growth. In: Fish Physiology. Academic Press, New York. p.599-675.
|
Brown P B, Robinson E H, Clark A E, Lawrence A L, Bordner C E. 1989. Apparent digestible energy coefficients and associative effects in practical diets for red swamp crayfish. Journal of the World Aquaculture Society, 20(3): 122-126.
DOI:10.1111/jwas.1989.20.issue-3 |
Chen J C, Lai S H. 1993. Effects of temperature and salinity on oxygen consumption and ammonia-N excretion of juvenile Penaeus japonicus Bate. Journal of Experimental Marine Biology and Ecology, 165(2): 161-170.
DOI:10.1016/0022-0981(93)90103-U |
Chen J C, Nan F H. 1995. Oxygen consumption and ammonia-N excretion of Penaeus chinensis (Osbeck, 1765) juveniles at different salinity levels (Decapoda, Penaeidae). Crustaceana, 68(6): 712-719.
DOI:10.1163/156854095X00223 |
Chen J, Bian P J. 1994. An influence of the low salinity upon tiger shrimp Penaeus japonicas. Journal of Zhejiang College of Fisheries, 13(4): 289-292.
(in Chinese with English abstract) |
Chiang S C, Du N S. 1979. Fauna Sinica: Crustacea (Freshwater Cladocera). Science Press, Beijing. 297p.
(in Chinese)
|
Cockcroft A C, Wooldridge T. 1985. The effects of mass, temperature and molting on the respiration of Macropetasma africanus Balss (Decapoda: Penaeoidea). Comparative Biochemistry and Physiology Part A: Physiology, 81(1): 143-148.
DOI:10.1016/0300-9629(85)90280-4 |
Cui Y B. 1989. Bioenergetics of fishes: theory and methods. Acta Hydrobiologica Sinica, 13(4): 369-383.
(in Chinese with English abstract) |
Dall W, Hill B J, Rothlisberg P C, Staples D J. 1990. The biology of the Penaeidae. Advances in Marine Biology, 27: 489.
|
Dalla Via G J. 1986. Salinity responses of the juvenile penaeid shrimp Penaeus japonicus: Ⅰ. Oxygen consumption and estimations of productivity. Aquaculture, 55(4): 297-306.
DOI:10.1016/0044-8486(86)90170-5 |
Dalla Via G J. 1987. Salinity responses in brackish water populations of the freshwater shrimp Palaemonetes antennarius: Ⅰ. Oxygen consumption. Comparative Biochemistry and Physiology Part A: Physiology, 87(2): 471-478.
DOI:10.1016/0300-9629(87)90153-8 |
Dong B, Li J, Wang H Y, Xue Q Z. 2003. Energy budget at different temperature and food concentration in manila clam Ruditapes philippinarum. Journal of Fishery Sciences of China, 10(5): 398-403.
(in Chinese with English abstract) |
Dong B, Xue Q Z, Li J. 2000. The effect of temperature on the filtration rate, clearance rate and absorption efficiency of manila clam, Ruditapes philippinarum. Marine Fisheries Research, 21(1): 37-42.
(in Chinese with English abstract) |
Dong S L, Du N S, Lai W. 1994a. Studies on the physioecology of Macrobrachium nipponense Ⅰ. Effects of temperature and body weight on metabolism. Oceanologia et Limnologia Sinica, 25(3): 233-237.
(in Chinese with English abstract) |
Dong S L, Du N S, Lai W. 1994b. Studies on the physioecology of Macrobrachium nipponense Ⅱ: Effects of temperature and body weight on energy budget. Oceanologia et Limnologia Sinica, 25(3): 238-242.
(in Chinese with English abstract) |
Gasca-Leyva J F E, Martinez-Palacios C A, Ross L G. 1991. The respiratory requirements of Macrobrachium acanthurus (Weigman) at different temperatures and salinities. Aquaculture, 93(2): 191-197.
DOI:10.1016/0044-8486(91)90217-U |
Heisey D, Porter K G. 1977. The effect of ambient oxygen concentration on filtering and respiration rates of Daphnia galeata mendotae and Daphnia magna. Limnology and Oceanography, 22(5): 839-845.
DOI:10.4319/lo.1977.22.5.0839 |
Hu Q X, Lu J S. 1990. Preliminary analysis of the relation of growth of Penaeus orientalis Kishinouye with environmental factors. Donghai Marine Science, 8(2): 59-62.
(in Chinese with English abstract) |
Jiang X Z. 1983. Aquatic Invertebrates in Tibet. Science Press, Beijing. p.443-492.
(in Chinese)
|
Kryutchkova N M. 1967. Utilisation of food for growth of Moina rectirostris Leydig. Zool. Zhurnal., 46(7): 1 030-1 036.
|
Kutty M N, Murugapoopathy G, Krishnan T S. 1974. Influence of salinity and temperature on the oxygen consumption in young juveniles of the Indian prawn Penaeus indicus. Marine Biology, 11: 125-131.
|
Lei C H, Armitage K B. 1980. Ecological energetics of a Daphnia ambigua population. Hydrobiologia, 70(1-2): 133-143.
DOI:10.1007/BF00015499 |
Lin X T, Liu L Y, Yu H D, Xu Z N. 1999. Metabolism of parent female Macrobrachium rosenbergii in relation to body size. Journal of Ji'nan University (Natural Science), 20(5): 107-111.
(in Chinese with English abstract) |
Lynch M, Weider L J, Lampert W. 1986. Measurement of the carbonbalancein Daphnia. Limnology and Oceanography, 31(1): 17-33.
DOI:10.4319/lo.1986.31.1.0017 |
Manca M, Cammarano P, Spagnuolo T. 1994. Notes on Cladocera and Copepoda from high altitude lakes in the Mount Everest Region (Nepal). Hydrobiologia, 287(3): 225-231.
DOI:10.1007/BF00006371 |
Mitchell B D, Williams W D. 1982. Population dynamics and Production of Daphnia carinata (King) and Simocephalus exspinosus (Koch) in Waste Stabilization Ponds. Australian Journal of Marine and Freshwater Research, 33(5): 837-864.
DOI:10.1071/MF9820837 |
Mullin M M, Sloan P R, Eppley R W. 1966. Relationship between carbon content, cell volume, and area in phytoplankton. Limnology and Oceanography, 11(2): 307-311.
DOI:10.4319/lo.1966.11.2.0307 |
Newell R C, Branch G M. 1980. The effect of temperature on the maintenance of metabolic energy balance in marine invertebrates. Advances in Marine Biology, 17: 329-396.
DOI:10.1016/S0065-2881(08)60304-1 |
Newell R C. 1979. Biology of Intertidal Animals. Marine Ecological Surveys: Faversham, Kent.
|
Petrusewicz K, Macfadyen A. 1970. Productivity of Terrestrial Animals: Principles and Methods. Blackwell, Oxford. 190p.
|
Pillai B R, Diwan A D. 2002. Effects of acute salinity stress on oxygen consumption and ammonia excretion rates of the marine shrimp Metapenaeus monoceros. Journal of Crustacean Biology, 22(1): 45-52.
DOI:10.1163/20021975-99990207 |
Porter K G, Gerritsen J, Orcutt J D Jr. 1982. The effect of food concentration on swimming patterns, feeding behavior, ingestion, assimilation, and respiration by Daphnia. Limnology and Oceanography, 27(5): 935-949.
DOI:10.4319/lo.1982.27.5.0935 |
Richman S. 1958. The transformation of energy by Daphnia pulex. Ecological Monographs, 28(3): 273-291.
DOI:10.2307/1942243 |
Rosas C, Vanegas C, Tabares I, Ramirez J. 1993. Energy balance of Callinectes Contreras 1930 in floating cages in a tropical coastal lagoon. Journal of the World Aquaculture Society, 24(1): 71-79.
DOI:10.1111/jwas.1993.24.issue-1 |
Schindler D W. 1968. Feeding, assimilation and respiration rates of Daphnia magna under various environmental conditions and their relation to production estimates. Journal of Animal Ecology, 37(2): 369-385.
DOI:10.2307/2954 |
Shen J R, Song D X. 1964. Preliminary study of Cladocera in Tibet. Zoo Sinica, 16(1): 61-69.
(in Chinese with English abstract) |
Shi Z F, Mei Z P, Luo Q Z, Zhang Y J, Song W H, Shen Y X, Gong R S. 1994. Preliminary studies on energy budget and utilization efficiency of Macrobrachium nipponensis. Journal of Fisheries of China, 18(3): 191-197.
(in Chinese with English abstract) |
Slobodkin L B. 1959. Energetics in Daphnia pulex populations. Ecology, 40(2): 232-243.
DOI:10.2307/1930033 |
Smirnov N N. 2017. Physiology of the Cladocera. Academic Press, Pennsylvania.
|
Strathmann R R. 1967. Estimating the organic carbon content of phytoplankton from cell volume or plasma volume. Limnology and Oceanography, 12(3): 411-418.
DOI:10.4319/lo.1967.12.3.0411 |
Tian X L, Dong S L, Wu L X, Wang F. 2005. A comparative study on the growth and energy budget of Chinese shrimp (Fenneropanaeus chinensis Osbeck) at constant and diel fluctuating temperature. Acta Ecologica Sinica, 25(11): 2 811-2 817.
(in Chinese with English abstract) |
Villarreal H. 1991. A partial energy budget for the Australian crayfish Cherax tenuimanus. Journal of the World Aquaculture Society, 22(4): 252-259.
DOI:10.1111/jwas.1991.22.issue-4 |
Wang J Q, Luo M, Zhang D Z, Pang C. 2004. Effect of water temperature and salinity on energy budget of Penaeus vannamei juveniles. Journal of Fisheries of China, 28(2): 161-166.
(in Chinese with English abstract) |
Wang L C, Liu XY. 1991. Shrimp Cultured. Nankai University Press, Tianjin. p.28-31.
|
Wang Y, He Z H. 1997. The effect of temperature and body length on feeding intensity of Moina mongolica Daddy. Journal of Dalian Fisheries University, 12(1): 1-7.
(in Chinese with English abstract) |
Wang Y. 1999. Temperature effect on oxygen consumption rates in Moina mongolica Daddy. Journal of Shantou University (Natural Science), 14(1): 59-63.
(in Chinese with English abstract) |
Wissing T E, Hasler A D. 1968. Calorific values of some invertebrates in Lake Mendota, Wisconsin. Journal of the Fisheries Research Board of Canada, 25(11): 2 515-2 518.
DOI:10.1139/f68-220 |
Yang H Y, He Z H. 1997. The effect of salinity on the assimilation, metabolism, growth and carbon budget of Daphnia magna. Journal of Fishery Sciences of China, 4(3): 33-38.
(in Chinese with English abstract) |
Zhang J M, He Z H. 1991. Handbook of Fishes and Natural Resources Investigation in Inland Waters. Agricultural Press, Beijing. p.12-75.
(in Chinese)
|
Zhang S, Dong S L. 2002. The effects of food and salinity on energy budget of juvenile shrimp of Penaeus chinensis. Journal of Dalian Fisheries University, 17(3): 227-233.
(in Chinese with English abstract) |
Zhang S, Wang F, Dong S L. 1998. Effects of temperature on energy partitioning and elemental composition (CNH) in Penaeus chinensis. Journal of Fishery Sciences of China, 5(1): 38-42.
(in Chinese with English abstract) |
Zhang W C, Wang R, Wang K. 2000. Effect of temperature on metabolic rates of Calanus sinicus. Marine Sciences, 24(2): 42-44.
(in Chinese with English abstract) |
Zhao W, He Z H, Yin S R. 2008a. Biology and Technology of Culture and Utilization in Marine Water for Cladocera in Inland Saline Waters. Science Press, Beijing. 163p.
(in Chinese with English abstract)
|
Zhao W, Huo Y Z, Gao J. 2006. Analysis and appraisement of nutrient compositions for Daphniopsis tibetana Sars. Journal of Fishery Sciences of China, 13(3): 446-451.
(in Chinese with English abstract) |
Zhao W, Jiang H, He Z H. 1996. Planktonic Crustaceans of inland saline waters in Sanbei District, Northern China. Journal of Dalian Fisheries University, 11(1): 1-13.
(in Chinese with English abstract) |
Zhao W, Liang X, Xie X, Zhang L, Wei J. 2008b. The effect of temperature, salinity and body length on ammonia excretion rate of Daphniopsis tibetana Sars. Acta Ecologica Sinica, 28(12): 6 385-6 389.
(in Chinese with English abstract) |
Zhao W, Wang Q H, Zheng M P, Zhao Y Y, Wang H L. 2002. A preliminary study on the biology of Daphniopsis tebitana Sar. Journal of Dalian Fisheries University, 17(3): 209-214.
(in Chinese with English abstract) |
Zhao W, Wang Q H. 2005. The morphological redescription of Daphniopsis tibetana Sar (Crustacea: Cladocera: Daphnidae). Journal of Dalian Fisheries University, 20(3): 165-173.
(in Chinese with English abstract) |
Zhao W, Zhang L, Huo Y Z. 2005b. The effect of salinity, temperature and body length on oxygen consumption of Daphniopsis tibetana Sars. Acta Ecologica Sinica, 25(7): 1 549-1 553.
(in Chinese with English abstract) |
Zhao W, Zhang P, Huo Y Z, Wang H L. 2004. Study of karyotype on Daphniopsis tibetana Sars (Cladocera: Daphniidae). Journal of Dalian Fisheries University, 19(3): 167-170.
(in Chinese with English abstract) |
Zhao W, Zhao Y Y, Wang Q H, Zheng M P, Wei J, Wang S. 2016. The community structure and seasonal dynamics of plankton in Bange Lake, northern Tibet, China. Chinese Journal of Oceanology and Limnology, 34(6): 1 143-1 157.
DOI:10.1007/s00343-016-5131-0 |
Zhao W, Zhao Y Y, Wang Q H, Zheng M P, Zhang P, He Z H. 2010. Spatial and temporal patterns of plankton assemblage structure in a high altitude saline lake, Namuka Co in Northern Tibet, China. CLEAN-Soil, Air, Water, 38(10): 960-968.
DOI:10.1002/clen.v38:10 |
Zhao W, Zheng M P, Xu X Z, Liu X F, Guo G L, He Z H. 2005a. Biological and ecological features of saline lakes in northern Tibet, China. Hydrobiologia, 541(1): 189-203.
DOI:10.1007/s10750-004-5707-0 |
Zhao W. 1991. A review on the Cladocera in inland saline waters. Journal of Dalian Fisheries University, 6(2): 31-41.
(in Chinese with English abstract) |
Zhou W Z, Li H T, Chen Y. 2006. Effects of salinity on food consumption and hunger weight loss of rice-field eel. Reservoir Fisheries, 26(5): 26-28.
(in Chinese with English abstract) |
Zhou X M, Wu C W, Zhang Y X. 2008. Effect of Temperature and body weight on bioenergetics of cultured Pseudosciaena crocea. Journal of Zhejiang Ocean University (Natural Science), 27(3): 291-296.
(in Chinese with English abstract) |
 2018, Vol. 36
2018, Vol. 36