Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZHAO Bin(赵斌), HU Wei(胡炜), LI Chenglin(李成林), HAN Sha(韩莎), XU Tao(徐涛)
- Effects of different levels of sweet potato in feeds on body composition and digestive enzymes activities of Apostichopus japonicas Selenka
- Chinese Journal of Oceanology and Limnology, 36(5): 1835-1842
- http://dx.doi.org/10.1007/s00343-018-7050-8
Article History
- Received Feb. 27, 2017
- accepted in principle Jun. 8, 2017
- accepted for publication Aug. 8, 2017
2 Shandong Province Fisheries Technology Extension Station, Jinan 250013, China;
3 Homey Foods Co. Ltd., Weihai 264305, China
Apostichopus japonicas Selenka (sea cucumber) is an economically important echinoderm species that is distributed along the northern coast of China. Since 2003, the aquaculture industry of A. japonicas has developed rapidly, and it is now one of the main cultured species in Shandong and Liaoning provinces (Li et al., 2010). Many economically valuable macroalgae such as Sargassum spp. have been used as ingredients of feed for A. japonicas, leading to the gradual exhaustion of natural resources. This feed shortage has increased the cost of A. japonicas and also had negative effects on the coastal environment. Therefore, recent research and development of feed for A. japonicas has focused on alternative feed sources or additives. Such materials must be inexpensive, but able to meet the nutrient needs of growing A. japonicas. The alternative materials tested to date include Zostera marina (Liu et al., 2013), Enteromorpha sp. (Liao et al., 2011), Eichhornia crassipes (Wang et al., 2013), alfalfa (Li et al., 2013) and Ophiopholis mirabilis (Jiang et al., 2012). Those studies have identified several materials that have potential as ingredients to enrich feeds for A. japonicas.
Sweet potato is an important economic crop that is widely cultivated in China. Sweet potato production in China accounts for 80% of the world's total (Du et al., 2006). In recent years, there have been several studies on the nutritive value of sweet potato and its functional components (Innami et al., 1998; Shahidul et al., 2003; Le Van An et al., 2003). Other studies have evaluated and analyzed the nutritive values of different parts of sweet potato (Koizumi and Yasui, 1991; O'Sullivan et al., 1996; Wang and Yeh, 1996; Ishida et al., 2000; Wang et al., 2009), and have provided a theoretical foundation for the use of sweet potato as alternative feed source or additive. Preliminary research indicated that addition of sweet potato ingredients to a final concentration of 30% of feed promoted the growth and food ingestion of A. japonicas (Zhao et al., 2016). The aim of this study was to expand the scope of research on the use of sweet potato materials as ingredients in feed for A. japonicas, and to determine whether these materials affected the quality of this important maricultured species. Therefore, we determined the effects of adding sweet potato tuber and stem powders to feed on the body composition of A. japonicas. Specifically, A. japonicas fed with different diets were analyzed to determine their nutrient and amino acid contents, and the activities of their digestive enzymes (protease, amylase, and lipase). The overall aim of this research was to explore the feasibility of using terrestrial plant materials (sweet potato tubers and stems) as the protein source in feed for sea cucumber, and to provide fundamental data and theoretical support for the development of green and novel feeds.
2 MATERIAL AND METHOD 2.1 MaterialHealthy sea cucumbers with average weights from 14.2 g to 17 g were obtained from the Germplasm Research Center of Marine Biology Institute, Shandong Province. Sweet potato tubers and stems were air-dried and ground into a powder with a particle size of < 150 μm.
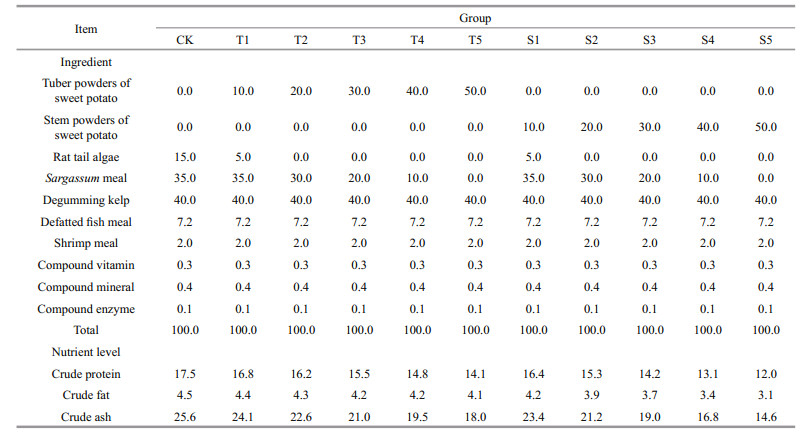
2.2 Experimental design and daily managementCompound feed was purchased from the Qingdao Longer Biological Technology Co., Ltd. The composition of the compound feed was 16% crude protein, 5% crude fat, 8% crude fiber, 6% total phosphorus, 25% crude ash, 0.8% lysine, and 10% moisture. To this basal feed, sweet potato tuber powder (T) or stem powder (S) was added to a final concentration of 10%, 20%, 30%, 40% or 50%, by weight (Table 1). Then, water was added to each group (40%–60%), and the mixtures were rolled, pressed into flakes, dried, and then kept sealed until use.
The compound feeds containing sweet potato materials were supplied at 8% of the total body weight of sea cucumbers. Good-quality dried sea mud was also supplied (5 g mud:1 g feed). The sea cucumbers were fed in plastic boxes (80 cm long, 60 cm wide, 48 cm high). Each box contained 20 sea cucumbers that were fed once per day at 17:00. The experiment consisted of five parallel groups. The salinity of sea water was 30.8–31.6, the pH ranged from 7.8 to 8.3, and water temperature was maintained at 15–20℃. Gentle aeration was supplied to the boxes and 100% of the water was replaced daily. The 60-day experiment began on April 17, 2016.
2.3 Determination methods and data calculations 2.3.1 The measure of the growth and body composition of A. japonicasThe sea cucumbers were starved for 1 day before the start of the experiment. The sea cucumbers were weighed at the beginning (W0 (g); initial body weight) and at the end of the experiment (Wt (g)), where "t" indicates the duration of the experiment. Each sea cucumber was removed from the water and dried in the shade before weighing. The specific growth rate (SGR, %/d) of A. japonicas was calculated as follows:
SGR(%/d)=100×(lnWt−lnW0)/t.
At the end of the experiment, the viscera were taken out and the digestive tract contents were removed. The ratio of viscera to body wall (RVBW) was calculated as follows:
RVBW%=100×(viscera weight/body wall weight).
The crude protein content was measured by the Kjeldahl Method, crude fat was determined by Soxhlet extraction, ash content was determined by combustion in a muffle furnace at 550℃, and moisture content was determined after oven-drying at 103℃.
A portion of each sample was hydrolyzed with hydrochloric acid (6 mol/L) under nitrogen gas for 24 h before detecting 17 amino acids using an automatic amino acid analyzer.
2.3.2 Determination of digestive enzyme activityTo collect coelomic fluid, the sea cucumbers were cut open and the digestive tract and respiratory tissues were removed. The tissues were washed with ultrapure water and then stored at -80℃ until analysis. The intestinal tissues were homogenized for 20 min in pre-cooled ultra-pure water using a glass homogenizer (10 vol water:1 vol intestinal tissue), and the resulting mixture was centrifuged (0–4℃, 1 000 r/min) for 30 min. The supernatant was stored at -20℃ until use in analyses.
The activities of digestive enzymes including protease, amylase, and lipase, were measured using kits developed by the Nanjing Jiancheng Bioengineering Institute. Protease activity was detected using the molybdate ammonium method, and one unit of enzyme activity was defined as the amount of enzyme producing 1 μg amino acids per minute at 37℃. Amylase activity was detected by the turbidimetry method, and one unit of amylase activity was defined as the amount of protein that hydrolyzes 10 mg starch in 30 minutes at 37℃. Lipase activity was measured by the xanthine oxidase method, and one unit of activity was defined as the amount of enzyme consuming 1 μmol substrate in 1 min at 37℃.
The protein content in the enzyme solution was determined by the Coomassie Brilliant Blue G250 Method, with bovine serum albumen as the standard. The activities of various enzymes were expressed as the number of units per mg soluble protein.
2.3.3 Data analysisData were analyzed by one-way analysis of variance and multiple comparisons were conducted using SPSS 22.0 software, with P < 0.05 indicating statistical significance.
3 RESULT 3.1 Effects of sweet potato tuber and stem powders in feed on growth and body composition of A. japonicas 3.1.1 Growth of A. japonicasThe growth of sea cucumbers fed with tuber and stem powders of sweet potato during the 60-day experiment is shown in Table 2. At the end of the experiment, the highest body weight was in the T2 (20%) group (average, 36.32 g). This was significantly higher than that in the control group (CK) (P < 0.05). The body weights of sea cucumbers in the T1 (10%) and T3 (30%) groups (average, 35.43 g and 33.47 g, respectively) were not significantly different from that in CK (P > 0.05). The average weights of sea cucumbers in the T4 (40%) and T5 (50%) groups were lower than that of CK (P < 0.05), with the average weight in T5 (28.43 g) the lowest among all the groups. Among all the groups fed with feed containing sweet potato stem powder, S1 (10%) had the highest average body weight (32.17 g), but it was not significantly different from that of CK (P > 0.05). The sea cucumbers in the S2 (20%), S3 (30%), S4 (40%) and S5 (50%) groups had lower average body weights than that of CK (P < 0.05), with the lowest in the S5 group (22.54 g).

|
The SGR of sea cucumbers in the T groups changed markedly during the experiment (P < 0.05) (Table 2). At the end of the experiment, compared with the SGR in CK, those in T1 and T2 were significantly higher (P < 0.05), that in T5 was significantly lower (P < 0.05), and those in T3 and T4 were not significantly different (P > 0.05). In contrast, the SGRs of the S groups decreased during the experiment (P < 0.05). At the end of the experiment, all S groups except S1 had a notably lower SGR than that of CK (P < 0.05), and the SGR decreased as the proportion of stem powder in feed increased.
There was no significant difference in RVBW among the T groups (P > 0.05), except that the RVBW in T2, 9.45%, was lower than that in CK (P < 0.05). The RVBWs were higher in the S groups than in CK (P < 0.05) and showed an increasing trend, except for those in S1 and S2 (P > 0.05).
3.1.2 Effects on body composition and amino acid compositionTable 3 summarizes the body composition of sea cucumbers fed diets containing sweet potato tuber and stem powders. Among all the components of the body wall, the most abundant was moisture (up to 91.1%), followed by crude protein, and then crude ash. The least abundant component was crude fat (< 1.00%). The stem and tuber of sweet potato had different effects on the nutrient contents of the body wall. The highest crude protein content in the body wall was in the T2 group (5.4%), and this value was significantly higher than those in other groups. The moisture and ash contents in the body wall did not differ significantly between the S and T groups.

|
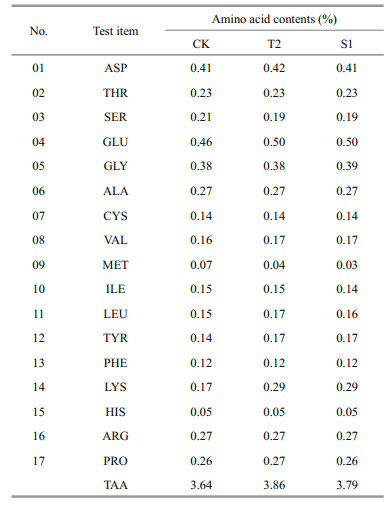
The CK and the two groups showing the best growth, T2 and S1, were selected for amino acid analyses (Table 4). The contents of glutamic acid (Glu) and lysine (Lys) were higher in T2 (0.50% and 0.29%), respectively, than in CK (0.46% and 0.17%, respectively). The methionine (Met) content was slightly lower in T2 (0.04%) and S1 (0.03%) than in CK (0.07%).
|
As the proportion of tuber powder increased, the protease activity in T groups first increased and then decreased, with no significant difference from CK (P > 0.05; Fig. 1). The highest protease activity was in T2 (3.75 U/mg prot), significantly higher than that in T5 (P < 0.05). However, protease activity did not differ significantly among the other T groups (P > 0.05). The protease activity in S groups was not significantly different from that in CK (P > 0.05) when less than 30% stem powder was included in feed, but was significantly lower than that in CK (P < 0.05) when 30% to 50% stem powder was included in feed. The protease activity did not differ significantly between the S and T groups when the feed contained less than 30% sweet potato materials, but was significantly lower in S groups than in T groups when the feed contained 30% to 50% sweet potato materials (P < 0.05).

|
| Figure 1 Protease activity in sea cucumbers supplied with feed containing sweet potato tubers and stems Different letters indicate significant differences within the same dietary group; * indicates significant differences between dietary groups (P < 0.05). |
The sweet potato tuber powder significantly affected amylase activity (P < 0.05, Fig. 2). Amylase activity was significantly higher in the groups supplied with feed containing 30% to 40% tuber powder than in CK. The highest amylase activity was in T4 (0.67 U/mg prot). The amylase activity in other T groups was only slightly higher than that in CK (P > 0.05). The effect of sweet potato stem powder on amylase activity was similar. The highest amylase activity was in S3 (0.66 U/mg prot). This value was significantly higher than that in CK, whereas the amylase activity in other S groups was not significantly different from that in CK (P > 0.05). There were no significant differences in amylase activity between the T and S groups (P > 0.05).

|
| Figure 2 Effects of tubers and stems of sweet potato in feeds on amylase activity of sea cucumbers Different letters indicate significant differences within the same dietary group (P < 0.05). |
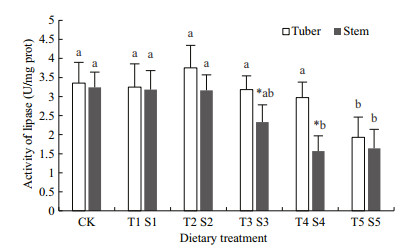
Figure 3 shows the lipase activity of sea cucumbers fed with different proportions of sweet potato tuber and stem powders. The lipase activity in T groups tended to decrease with increasing proportion of tuber powder in feeds, but the only significant difference in lipase activity was between CK and the T5 group (1.93 U/mg protein); this value was significantly lower than that in CK (P < 0.05). The lipase activity in S groups also tended to decrease with increasing proportions of stem powder in feed. When stem powder was added to a final concentration of 40% or higher, the lipase activity was significantly lower than that in CK (P < 0.05). There were striking differences in the lipase activity between T and S groups containing sweet potato materials at 30% to 50% (P < 0.05), but no significant difference in lipase activity between the other T and S groups (P > 0.05).
|
| Figure 3 Effects of sweet potato tuber and stem powders in feed on lipase activity of sea cucumbers Different letters indicate significant differences within the same dietary group; * indicates significant difference between dietary groups (P < 0.05). |
Sea cucumbers have many tiny calcareous ossicles under the surface. Therefore, their bodies have a higher ash content compared with other aquatic animals including eels, Trionyx sinensis, hairtails, prawns, ricefield eels, mandarin fish, and Pinctada martensii (Liu et al., 2011), while their crude protein and crude fat contents are lower. The substances that increase protein content are exclusively dietary, so the proteins supplied in the feed are a key factor in sea cucumber breeding. In this study, the ash content was high, but it was not related to the variations in the feed. However, the protein content was closely related to the proportion of sweet potato tuber and stem powders in the feed. As one of the terrestrial plants added into feeds, sweet potato is rich in starch, vitamins, and minerals (Wang et al., 2005; Bovell-Benjamin, 2007). In tubers of sweet potato, sporamin, a special kind of monomer protein in root plants (Maeshima et al., 1985) accounts for 60% to 80% of soluble protein (Harvey and Boulter, 1983). In this experiment, the highest contents of crude protein were in the T2 and S1 groups, and the crude protein content was higher in the T groups than in the S groups. This is consistent with the research results mentioned above.
Sweet potato is an ideal feed source of protein because its amino acids are abundant and wellbalanced. In this experiment, the amino acid contents of sea cucumbers differed between those fed with sweet potato tuber and stem powders and those in the CK group. The glutamic acid and lysine contents were higher in sea cucumbers fed with sweet potato tuber powder than in those in the CK group, while the methionine contents were slightly lower (0.07% in CK, compared with 0.04% in the T group and 0.03% in the S group). These results represent the amino acid composition of the sea cucumber body wall under these conditions, although the composition can vary depending on the testing methods, environmental factors (season, temperature, salinity), and the growth stage of sea cucumbers. Glutamic acid is important for brain cell function, and for stimulating erythropoiesis. Compared with CK, both the T and S groups showed higher glutamic acid contents, providing further evidence that sweet potato as a feed ingredient effectively improved the quality of sea cucumbers.
4.2 Effects of different proportions of sweet potato materials on digestive enzyme activitiesThe digestive tract is the most important organ for the digestion of food and the assimilation of nutrients. It is characterized by its high plasticity and strong ability to adapt to changes in feed composition, ingestion rate, and metabolic status (Hirche, 1989; West-Eberhard, 1989). The structure and function of the digestive tracts of animals in the Holothuroidea can adjust to changes in food sources and seasons (Fankboner and Cameron, 1985). In this study, there were significant differences in RVBW between S groups and CK, indicating that sea cucumbers can probably improve their ability to absorb protein and other nutrients from food by adjusting the length of the digestive tract. Echinoderms, including sea cucumbers, do not have specialized digestive glands, but their intestinal tracts perform the same function. Their intestinal tracts have abundant blood sinuses under the midgut epithelium to absorb nutrients effectively (Franchini and Ottaviani, 1992), and promote the digestion and assimilation of nutrients in feeds by secreting various digestive enzymes including protease, amylase, and lipase (Cui et al., 2000). To date, more than ten kinds of digestive enzymes have been detected in the digestive tracts of sea cucumbers, including protease, lipase, esterase, glycosidase (amylase, cellulase, alginate lyase, disaccharidase, invertase, and chitinase) and phosphatase (acid phosphatase and alkaline phosphatase) (Choe, 1963). Three important digestive enzymes (protease, amylase, and lipase) were selected and analyzed in this experiment. Sea cucumbers can adapt to different food sources by adjusting the physiological function of the digestive tract and the activities of digestive enzymes. In addition, the activities of various digestive enzymes in intestinal tracts were shown to differ among various feeding stages (Fankboner and Cameron, 1985). In this experiment, the activities of digestive enzymes differed among groups fed with different proportions of sweet potato tuber and stem powders. The protease and lipase activities first increased and then decreased, with the highest protease activity in T2 (20%) and the highest lipase activity in S1 (10%). This result indicated that the addition of tuber and stem sweet potato into feeds in certain proportions can activate certain digestive enzymes, thereby improving the digestive ability of sea cucumbers and promoting their growth. The changes in amylase activity were not consistent with the proportions of tuber and stem powders added to feed. Amylase activity increased as the proportion of sweet potato materials in feed increased up to 40%, but then decreased. According to Dybas and Fankboner (1986), the increase in amylase activity in groups fed with sweet potato was related to microbes in the intestine, rather than the animal itself. Further research is required to explore these responses in more detail.
5 CONCLUSIONThe results of this study show that sweet potato materials as feed ingredients can stimulate digestive enzyme activities in A. japonicas. Therefore, the addition of different proportions of tubers and stems of sweet potato to compound feeds can improve the digestive ability and growth of cultured A. japonicas.
6 DATA AVAILABILITY STATEMENTThe authors declare that all data supporting the findings of this study are available within the article and its supplementary information files.
Bovell-Benjamin A C. 2007. Sweet potato: a review of its past, present, and future role in human nutrition. Advances in Food and Nutrition Research, 52: 1-59.
DOI:10.1016/S1043-4526(06)52001-7 |
Choe S. 1963. Study of Sea Cucumber: Morphology, Ecology and Propagation of Sea Cucumber. Kaibundou Publishing House, Tokyo. 219p.
|
Cui L B, Dong Z N, Lu Y H. 2000. Histological and histochemical studies on the digestive system of Apostichopus japonicus. Chinese Journal of Zoology, 35(6): 2-4.
(in Chinese with English abstract) |
Du L Q, Zhao Y G, Li R F. 2006. Analysis and evaluation on the nutritional components in stem apex of sweet potato. Chinese Agricultural Science Bulletin, 22(6): 99-101.
(in Chinese with English abstract) |
Dybas L, Fankboner P V. 1986. Holothurian survival strategies: Mechanisms for the maintenance of a bacteriostatic environment in the coelomic cavity of the sea cucumber, Parastichopus californicus. Developmental & Comparative Immunology, 10(3): 311-330.
|
Fankboner P V, Cameron J L. 1985. Seasonal atrophy of the visceral organs in a sea cucumber. Canadian Journal of Zoology, 63(12): 2 888-2 892.
DOI:10.1139/z85-432 |
Franchini A, Ottaviani E. 1992. Intestinal cell types in the freshwater snail planorbarius corneus: histochemical, immunocytochemical and ultrastructural observations. Tissue and Cell, 24(3): 387-396.
DOI:10.1016/0040-8166(92)90055-C |
Harvey P J, Boulter D. 1983. Isolation and characterization of the storage protein of yam tubers (Dioscorea rotundata). Phytochemistry, 22(8): 1 687-1 693.
DOI:10.1016/S0031-9422(00)80252-6 |
Hirche H J. 1989. Spatial distribution of digestive enzyme activities of Calanus finmarchicus and C. hyperboreus in Fram strait/Greenland sea. Plankton Research, 11(3): 431-443.
DOI:10.1093/plankt/11.3.431 |
Innami S, Tabata K, Shimizu J, Kusunoki K, Ishida H, Matsuguma M, Wada M, Sugiyama N, Kondo M. 1998. Dried green leaf powders of Jew's mellow (Corchorus), persimmon (Diosphyros kaki) and sweet potato (Ipomoea batatas poir) lower hepatic cholesterol concentration and increase fecal bile acid excretion in rats fed a cholesterolfree diet. Plant Foods for Human Nutrition, 52(1): 55-66.
DOI:10.1023/A:1008031028484 |
Ishida H, Suzuno H, Sugiyama N, Innami S, Tadokoro T, Maekawa A. 2000. Nutritive evaluation on chemical components of leaves, stalks and stems of sweet potatoes (Ipomoea batatas poir). Food Chemistry, 68(3): 359-367.
DOI:10.1016/S0308-8146(99)00206-X |
Jiang S H, Liang J, Sun X. 2012. Study on biochemical compositions of ophiopholis mirabilis and its influence on growth and survival of Apostichopus japonicus as feed ingredients. Feed Industry, 33(16): 13-17.
(in Chinese with English abstract) |
Koizumi H, Yasui A. 1991. Determination of vitamins polyphenols and minerals in roots of sweet potatoes. Report of National Food Research Institute, 55: 1-8.
|
Le V A, Frankow-Lindberg B E, Lindberg J E. 2003. Effect of harvesting interval and defoliation on yield and chemical composition of leaves, stems and tubers of sweet potato (Ipomoea batatas L. (Lam.)) plant parts. Field Crops Research, 82(1): 49-58.
DOI:10.1016/S0378-4290(03)00018-2 |
Li C L, Song A H, Hu W, Zhang Y P, Zhao B, Li Q C, Ma D P. 2010. Status analysis and sustainable development strategy of sea cucumber Apostichopus japonicus Selenka aquaculture industry in Shandong province. Progress in Fishery Science, 31(4): 126-133.
(in Chinese with English abstract) |
Li X, Zhang S Y, Chen S Q, Zou A G, Gao F, Sun H L, Yan J P, Liu C L. 2013. Effects of four feedstuffs on the growth, body composition and digestive physiology of Apostichopus japonicus. Feed Industry, 34(8): 36-40.
(in Chinese with English abstract) |
Liao M J, Hao Z K, Shang D R, Jiang Y, Chen G P, Wang Y G. 2011. Nutrition components analysis of Enteromorpha prolifera and its preliminary study on feeding Apostichopus japonicas. Fishery Modernization, 38(4): 32-36.
(in Chinese with English abstract) |
Liu X F, Xue C H, Wang Y M, Li H Y. 2011. Comparative analysis of nutritive composition in body wall and internal organs of sea cucumber (Apostichopus japonicus) at Rushan. Journal of Fisheries of China, 35(4): 587-593.
(in Chinese with English abstract) |
Liu X J, Zhou Y, Yang H S, Ru S G. 2013. Evaluation of eelgrass debris as a food source for Apostichopus japonicas. Marine Science, 37(10): 32-38.
(in Chinese with English abstract) |
Maeshima M, Sasaki T, Asahi T. 1985. Characterization of major proteins in sweet potato tuberous roots. Phytochemistry, 24(9): 1 899-1 902.
DOI:10.1016/S0031-9422(00)83088-5 |
O'Sullivan J N, Blamey F P C, Asher C J. 1996. Diagnostic criteria for nutrition disorders of sweet potato. Ⅱ: critical nutrient concentrations in leaves. In: Mineral Nutrient Disorders of Root Crops in the Pacific: Proceedings of a Workshop. Nuku'alofa, Kingdom of Tonga. p.39-43.
|
Shahidul I M, Yoshimoto M, Yamakawa O. 2003. Distribution and physiological functions of caffeoylquinic acid derivatives in leaves of sweetpotato genotypes. Journal of Food Science, 68(1): 111-116.
DOI:10.1111/jfds.2003.68.issue-1 |
Wang H Y, Yeh K W. 1996. Cultivar differences in trypsin inhibitory activities of sweet potato leaves and tuberous roots. Taiwania, 41(1): 27-34.
|
Wang W L, Du F L, Xu T C. 2009. Research of nutrition value in sweet potato's stems and leaves and its developments. Food and Nutrition in China, (7): 29-31.
(in Chinese with English abstract) |
Wang W X, Bai Y, Hou W J, Wang H. 2013. Application of Eichhornia crassipes sioms in Apostichopus japonicus feed. Cereal & Feed Industry, (8): 51-54.
(in Chinese with English abstract) |
Wang X C, Hu X Z, Wang J Z. 2005. Study on polysaccharide extracting technology from stem of sweet potato (Ipomoea batatas(L.) Lam.). Journal of Agricultural University of Hebei, 28(2): 51-53, 60.
(in Chinese with English abstract) |
West-Eberhard M J. 1989. Phenotypic plasticity and the origins of diversity. Annual Review of Ecology and Systematics, 20: 249-278.
DOI:10.1146/annurev.es.20.110189.001341 |
Zhao B, Hu W, Li C L, Han S, Yan F. 2016. The nutritional effects of two feedstuffs from sweet potato on feeding and growth of sea cucumber Apostichopus japonicus. Progress in Fishery Sciences, 37(1): 80-86.
|
 2018, Vol. 36
2018, Vol. 36