Institute of Oceanology, Chinese Academy of Sciences
Article Information
- Alexandr V. PRAZUKIN, Elena V. ANUFRⅡEVA, Nickolai V. SHADRIN
- Cladophora mats in a Crimean hypersaline lake: structure, dynamics, and inhabiting animals
- Chinese Journal of Oceanology and Limnology, 36(6): 1930-1940
- http://dx.doi.org/10.1007/s00343-018-7313-4
Article History
- Received Nov. 4, 2017
- accepted in principle Mar. 8, 2018
- accepted for publication Mar. 27, 2018
Filamentous green algae play an important functional role in element cycling and productivity in different water bodies, reaching high biomass. Due to eutrophication during the last decades they have become more abundant in lagoons (Curiel et al., 2004), lakes (Higgins et al., 2012), estuaries (Gubelit and Berezina, 2010), streams (Okada and Watanabe, 2002), and reservoirs (Dondajewska et al., 2007), creating some problems for the human population. Cladophora spp. are among most frequent of those green algae. Phosphorus is generally the limiting nutrient for filamentous green algae development in lakes (Higgins et al., 2012; Song et al., 2017). Only few reports have mentioned nitrogen-limited filamentous green algae growth in aquatic systems (Song et al., 2017); heterotrophic nitrogen-fixing bacteria among Cladophora epibionts may be one of the factors enabling their development (Young et al., 2010; Zulkifly et al., 2012). Cladophora can forms different types of mats, which are very complicated systems including different epibiontic organisms (bacteria, microalgae, infusorians) and free living protists and animals (Pavlovskay et al., 2009; Zulkifly et al., 2012; Shadrin and Anufriieva, 2013). To avoid phosphorus limitation, Cladophora can accelerate P regeneration by excreting alkaline exophosphatase (Song et al., 2017). Epibiontic microalgae and bacteria as well as mat-dwelling infusorians and animals also accelerate phosphorus cycling, thus promoting high development of filamentous green algae.
In hypersaline lakes and lagoons worldwide, filamentous green algae are present and Cladophora spp. dominate when lake salinity exceeds 100 g/L (Gordon et al., 1980; Hammer et al., 1983; Velasco et al., 2006). In hypersaline lakes and lagoons of the Crimea, they may form bottom and floating mats and occupy the large areas with high biomass, up to 4-5 kg (wet biomass)/m2 (Ivanova et al., 1994; Prazukin et al., 2008; Balushkina et al., 2009; Shadrin and Anufriieva, 2013). Animal and infusorian abundance may reach high values in mats (Pavlovskay et al., 2009; Shadrin et al., 2016, 2017). Cladophora mats also influence the aquatic environment, modifying evaporation rate, regimes of temperature, oxygen, pH, Eh, etc. (Prazukin et al., 2008; Shadrin, 2017). Despite this, our knowledge on these mats is very limited. The main goal of this article is to describe and discuss the results of our multiyear study on the structure and dynamics of Cladophora mats in a small Crimean hypersaline lake.
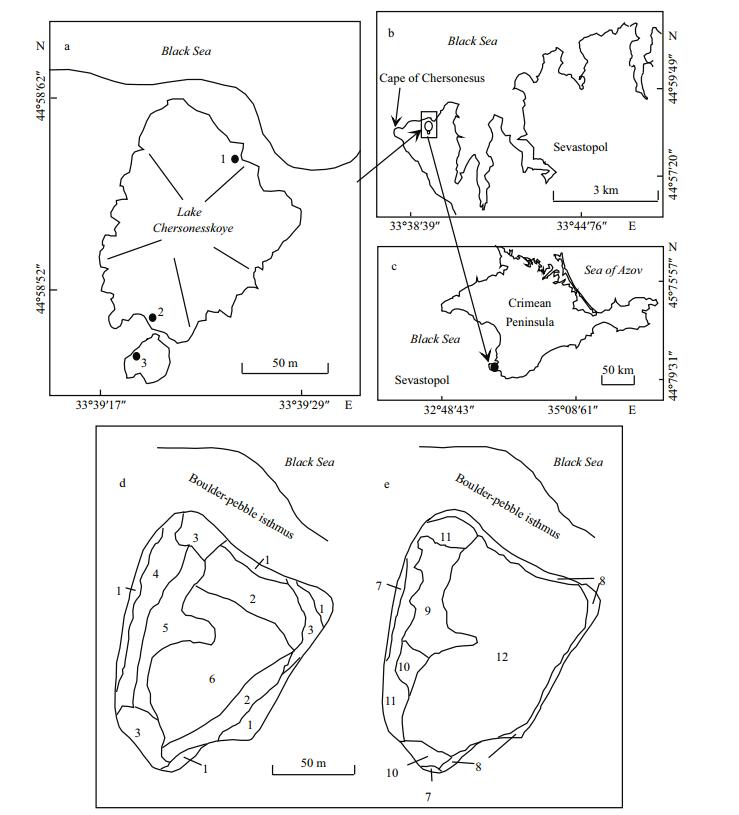
2 MATERIAL AND METHOD 2.1 Study areaIn 2003-2017, complex studies were conducted on Lake Chersonesskoye (44°35′09″N, 33°23′39″E) in the southwestern part of the Crimea, the largest peninsula in the Black Sea (Fig. 1); their main results were published (Kolesnikova et al., 2008; Prazukin et al., 2008; Senicheva et al., 2008; Batogova et al., 2009; Pavlovskay et al., 2009; Gubanov and Bobko, 2012; Prazukin, 2009, 2015), but many results related to mats have not been published before. The lake is shallow, has an oval-oblong shape, the area of the water is 0.05 km2 with a catchment area of 0.92 km2; the average depth is 0.38 m and the largest—about 1.3 m. In summer, due the water level decrease, a shallow small part of the lake is separated by an isthmus. High spatial-temporal variability of abiotic parameters was observed. Intensive water heating is observed starting in April and reaches its maximum in August (30-43℃). In winter the temperature can drop below 0℃; the lowest value in the coldest winter was little below -7°С. The maximum salinity value during the observation period was 360 g/L in the 'small lake' and 160 g/L in the other lake part; the minimum salinity was 35 g/L. The ratio of major ions in the water, as in other hypersaline lakes of marine origin, does not differ from that in the Black Sea. The average daytime pH value in the lake was 8.64; the maximum value can reach 10.0, which is due to the high intensity of photosynthesis. In 2005, the absolute minimum pH (7.73) in upper layer of mat was registered in September; it was associated with the development of sulfate reduction processes under floating mats. In 2005, annual average daytime absolute concentration of oxygen was 5.21 mL/L (102% of saturation). In August 2005, in the presence of hydrogen sulfide in a deeper layer, an absolute maximum of the relative oxygen content (202%) was recorded. In 2005, average phosphate concentration (PO4) was 0.58±0.3 μmol/L, nitrate concentration (NO3) was 2.35±2.06 μmol/L and silicate concentration (SiO3) was 7.94±7.74 μmol/L.
|
| Fig.1 Lake Chersonesskoye in the Crimea (a-c) and schemes of algal mat biomass distribution in the lake (d, e) a. straight lines show the transects for sampling in the lake; d. July 12, 2005; e. August 23, 2005; 1-12: the lake areas as in Table 1. |
The lake surface was almost completely covered by the floating Cladophora mat at the observed point. During the period of observations on the lake phytoplankton, 61 microalgae species were recorded: Dinoflagellata: 19 species, Bacillariophyceae: 15, Chlorophyta: 9, Cyanobacteria: 7, Chrysophyta: 6, Cryptophyta: 3, Euglenoidea: 2. Seventy species of benthic diatoms were noted. Macrophytes were represented by 6 species, 5 of them belong to the filamentous Chlorophyta and one species—to seagrasses (Angiospermae). Twenty-four infusorian and more than 40 animal (Nematoda, Turbellaria, Rotifera, Diptera, Coleoptera, Crustacea) species inhabit the lake. As it was shown before, most part of benthic animals in hypersaline waters (Chironomidae, Harpacticoida, Ostracoda, Nematoda) transits to planktonic life due to high density of water, hypoxic/ anoxic condition near bottom, salt sedimentation (Shadrin and Anufriieva, 2013; Shadrin et al., 2017). This lake is not exclusion from such regularity.
2.2 Sampling and sample processingObservation and sampling of mats, inhabiting them animals and zooplankton was conducted in 2000- 2017 in 2-4 points of the lake. Only in 2005, sampling and observations were made in the summer-autumn period on five transects several times, in 5-10 points on every transect (Fig. 1). On transects, starting from the shore and up to depths of 50 to 55 cm, samples were taken with a cylindrical sampler with a crosssectional area of 452 cm2, which allows seaweed to be taken horizontally throughout the water column. The algae biomass samples were washed in freshwater, dried with filter paper and weighed on an electronic balance WT-250 (Techniprot, Poland). To determine the dry mass, the algae were dried at a temperature of 105℃ to constant weight and weighed on the same balance. On the basis of the obtained data, the volumetric concentration of plant mass in different parts of the lake was calculated from the equations (Prazukin, 2015):
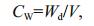 (1)
(1)where CW: dry weight algal biomass per unit of mat volume, mg (dry weight)/cm3; Wd: dry mass of mat sample, mg; V: mat sample volume, cm3.
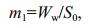 (2)
(2)where m1: wet weight algal biomass per unit of water surface in sampling point, kg (wet weight)/m2; Ww: wet weight of mat sample, kg; S0: the surface area from which the sample was taken, m2.
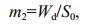 (3)
(3)where m2: dry weight algal biomass per unit of mat surface, mg (dry weight)/cm2.
Animals were selected from the clumps of algae of known mass. The relative number of animals in the mats was determined by dividing the number of counted individuals by the mat sample weight. Zooplankton samples were collected by filtration of 100-150 L of water through a plankton net with a mesh size of 110 μm. Samples were fixed with 4% buffered formalin solution and examined under an Olympus SZ-ST stereomicroscope. Identifications were made using a Carl Zeiss Axio Scope A1 light microscope. Totally more than 150 plankton and mat samples with animals were analyzed. In the lake, benthos, infusorians and plankton were studied in 2005-2006; methods and partly results were described previously (Kolesnikova et al., 2008; Pavlovskay et al., 2009; Anufriieva and Shadrin, 2012). The authors use these results here. At the sampling points, temperature and salinity were evaluated. Temperature was measured by a PHH-830 electronic thermometer, salinity—by a manual Kelilong WZ212 refractometer. The water temperature was measured directly in the floating and bottom mats and in the water outside the mat, the salinity in the floating mats, and the incident radiation at a distance of 3 cm above the surface of the floating mat. Data were subjected to standard statistical processing in Grapher-7, Excel 2007.
3 RESULT 3.1 Structure and dynamics of the mats in the lakeIn 2000-2017, the authors found five species of filamentous green algae (Chlorophyta) in the lake: Cladophora vadorum (Aresch.) Kütz., C. siwaschensis C. Meyer, C. echinus (Biasol.) Kütz., Ulothrix implexa (Kütz.) Kütz., Rhizoclonium tortuosum (Dillw.) Kütz, and seagrass (Angiospermae) Ruppia cirrhosa (Petagna) Grande. Usually each sample contained between one and three species; their composition varied in time. Cladophora spp. and R. cirrhosa were found in the lake throughout the year, other types of algae—during some periods.
Every year, excluding only three of 2000-2017, Cladophora spp. formed bottom and floating mats at salinities at least up to 200 g/L, accompanied by the purple bacteria Chromatium and Ectothiorhodospira (identification was made by L. Gerasimenko, see Shadrin et al., 2008) during certain periods. For July and August 2005, maps were made for the distribution of mats in the lake water area (Fig. 1d, e). In the lake sections where Ruppia was present, Ruppia and Cladophora jointly formed a bottom mat. According mat biomass and structure, characteristic areas in the lake were identified (Table 1). In July and August 2005, Cladophora mats occupied almost the entire water area of the lake (Fig. 2d). The coastal area of the lake up to depths not exceeding 20 cm (the 1st lake area) covered 13% of the total lake area in July and had high biomass (4.1-5.5 kg (wet weight)/m2, Fig. 2c) contributing 29% of total mat biomass in the lake. In August, coastal areas (lake areas 7 and 8) occupied 9% of the lake area and gave 19% of the total macroalgae biomass. In both surveys, in the central part of the lake, mat biomass varied between 1.1 and 2.8 kg (wet weight)/m2). In July, 69% of the lake area was covered by floating mats, and in August—only 37%. In July and August, the total stock of macrophytes was close—30.98 and 31.13 tons (wet weight), respectively; 82%-84% of total biomass was in the bottom mats. In the winter months there was only a thin strip of Cladophora spp. along the shore (Fig. 2a); and small thickets of R. cirrhosa in some sections. In the middle of March, Cladophora bottom and floating mats were beginning to form along the coastline (Fig. 2b), and by mid-August these can occupy 60%-70% of the lake area (Fig. 2d). As a rule, Cladophora spp. dominated in the lake macrophyte biomass in most part of the water area, and only in certain parts, there Ruppia was present in equal proportion. For 17 years of our observations a complete lack of Cladophora spp. in the winter season in three times; and only three times, we observed a very low abundance of Cladophora in the following summer-autumn period without mat forming.
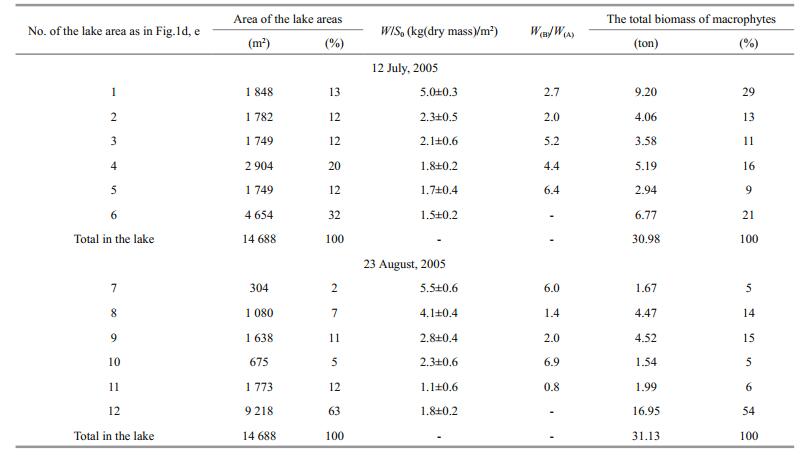
|

|
| Fig.2 Cladophora mats in different seasons of the year in Lake Chersonesskoye a. a thin strip of Cladophora spp. along the shore in the winter months; b. Cladophora spp. in the middle March; c, d. the floating mats in the middle August: c. in the coastal lake part; d. in the central lake parts. |
Distribution of Cladophora was not homogeneous; it is possible to distinguish bottom and floating mats (Fig. 3a). The ratio between them varied with distance from the shore and depth (Fig. 4, I-V). In turn, each mat had a characteristic vertical structure. Near the lake coastline to depths not exceeding 10 cm, a dense multi-layered mat was formed (Fig. 4; 'A', I (a), I (b), Fig. 3a-d). As a rule, there was a dense (36-67 mg (dry)/cm3) interlacing of Cladophora filaments and shoots of Ruppia (Fig. 5). In the vertical structure of the mat, two or three layers were detected, differing in density and color. The top layer (A1, Fig. 4) was either a dense crust of algae, covered with a thin layer of mineral salts (Fig. 3b), or a thin, loose mass of algae with white or light-green color (Fig. 3d, e). Below there was a relatively thick layer of dark-green algae (A2, Fig. 4), sometimes with a large number of different-sized caverns of unknown origin (Fig. 5, Fig. 3d-f) often there were also channels in A1-A2 made by animals (Amphipoda, Coleoptera). The third, lower layer (A3, Fig. 4) was a layer of decaying macrophytes. Visually here the cladophora filaments were green, black and purple-pink in color (Fig. 3b, c). In the general mass, the purple-pink color predominated, which was due to the presence of anoxygenic phototrophs—purple bacteria.

|
| Fig.3 Floating and bottom mats near the coast in Lake Chersonesskoye in August a. floating (1) and bottom (2) mats; b. view from above on the floating mat; c. bottom view on the floating mat; d and e. the floating mat in different points; f. cavities of different size in the upper layer of the floating mat; g. upper layer of the bottom mat (B1, see Figs. 4, 5). 3, 4, 5, 6, 7: layers of the floating mat: 3, 6: upper layer, 4: middle layer, 5, 7: lower layer; 9: Cladophora 'balls'; 10: Ruppia cirrhosa. |
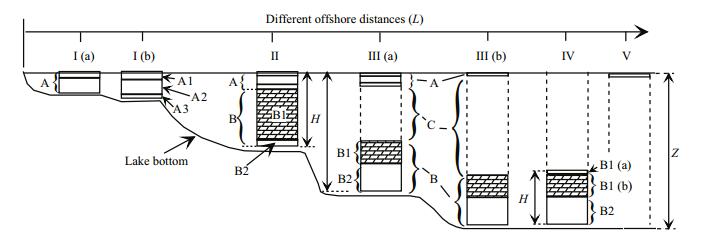
|
| Fig.4 Different states (I-V) of the vertical structure of the floating (A) and bottom (B) Cladophora mats at different depths (Z) in the lake and different offshore distances (L) A1, A2, A3, B1, B2: vertical layers of floating (A) and bottom (B) mats; H: the thickness of the mat; C: space free from macroalgae. |
On the lake sections with depths of 10-20 cm, there were the floating mat (A) and the bottom mat (B) (Fig. 4, II). The morphological structure of the floating mat was identical to the structure of the mat located near the coastline (Fig. 4, I (a), I (b)). Ruppia may 'pierce' floating mats with its shoots, making it motionless (Fig. 5). The bottom mat covered the space from the lower boundary of the floating mat to the bottom and, in turn, was divided into the upper (B1) and lower (B2) layers (Figs. 4, 5). The top layer was formed by the freely floating Cladophora 'balls' of green or salad-green color (Fig. 3g). In the lower layer, there were algae with signs of decay, colored green, black and purple-pink.
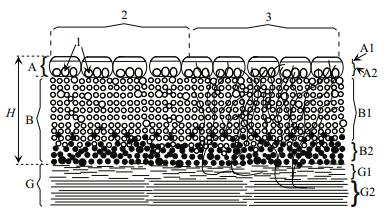
|
| Fig.5 The vertical structure of the floating and bottom mats at depths 10-20 cm Upper (A1) and lower (A2) layers of the floating mat (A); upper (B1) and lower (B2) layers of the bottom mat (B); 'liquid' (G1) and 'solid' (G2) layers of bottom biogenic sediments (G). H: the thickness of the mat in the lake; 1: cavities of different sizes in the upper layer of the floating mat; 2: mat formed only by Cladophora spp.; 3: mat formed by Cladophora spp. and Ruppia cirrhosa. |
At sites with a depth of more than 20-25 cm, the relative location of the floating and bottom mats and their characteristic vertical structure were variable. In some cases, both mats were simultaneously present (Fig. 4, (III (a), III (b)), and there was a space between them, free from macrophytes (Fig. 4, zone 'C'). In other cases, only the bottom (Fig. 4, IV) or only the floating (Fig. 4, V) mat formed. The vertical structure of the bottom mat (Fig. 4, IV) was similar in many respects to the structure of the floating mat: a thin surface layer of light green color (B1 (a)), beneath which there is a layer of freely floating Cladophora green 'balls' (B1 (b)) and below that—a layer of decaying algae (B2). In all cases, below the zone 'B' there was a 'liquid' layer of bottom sediments of biogenic origin (Fig. 5; G1)—a mineral-organic complex formed as a result of the decomposition of algae and animal remains that emerged over a short period of time, for example, for the spring-summer season. A thick 'solid' layer was below this (Fig. 5; G2), accumulated during the period of existence of the lake.
Figure 6 shows the structural characteristics of mats, located at different distances from the shore with different depth. The mat thickness (h(A)) decreased with increasing depth (Fig. 6a). The thickness of the mat lying near the coastline and the floating mat near the shore reached 6-7 cm; thefl oating mat of the open lake sections did not exceed one centimeter. The concentration of dry mass per volume unit of the mat (CW) also changed with depth (Fig. 6b). In September, in the shallow lake part, to the depth not exceeding 30 cm, two maximum of CW were detected: the first near the shore, and the second at a considerable distance from the shore. In the latter case, the floating mat was a thin (0.9 cm), dense (85 mg (dry)/cm3) continuous algae film at the waterair boundary. In other cases (Fig. 6b), especially in the open part, the floating mat was not dense and had an open-work appearance, while the volumetric concentration of dry matter in floating mat varied from 4.6 to 30 mg (dry)/cm3. The amount of dry mass calculated per unit surface (W(A)/S0, W(B)/S0) varied widely and decreased with depth (Fig. 6c, d). In most cases, the bottom mat biomass exceeded that of the floating mat (Fig. 6f). In general, the total biomass of the bottom and floating mats in the lake areas at depth up to 30 cm was within a stable range of values from 100 to 290 mg (dry)/cm2 (Fig. 6e).
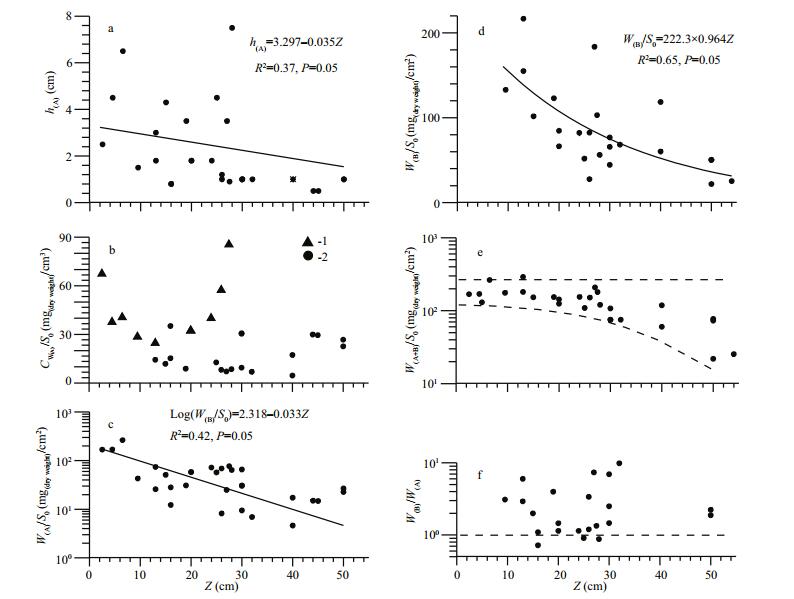
|
| Fig.6 The structural characteristics of Cladophora mats at different depths in the lake (Z) varying with offshore distance (L) (see also Fig. 4) a. the thickness of the floating mat, h(A); b. dry mass of the floating mat, calculated per unit water volume (CW(A)) (1: for near shore section at 14 September 2005, 2: June-September 2005 average for whole lake); c. dry mass of the floating mat, calculated per surface unit (W(A)/S0); d. dry mass of the bottom mat, calculated per surface unit (W(B)/S0); e. total dry mass of both mats, calculated surface unit (W(A+B)/S0); f. the biomass ratio between the bottom and floating mats (W(B)/W(A)). |
All animal species found in the lake were encountered also in the mats. Some groups were found only in the mats such as Amphipoda (Orchestia gammarellus (Pallas, 1766), O. mediterranea (Costa, 1853)) and several species of Coleoptera. The most common and abundant in mats were the same animal species as in the plankton: HarpacticoidaCletocamptus retrogressus Schmankevitsch, 1875 and Ostracoda Eucypris mareotica (Fischer, 1855). Amphipoda and some Coleoptera made the channels in dense mats near the lake coastline, promoting ventilation of the mats. In all cases, abundance of animals in the mats was higher by order of magnitude than in the plankton far from mats (Table 2). Under mats, animal abundance was much low or animal were absent due to H2S presence.
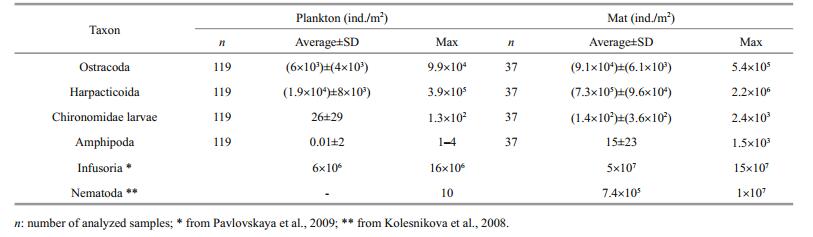
|
The formation of mats of filamentous algae usually in shallow water bodies with high concentrations of nutrients as in our case and their effects on the environment have been discussed (McGlathery et al., 1997; Scheffer et al., 2003; Prazukin et al., 2008; Prazukin, 2015). There are competitive relationships for solar radiation and nutrient elements between the bottom and floating mats (McGlathery et al., 1997; Scheffer et al., 2003; Green and Fong, 2015). In our case, the strongest competitive relations between the floating and bottom mats were shown to exist in the shallow part of the lake (Fig. 4).
Such competition can be explained by an observed decrease of intensity of light, temperature, dissolved oxygen, C/P and C/N ratios with the depth (Phillips et al., 1978; O'Neal and Lembi, 1983; Eiseltová and Pokorný, 1994; Gubanov and Bobko, 2012; Saunders et al., 2012). In a floating Pithophora mat, only 1% of the incident light reached a depth of 1 cm (O'Neal and Lembi, 1983), in some Cladophora mats, the percentage of sun radiation penetrating was 2% (Eiseltová and Pokorný, 1994), and in the dense mats of Chaetomorpha linum (Müller) Kütz., light penetration was restricted to 8 cm (Krause-Jensen et al., 1996). Due to the high levels of solar radiation the upper layers of the mats may suffer from oxidative stress and photoinhibition (Jiang and Qiu, 2005), whereas algae in the lower layer may have to cope with very low levels of photosynthetically active radiation (Vergara et al., 1997). Earlier, the authors showed that the concentration of carotenoids and chlorophyll a and b per biomass unit in the layer of cladophora 'balls' (B1) was approximately twice as high as in the lower layer of the floating mat (A1) (Prazukin et al., 2008), which indicates an adaptive response of algae to a reduced light intensity in the lower part of the mat (Berner et al., 1989). It was shown that the intensity of photosynthesis in a mat decreased from 0.7 μg C/(mg·h) (upper 1-4 cm) to 0.2 μg C/(mg·h) at a depth of 9 cm (Prazukin, 2009). Perhaps below the layer of Cladophora 'balls' (B1), oxygenic photosynthesis was practically impossible. In the 'B2' layer, decomposition processes predominantly occurred, the Cladophora filaments were mostly colored black and purple-pink due to the presence of anoxygenic phototrophs—purple bacteria (Fig. 3b, c, e). That's why highest O2 concentration (200% of saturation) was observed in an upper layer of a mat, and H2S was detected under the floating mat (Gubanov and Bobko, 2012), and anoxic zones are a common phenomenon under such mats in the Crimean hypersaline lakes (Pavlovskay et al., 2009; Shadrin and Anufriieva, 2013; Shadrin et al., 2016).
Animals are a common component of the mat, reaching abundance as high as in mats of other Crimean hypersaline lakes (Ivanova et al., 1994; Balushkina et al., 2009; Shadrin and Anufriieva, 2013; Shadrin et al., 2016, 2017). These animals are highly halotolerant and can dwell at salinities up to 280-350 g/L (Anufriieva, 2014, 2015; Shadrin et al., 2017). Such a high concentration of infusorians and animals (Table 2) and their small sizes (high intensity of metabolism) provide a high rate of nutrient regeneration in mats. Inhospitable conditions on the bottom (oxygen deficit first) push benthic animals transit to live in mats or plankton. Cladophora mats contribute to the high number and diversity of animals in them first providing food—a lot of epibiontic microalgae on cladophora filaments, etc.
Ecosystems of some hypersaline lakes in the Crimea, like the lake studied, can be in various alternative states, when in one of them, the main primary producer is Cladophora mats, and in the other—phytoplankton (Ivanova et al., 1994; Balushkina et al., 2009; Shadrin and Anufriieva, 2013; Shadrin, 2014; Prazukin, 2015). Ecosystems with mats are characterized by an increased intensity of primary production. For example, in Lake Tobechikskoye in August 2005, there was intensive development of Cladophora mats, and their primary production (47 g C/(m2·d)) exceeded the primary production of plankton by 69 times (Balushkina et al., 2009). A similar thing was noted for Lake Bakalskoye (Shadrin and Anufriieva, 2013). It should also be noted that the intensive development of Cladophora spp. with mat formation in the lakes contributes to the growth of the number and diversity of animals and to the decrease of the sedimentation and resuspension of bottom sediments, thereby increasing the transparency of the water (Balushkina et al., 2009; Shadrin and Anufriieva, 2013).
Floating and bottom mats constitute a whole integrated spatially organized system, which in addition to Cladophora spp. includes different groups of organisms: plants, algae, animals, and bacteria. Our results allow us to conclude that in the vertical structure of the mat (Fig. 2), there is a zone where photosynthesis predominates (A, B1) and a decomposition zone (B2); together these two processes (production and decomposition of organic matter) within the boundaries of the mat form a cycle of matter, which determines the structural and functional integrity of the system. Heterotrophic organisms (numerous bacteria, infusorians and animals) play an important role in the regeneration of nutrients within the mat. Internal circulation of substances takes place inside the mat contributing to the maintenance of high algal biomass. Due to all the above, the aquatic ecosystem with floating mats is a stable system (Scheffer et al., 2003) capable to exist in a wide range of temperatures and salinities. Further comprehensive study of these unique communities will expand our understanding of the patterns of functioning, dynamics and transformation of aquatic ecosystems.
6 ACKNOWLEDGMENTThe author thanks to anonymous reviewers for valuable comments. The study was carried under support of the Russian Academy of Sciences for the A. O. Kovalevsky Institute of Marine Biological Research of RAS.
Anufriieva E V, Shadrin N V. 2012. Crustacean diversity in hypersaline Chersonessus Lake (Crimea). Optimization and Protection of Ecosystems, 7: 55-61.
|
Anufriieva E V. 2015. Do copepods inhabit hypersaline waters worldwide? A short review and discussion. Chinese Journal of Oceanology and Limnology, 33(6): 1 354-1 361.
DOI:10.1007/s00343-014-4385-7 |
Anufriieva E. 2014. Copepods in hypersaline waters worldwide: diversity, environmental, social, and economic roles. Acta Geologica Sinica (English Edition), 88(S1): 43-45.
DOI:10.1111/1755-6724.12266_2 |
Balushkina E V, Golubkov S M, Golubkov M S, Litvinchuk L F, Shadrin N V. 2009. Effect of abiotic and biotic factors on the structural and functional organization of the saline lake ecosystems. Zhurnal Obshchei Biologii, 70(6): 504-514.
|
Batogova E A, Gerasimova O V, Shadrin N V. 2009. Cladophora mats as unique communities of hypersaline lakes. In: Proceedings of the International Conference of Young Scientists "Actual problems of botany and ecology" (11-15 August 2009, Kremenets). Ternopil. p.17-18.
|
Berner T, Dubinsky Z, Wyman K, Falkowski P G. 1989. Photoadaptation and the "package" effect in Dunaliella tertiolecta (Chlorophyceae). Journal of Phycology, 25(1): 70-78.
DOI:10.1111/jpy.1989.25.issue-1 |
Curiel D, Rismondo A, Bellemo G, Marzocchi M. 2004. Macroalgal biomass and species variations in the Lagoon of Venice (Northern Adriatic Sea, Italy): 1981-1998. Scientia Marina, 68(1): 57-67.
DOI:10.3989/scimar.2004.68n1 |
Dondajewska R, Frankowski T, Wojak P. 2007. Changes in the vegetation of filamentous green algae in the Antoninek preliminary reservoir. Oceanological and Hydrobiological Studies, 36(S1): 121-128.
|
Eiseltová M, Pokorný J. 1994. Filamentous algae in fish ponds of the Třeboň Biosphere Reserve-ecophysiological study. Vegetatio, 113(2): 155-170.
|
Gordon D M, Birch P B, McComb A J. 1980. The effect of light temperature and salinity on photosynthetic rates of an estuarine cladophora. Botanica Marina, 23: 749-755.
|
Green L, Fong P. 2015. The good, the bad and the Ulva: the density dependent role of macroalgal subsidies in influencing diversity and trophic structure of an estuarine community. Oikos, 125(7): 988-1.
DOI:10.1111/oik.02860 |
Gubanov V I, Bobko NI. 2012. Hydrological and hydrochemical characteristics of the salt lake at Cape Сhersones (Sevastopol, Crimea). Mar. Ecol. J., 11(4): 18-26.
|
Gubelit Y I, Berezina N A. 2010. The causes and consequences of algal blooms: the Cladophora glomerata bloom and the Neva estuary (eastern Baltic Sea). Marine Pollution Bulletin, 61(4-6): 183-188.
DOI:10.1016/j.marpolbul.2010.02.013 |
Hammer U T, Shamess J, Haynes R C. 1983. The distribution and abundance of algae in saline lakes of Saskatchewan, Canada. Hydrobiologia, 105(1): 1-26.
DOI:10.1007/BF00025173 |
Higgins S N, Pennuto C M, Howell E T, Lewis T W, Makarewicz J C. 2012. Urban influences on Cladophora blooms in Lake Ontario. Journal of Great Lakes Research, 38(S4): 116-123.
|
Ivanova M, Balushkina E, Basova S. 1994. Structural functional reorganization of ecosystem of hyperhaline Lake Saki (Crimea) at increased salinity. Russian Journal of Ecology, 3(2): 111-126.
|
Jiang H B, Qiu B S. 2005. Photosynthetic adaptation of a bloom‐forming cyanobacterium Microcystis aeruginosa (Cyanophyceae) to prolonged UV‐B exposure. Journal of Phycology, 41(5): 983-992.
DOI:10.1111/jpy.2005.41.issue-5 |
Kolesnikova E A, Mazlumyan S A, Shadrin N V. 2008. Seasonal dynamics of meiobenthos fauna from a salt lake of the Crimea. In: Proceedings of the 5th International Conference of Environmental Micropaleontology, Microbiology and Meiobenthology (EMMM). University of Madras, Chennai, India. p.155-158.
|
Krause-Jensen D, McGlathery K, Rysgaard S, Christensen P B. 1996. Production within dense mats of the filamentous macroalga Chaetomorpha linum in relation to light and nutrient availability. Marine Ecology Progress Series, 134: 207-216.
DOI:10.3354/meps134207 |
McGlathery K J, Krause-Jensen D, Rysgaard S, Christensen P B. 1997. Patterns of ammonium uptake within dense mats of the filamentous macroalga Chaetomorpha linum. Aquatic Botany, 59(1-2): 99-115.
DOI:10.1016/S0304-3770(97)00026-0 |
Okada H, Watanabe Y. 2002. Effect of physical factors on the distribution of filamentous green algae in the Tama River. Limnology, 3(2): 121-126.
DOI:10.1007/s102010200014 |
O'Neal S W, Lembi C A. 1983. Effect of simazine on photosynthesis and growth of filamentous algae. Weed Science, 31(6): 899-903.
|
Pavlovskay T V, Prazukin A V, Shadrin N V. 2009. Seasonal phenomena in infusoria community in hypersaline Lake Khersonesskoye (Crimea). Marine Biological Journal, 8(2): 53-63.
|
Phillips G L, Eminson D, Moss B. 1978. A mechanism to account for macrophyte decline in progressively eutrophicated freshwaters. Aquatic Botany, 4: 103-126.
DOI:10.1016/0304-3770(78)90012-8 |
Prazukin A V, Bobkova A N, Evstigneeva I K, Tankovska I N, Shadrin N V. 2008. Structure and seasonal dynamics of the phytocomponent of the bioinert system marine hypersaline lake on cape of Chersonesus (Crimea). Marine Biological Journal, 7(1): 61-79.
|
Prazukin A V. 2009. Photosynthetic activity of the vegetation in Lake Chersonesskoye (Crimea) and its structural and functional organization. Systems of control for environment. Tools and Monitoring, 5: 370-376.
|
Prazukin A V. 2015. Ecological phytosystemology. Pero Press, Moscow. 375p. (in Russian)
|
Saunders L L, Kilham S S, Winfield Fairchild G, Verb R. 2012. Effects of small‐scale environmental variation on metaphyton condition and community composition. Freshwater Biology, 57(9): 1 884-1 895.
DOI:10.1111/fwb.2012.57.issue-9 |
Scheffer M, Szabó S, Gragnani A, van Nes E H, Rinaldi S, Kautsky N, Norberg J, Roijackers R M M, Franken R J M. 2003. Floating plant dominance as a stable state. Proceedings of the National Academy of Sciences of the United States of America, 100(7): 4 040-4 045.
DOI:10.1073/pnas.0737918100 |
Senicheva M I, Gubelit Y, Prazukin A V, Shadrin N V. 2008. Phytoplankton of the Crimean hypersaline lakes. In: Tokarev Yu N, Finenko Z Z, Shadrin N V eds. Microalgae of the Black Sea: Problems of biodiversity conservation and biotechnological use. ECOSI-Gidrofizika, Sevastopol. p.93-100. (in Russian)
|
Shadrin N V, Anufriieva E V, Belyakov V P, Bazhora A I. 2017. Chironomidae larvae in hypersaline waters of the Crimea: diversity, distribution, abundance and production. The European Zoological Journal, 84(1): 61-72.
DOI:10.1080/11250003.2016.1273974 |
Shadrin N V, Anufriieva E V. 2013. Climate change impact on the marine lakes and their Crustaceans: the case of marine hypersaline Lake Bakalskoye (Ukraine). Turkish Journal of Fisheries and Aquatic Sciences, 13: 603-611.
|
Shadrin N V, Mykhodyuk O S, Naidanova O G, Voloshko L N, Gerasimenko L M. 2008. Benthic cyanobacteria in hypersaline lakes of the Crimea. In: Tokarev Yu N, Finenko Z Z, Shadrin N V eds. Microalgae of the Black Sea: Problems of Biodiversity conservation and biotechnological use. ECOSI-Gidrofizika, Sevastopol. p.100-112. (in Russian)
|
Shadrin N V, Sergeeva N G, Latushkin A A, Kolesnikova Е А, Kipriyanova L M, Anufriieva E V, Chepyzhenko A A. 2016. Transformation of Gulf Sivash (the Sea of Azov) in conditions of growing salinity: changes of meiobenthos and other ecosystem components (2013-2015). Journal of Siberian Federal University Biology, 9(4): 452-466.
|
Shadrin N V. 2017. Hypersaline lakes as polyextreme habitats for life. In: Zhemg M, Deng T, Oren A eds. Introduction to Salt Lake Sciences. Science Press, Beijing, China. p.173- 178.
|
Shadrin N. 2014. Alternative states of saline lake ecosystems and development of salinology. Acta Geologica Sinica (English Edition), 88(S1): 434-435.
DOI:10.1111/1755-6724.12280_9 |
Song C L, Cao X Y, Zhou Y Y, Shadrin N. 2017. Filamentous green algae, extracellular alkaline phosphatases and some features of the phosphorus cycle in ponds. Marine Biological Journal, 2(1): 66-78.
DOI:10.21072/mbj.2017.02.1.07 |
Velasco J, Millán A, Hernández J, Gutierrez C, Abellán P, Sánchez D, Ruiz M. 2006. Response of biotic communities to salinity changes in a Mediterranean hypersaline stream. Saline Systems, 2: 12.
DOI:10.1186/1746-1448-2-12 |
Vergara J J, Pérez-Lloréns J L, Peralta G, Hernández I, Niell F X. 1997. Seasonal variation of photosynthetic performance and light attenuation in Ulva canopies from Palmones river estuary. Journal of Phycology, 33(5): 773-779.
DOI:10.1111/j.0022-3646.1997.00773.x |
Young E B, Tucker R C, Pansch L A. 2010. Alkaline phosphatase in freshwater Cladophora-epiphyte assemblages: regulation in response to phosphorus supply and localization. Journal of Phycology, 46(1): 93-101.
DOI:10.1111/(ISSN)1529-8817 |
Zulkifly S, Hanshew A, Young E B, Lee P, Graham M E, Graham M E, Piotrowski M, Graham L E. 2012. The epiphytic microbiota of the globally widespread macroalga Cladophora glomerata (Chlorophyta, Cladophorales). American Journal of Botany, 99(9): 1 541-1 552.
DOI:10.3732/ajb.1200161 |
 2018, Vol. 36
2018, Vol. 36


