Institute of Oceanology, Chinese Academy of Sciences
Article Information
- Anastasia KOMOVA, Anna MELNIKOVA, Zorigto NAMSARAEV, Roman ROMANOV, Vera STRAKHOVENKO, Ekaterina OVDINA, Nadezhda ERMOLAEVA
- Chemical and biological features of the saline Lake Krasnovishnevoye (Baraba, Russia) in comparison with Lake Malinovoe (Kulunda, Russia): a reconnaissance study
- Chinese Journal of Oceanology and Limnology, 36(6): 1993-2001
- http://dx.doi.org/10.1007/s00343-018-7333-0
Article History
- Received Feb. 21, 2018
- accepted in principle May. 10, 2018
- accepted for publication Jun. 13, 2018
2 Central Siberian Botanical Garden of the Siberian Branch of the Russian Academy of Sciences, Novosibirsk 630090, Russia;
3 V. S. Sobolev Institute of Geology and Mineralogy of the Russian Academy of Sciences, Novosibirsk 630090, Russia;
4 Institute for Water and Environmental Problems, Siberian Branch of the Russian Academy of Sciences, Barnaul 656038, Russia
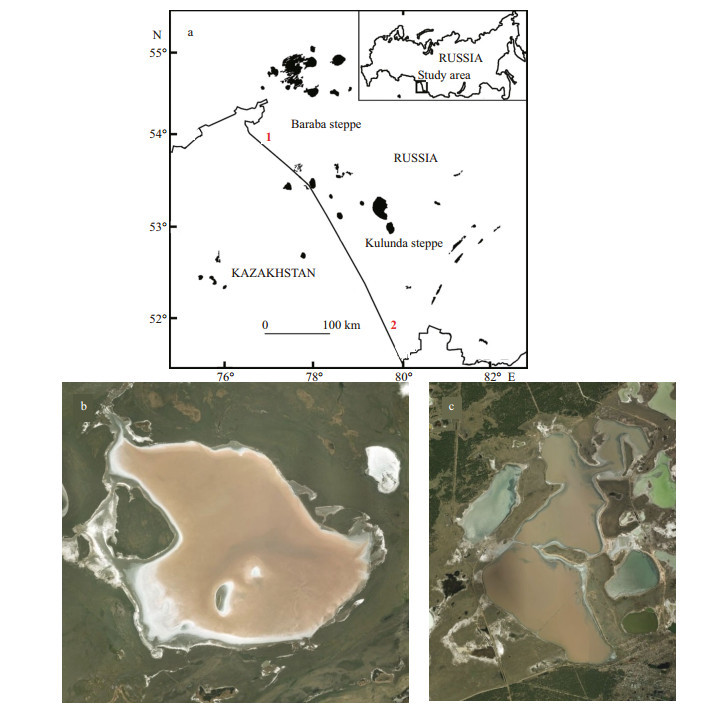
Saline and soda lakes are extreme environments that, in spite of the harsh conditions, can be highly productive (Melack and Kilham, 1974; Grant, 2006). Besides, the microbial communities of such lakes drive the main biogeochemical processes (Sorokin et al., 2011) and have been proposed as an analogue of the terrestrial ecosystems of early Proterozoic era (Zavarzin, 1993). The Baraba and Kulunda steppes are situated in southwestern Siberia, about 300 km distant from each other (Fig. 1). The soils in these steppes are similar in chemical and granulometrical content. The saline and soda lakes of the area are typical for the cryo-arid zone. The main geochemical characteristics of the lakes of Baraba and Kulunda steppes have been studied earlier (Strakhovenko et al., 2010; Strakhovenko, 2011). In Baraba steppe the lakes are mostly of the sodium bicarbonate type (chloride or sulfate are present to a lesser extent), with magnesium and sodium as major cations. The salinity varies from 0.131 to 1.902 g/L, pH 8–10. The majority of lakes of Kulunda steppe belong to the chloridesodium type, and soda lakes are less common. The salinity varies from 0.79 to 396 g/L, pH 7.6–10.6 (Savchenko, 1997; Sorokin et al., 2011; Strakhovenko et al., 2017).
|
| Fig.1 Location of Baraba and Kulunda steppes and satellite images of the studied lakes captured in July 2015 a. 1: Lake Krasnovishnevoye; 2: Lake Malinovoe; b. Lake Krasnovishnevoye; c. Lake Malinovoe (satellite images: courtesy of the U.S. Geological Survey, LandsatLook Viewer). |
The microbial processes (Kompantseva et al., 2009; Sorokin et al., 2010; Tourova et al., 2014; Sorokin et al., 2015; Namsaraev et al., 2018) and microbial diversity (Foti et al., 2008; Gorlenko et al., 2009; Kompantseva et al., 2010; Samylina et al., 2014) of Kulunda hypersaline lakes were intensively studied. Nevertheless, the majority of the microbiological studies in Kulunda steppe focused on the Tanatar group of soda lakes where extremely high salinity and alkalinity lead to the formation of haloalkaliphilic communities. The information about biota in the saline neutral lakes of the Kulunda region is scarce. Little is known about the hydrobionts and phototrophic communities in the saline lakes of the Baraba steppe (Vesnina et al., 2005; Bryanskaya et al., 2016). The aim of this work was to elucidate the main chemical, mineralogical, and biological features of Lake Krasnovishnevoye, a representative of the saline lakes of Baraba steppe, in comparison with saline Lake Malinovoe from Kulunda.
2 MATERIAL AND METHOD 2.1 Sampling siteThe general characteristics of Lake Krasnovishnevoye (Baraba steppe, South Siberia, Russia) and Lake Malinovoe (Kulunda steppe, South Siberia, Russia) are summarized in Table 1. Both are typical shallow drainless steppe lakes, very similar in its water regime and functioning. Samples of water, bottom sediments, salt crust, phyto- and zooplankton from the lakes Krasnovishnevoye and Malinovoe were collected in July–August 2015; phyto- and zooplankton samples in Lake Krasnovishnevoye were also taken in August 2003.
|
Water and bottom sediments from Lake Krasnovishnevoye and from Lake Malinovoe were collected for chemical analysis. The amount of total dissolved solids (TDS) and pH of water were measured by EcoSkan and Anion 4150 pH-meters/conductometers. The bottom sediments were sampled with a vacuum lock cylindrical corer (NPO Taifun), 82 mm×50 cm. The elemental composition of the lake waters and sediments were determined by the atomicabsorption method (Solaar M6, Thermo Electron Corporation). The mineral composition was determined by X-ray phase analysis on an ARL X'TRA X-ray diffractometer. The morphology, phase and elemental composition of the sediments were studied with a MIRA 3 TESCAN (SEM) with Si(Li) detector (Oxford Instruments, UK). The anion composition of the lake waters was determined by high performance liquid chromatography (HPLC) with the use of a Prominence 20 LC-HPLC system (Shimadzu, Japan) with a conductivity meter and a Star-Ion A300 column 10, 4.6 mm (Phenomenex, United States). The content of bicarbonates was determined by potentiometric titration with the use of an ATP-02 automatic titrator (Akvilon, Russia) in accordance with the procedure PND F 14.2.99-97 (State Standard PND F 14.2.99-97, 1997).
Samples of phytoplankton were collected from the near-surface layer and were concentrated on membrane filters with pore diameter of 0.55–0.65 μm at lowest possible pressure after light fixation with formaldehyde solution. The samples were counted by light microscopy. To calculate the biomass (biovolume), cell volumes were approximated as simple or combined geometric figures. Phytoplankton was identified with the use of a guide (Komárek and Fott, 1983).
Samples of zooplankton from Krasnovishnevoye and Malinovoe lakes were collected by filtering 100 L of lake water through an Apstein net (mesh width 64 μm). Zooplankton was identified with the use of a guide to freshwater invertebrates (Tsalolikhin, 1995).
For the studies of anoxygenic phototrophic bacteria (APB) samples of both sediments (below the salt crust) and water of Lake Krasnovishnevoye and sediments of upper layer and water of Lake Malinovoe were collected. Sterile 50-mL centrifuge tubes were filled 3/4 of the volume with sediments and water to avoid excessive process of sulfate reduction. The tubes were then kept in the dark in the refrigerator at 4℃.
In order to reveal the culturable diversity of APB in Lake Krasnovishnevoye, a series of enrichment cultures on the media covering various physicochemical conditions (pH and NaCl concentration) was prepared. The enrichment cultures were then transferred to the agar shakes in order to study the appearing colonies and to isolate pure cultures. For the enrichment cultures preparation a culture medium was made with a range of NaCl content (g/L): 10, 20, 50, 70, 100, 150, 200) and pH (6.8, 7.0, 7.5, 8.0, 8.5, 9.0) using a basic liquid medium containing (g/L): K2SO4: 0.53, MgSO4: 10, Na2S2O3: 0.5, NH4Cl: 0.2, KH2PO4: 0.2, CaCl2·6H2O: 0.4, NaHCO3: 0.3, Na2S·9H2O: 0.05, C2H3NaO2: 1, yeast extract: 0.1, vitamin B12: 20 μg, trace element solution (Pfennig and Lippert, 1966): 1 mL. pH was maintained using the phosphate buffer (pH 6.8–8.0) and carbonate-bicarbonate buffer (8.5–9.0). The 60-mL glass vials with rubber stoppers were completely filled with the medium and 1 mL of the lake sediments added as inoculum. The enrichment cultures were incubated in a luminostat at temperature of 20–25℃ and light intensity of about 2 000 lx for 1 month. The obtained enrichment cultures of anoxygenic phototrophic bacteria were examined under the Nikon Eclipse E200MV light microscope with a phase contrast system.
Pure cultures of APB were isolated using the serial agar shake dilution method (Biebl and Pfennig, 1981; Herbert, 1990) using medium with a composition corresponding to that of the enrichment culture. The colonies were then transferred to liquid medium to perform physiological tests. The optimum NaCl content was determined using the basic liquid medium with various amounts of NaCl added (in percentage of 0, 1, 2, 3, 5, 7, 10, 12, 15, 17, 20, and 22) and pH 8.0. The optimum of pH was determined in the basic liquid medium with various initial pH (6.5, 7.5, 8.5, 9.0, 9.5, and 10.0) and 3% of NaCl. Growth was determined by measuring OD650 using a Thermo Fisher Scientific G10S UV-Vis spectrophotometer after incubation for 5 days. Autotrophic growth with sulfide (Na2S·9H2O: 0.3 g/L), thiosulfate (Na2S2O3: 0.5 g/L) and elemental sulfur was tested on the medium with the optimal NaCl content and pH and with yeast extract and acetate omitted. The 16S rRNA analysis was outsourced to Evrogen JSC (Moscow, Russia).
The APB from Lake Malinovoe were detected using the serial agar shake dilution method. The medium contained (g/L): K2SO4: 0.4, MgSO4: 5, Na2S2O3: 0.5, NH4Cl: 0.2, KH2PO4: 0.2, CaCl2·6H2O: 0.4, NaCl: 200, NaHCO3: 1, Na2S·9H2O: 0.1, C2H3NaO2: 1, yeast extract: 0.1, vitamin B12: 20 μg, trace element solution (Pfennig and Lippert, 1966): 1 mL, pH 7.8. The grown colonies were examined under the Nikon Eclipse E200MV light microscope with a phase contrast system.
The APB were identified to the genus level according to the morphology of cells and colonies, as well as the absorption spectra of living cells as outlined by Imhoff (2005).
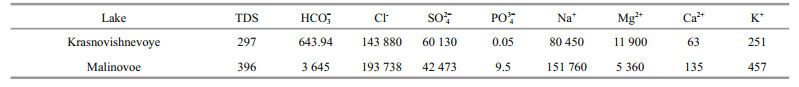
3 RESULT 3.1 Water chemistryThe chemical composition of lakes Krasnovishnevoye and Malinovoe in 2015 is given in Table 2. The results show that both Krasnovishnevoye and Malinovoe lakes were near neutral (pH 7.88 and 7.63, correspondingly) and belonged to the bicarbonate-chlorine sodium type. The content of bicarbonate and sulfate (percentage of TDS) in Lake Malinovoe was 5 and 2 times higher, correspondingly, while magnesium content in Lake Krasnovishnevoye was thrice as high. The amount of phosphates in Lake Krasnovishnevoye was almost 2.5 orders of magnitude lower than in Lake Malinovoe. Close to the moment of sampling in 2015 the area had been experiencing rains that led to slight decrease of salinity and increase in pH of Lake Krasnovishnevoye in comparison with 2003 (2003: TDS=307 g/L, pH 7.41; 2015: TDS= 297.3 g/L, pH 7.88).
|
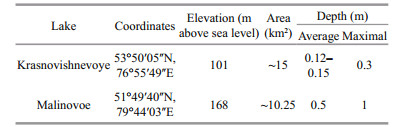
The studies of the bottom sediments showed that two mineral associations (terrigenous and evaporitic) strongly dominated in the lakes (Table 3). In Lake Krasnovishnevoye, quartz was the major type of mineral; NaCl, plagioclase, potash feldspar and others were also detected. Ash content was 96%. In Lake Malinovoe aragonite was dominant while NaCl, quartz, plagioclase, calcite and others were minor components. Ash content was 80%. Based on the mineral composition and morphology of grains terrigenous and evaporitic fractions were identified.
Both Krasnovishnevoye and Malinovoe lakes had a prominent similarity in phytoplankton composition but markedly differed in its abundance. The green alga Dunaliella salina Teod. formed mono-dominant plankton of Lake Krasnovishnyovoye reaching 230×106 cells/L, 85.6 g/m3 in 2003, and 3.01×106 cells/L, 1.24 g/m3 in 2015. Two green algae, D. salina (2.02–2.36)×106 cells/L; 0.45–0.53 g/m3) and Oocystis cf. submarina Lagerh.(1.65–2.36 cells/L; 0.07 g/m3) constituted nearly all the summer phytoplankton in Lake Malinovoe in 2015. Its abundance varied within (3.06–4.72)×106 cells/L, 0.54–0.61 g/m3.
3.4 ZooplanktonZooplankton was not found in Lake Krasnovishnevoye neither in 2003 nor in 2015. Zooplankton of Lake Malinovoe was represented exclusively by Artemia sp. Only females (with egg sacks and with exuvius as well as sex pubertal with soft tegument) were found in the samples. The amount was not high (90–120 ind./m3). The size of the individuals was 9.1–9.3 mm.
3.5 Anoxygenic phototrophic bacteriaThere was no visible accumulation of APB or cyanobacterial mats on the bottom or on the salt crust of the lakes Krasnovishnevoye and Malinovoe.
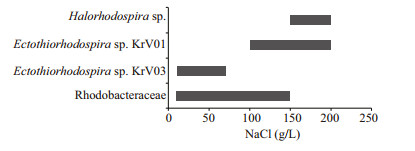
No growth of APB occurred in the enrichment cultures at pH lower than 7.5 and higher than 9.5, showing that the APB from Lake Krasnovishnevoye are alkalitolerant or alkaliphilic. APB were found over the entire salinity range (Fig. 3). Purple sulfur bacteria of the family Ectothiorhodospiraceae of two main morphotypes dominated: the first one at the NaCl concentration range of 100 to 200 g/L (KrV01: curved rods to short spirals, 0.4–0.6 by 1–2.5 μm), the second at 10–70 g/L NaCl (KrV03: curved rods or short spirals to cocci, 0.8–1.5 by 1–3 μm). These two morphotypes represented 90% of ABP in the corresponding salinities. Sphaeroidene-containing Rhodobacteraceae and aerobic bacteriochlorophyll-a containing bacteria Rhodobaca sp. (mostly at 10–150 g/L of NaCl), and Halorhodospira sp. (150– 200 g/L of NaCl) appeared to a much lesser extent. Only few members of Chromatiaceae sp. were detected in 50–200 g/L of NaCl.

|
| Fig.2 SEM images of the minerals a. Lake Krasnovishevoye: individual crystals of halite (1) with a thin crust of the plate astrakhanite crystals (2), bar=700 μm (300 μm on the insert); b. Lake Malinovoe: individual crystals of halite (1) with a thin crust consisting of the tangled fibrous astrakhanite aggregates (2), bar=1 mm; c. Lake Krasnovishevoye: bottom sediment consisting of plagioclase chip (1), quartz grain (2), fibrous illite aggregate (3), plate crystals of mica (4), calcite crystals (5), bar=20 μm; d. Lake Malinovoe: bottom sediment consisting of aragonite crystals aggregates (1), bar=10 μm. |
|
| Fig.3 Distribution of APB from Lake Krasnovishnevoye along the salinity range |
During the study of the physiology of the isolates it was found that, surprisingly, KrV01 (a strain isolated from a 150 g/L NaCl enrichment and representing the dominant morphotype in the high salinity enrichments) appeared to have its NaCl optimum at 1–3%, while the other strain KrV03, isolated from a 10-g/L NaCl enrichment and representing the dominant morphotype at the lower salinities, had its NaCl optimum at 5%– 10% and had a higher salt tolerance (Table 4). Both strains were alkaliphilic with a narrow pH optimum of 8–9. The cells of both strains grew under anoxic conditions in the light with a number of organic carbon sources (acetate, succinate, pyruvate etc.). Photoautotrophic growth occurred with sulfide, thiosulfate and elemental sulfur as electron donors.
|
According to 16S rRNA analysis, both isolates from Lake Krasnovishnevoye had 99% similarity with Ectothiorhodospira variabilis.
The APB from Lake Malinovoe were represented by purple sulfur bacteria Halorhodospira sp. in low numbers (2×103 cells/mL).
4 DISCUSSIONThe anionic composition of the lakes of Baraba steppe is dominated by bicarbonate (HCO3-) > chloride (Cl-) > sulfate (SO42-); the cationic composition: sodium (Na+) > magnesium (Mg2+) > calcium (Ca2+), with potassium (K+) as a minor fraction. The waters of the Baraba group of lakes are alkaline (pH 8.1–10) and mainly belong to the bicarbonate magnesiumsodium or bicarbonate-sodium type. The majority of lakes are freshwater with a salinity of 0.2–0.6 g/L, but the brackish lakes (1–3 g/L of TDS) and saline (3.3 g/L of TDS) lakes also occur. The anionic and cationic pattern of the waters of the Kulunda steppe lakes is mostly the same as that of the Baraba steppe. The waters belong to bicarbonate-chlorine sodium type that is characteristic for the lakes with the high total salinity, while in the less saline lakes the waters belong to bicarbonate magnesium-sodium type. The total salt concentration varies strongly from brackish waters to brine (2.1–396 g/L) with pH 7.63–10.6 (Savchenko, 1997; Kompantseva et al., 2010; Sorokin et al., 2011; Strakhovenko et al., 2017).
According to the results obtained in this work (Table 2), Lake Malinovoe can be classified as a typical saline neutral Kulunda lake, while the chemical composition of water of Lake Krasnovishnevoye is more similar to the saline neutral lakes of Kulunda steppe than to the neighbor Baraba steppe lakes. The mineral associations in the lakes were similar (Table 3, Fig. 2). The majority of terrigenous minerals in both of the investigated lakes are brought by wind. The depression of Lake Malinovoe is situated on the border of a band of conifers and thus is partially protected from dust storms coming from the Kazakhstan steppe. The aeolic fraction was therefore more pronounced in Lake Krasnovishnevoye. Evaporite associations in the lakes were also different. The presence of significant amounts of aragonite in the bottom sediments of Lake Malinovoe may indicate the processes of biogenic mineral formation (Rivadeneyra et al., 1998; Lepot et al., 2014). A broader diversity of minerals in the bottom sediments of Lake Malinovoe can also be a result of other seasonal changes in hydrodynamic, physicochemical and temperature regime of evaporite formation. The presence of pyrite framboids in the bottom sediments obviously points to biogeochemical processes occurring on the bottom sediment /water boundary (Popa et al., 2004).
The dominance of the cyanobacterium Aphanocapsa sp. or Dunaliella cf. viridis Teod., or D. salina with the cyanobacterium Aphanothece stagnina (Spreng.) A. Braun in Rabenh. was earlier reported for summer phytoplankton of Lake Malinovoe (Woronichin, 1929, 1934; Vesnina et al., 2005). The record of D. viridis from this lake seems to be unreliable because this species is less tolerant to the high insolation intensity (Oren, 2014) which is typical for the lakes studied. In addition, the names of the lakes ("Malinovoe" means crimson color and "Krasnovishnevoye" for red-cherry) point towards to characteristic water coloring due to outbreaks of redpigmented D. salina or halobacteria but not of greenpigmented D. viridis. A keystone role of D. salina can be suggested for the studied lakes. It is a well-known phenomenon for ecosystems of hypersaline water bodies (Hammer, 1986; Oren, 2014). In contrast to the phytoplankton of Tanatar lakes comprised by Dunaliella as well as by cyanobacteria and diatoms (Samylina et al., 2014), in Lake Malinovoe oxygenic phototrophs are represented almost only by green algae (D. salina and Oocystis cf. submarina Lagerh.).
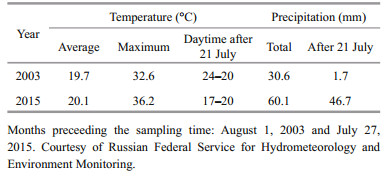
The cell number of phytoplankton in Lake Krasnovishnevoye in 2003 and 2015 reached 230×106 cells/Land 3.01×106 cells/L, correspondingly. The difference in the phytoplankton quantity can be a result of extremely unstable water regime due to the small lake depth (12–15 cm) and an uneven precipitation distribution during the month of July (the month preceding the sampling time, August 1, 2003 and July 27, 2015) (Table 5). In 2003, in spite of the high mineralization in Lake Krasnovishnevoye, the stable weather conditions (absence of rains and high temperature) favoured the bloom of phytoplankton. In 2015, apparently, a rapid decrease of water salinity from almost 300 g/L to 170 g/L during the rainfall (Table 5) together with lower temperatures caused the drop in the phytoplankton quantity, and it probably did not have time to recover.

|
Artemia sp. is present as the sole zooplankton species in Lake Malinovoe but is absent in Lake Krasnovishnevoye. The general features of both lakes were similar except for the phosphorus concentration (Table 2). The soluble phosphorus deficiency is characteristic for all the area of South Western Siberia (Moiseenko et al., 2013), and there is no anthropogenic impact on Lake Krasnovishnevoye in particular. Nevertheless, it does not seem to limit the phytoplankton as the food source for Artemia. Apparently, the reason for the absence of Artemia in Lake Krasnovishnevoye is the dynamics of the physicochemical parameters. Heavy rain prior to the sampling date in 2015 (Table 5) induced the intensive freshening of the shallow Lake Krasnovishnevoye and the increase of water level by 4 cm. After the subsequent evaporation during 1–2 sunny days immediately prior to the sampling date the mineralization regained the value of 297 g/L (Tables 2, 5). Although Artemia is tolerant to a wide range of water mineralization, the rapid fluctuations of salts concentration in short time periods may be unfavorable for its growth and development.
Obviously, Artemia sp. from Lake Malinovoe makes up a parthenogenetic population similar to those found earlier in the lakes of Novosibirsk region (Shadrin and Anufriieva, 2012). Nevertheless, additional research is needed for the detailed classification of the organism.
As no cyanobacterial mats or accumulations of APB were found in Lake Krasnovishnevoye, the share of APB in the phototrophic community of the lake is likely to be small. In some of the Kulunda lakes, the rates of anoxygenic photosynthesis reached 34% of total primary production. However, activity of anoxygenic photosynthesis was observed only in the alkaline lakes where visual presence of APB was detected, while no anoxygenic photosynthesis was registered in the saline near-neutral and alkaline lakes Lomovoe (TDS 280 g/L, pH 8.1), Kochkovoe (TDS 240 g/L, pH 8.5), Pechatnoe (TDS 200 g/L, pH 9.5) (Kompantseva et al., 2009). The features of culturable diversity of APB from Lake Krasnovishnevoye (Fig. 3) were similar to the community pattern of APB from the Kulunda lakes, saline neutral lakes in particular (Kompantseva et al., 2010): the low diversity of species composition, the dominance of representatives of the Ectothiorhodospiraceae along with low numbers of Chromatiaceae, and the absence of green sulfur bacteria. The conditions in Lake Malinovoe were close to those in other saline lakes of Kulunda (Lomovoe in particular), and APB composition consisting in the halophilic members of Ectothiorhodospiraceae (Halorhodospira sp.) was also similar (Kompantseva et al., 2010).
The phototrophic microbial communities and the mineral components of the lakes are tightly interconnected with each other. The transformation of aluminosilicate minerals and volcanic ash by nonsulfur purple bacteria (Naimark et al., 2009; Kompantseva et al., 2011; Kompantseva et al., 2013), transformation of chemical and mineralogical composition of hydrocarbonate thermal waters (Tatarinov et al., 2005), and the impact of bacteria on the biogenic mineral formation in Antharctic lakes (Lepot et al., 2014) are well known examples of this interaction. The formation of aragonite crystals and pyrite framboids in the sediments of Lake Malinovoe can also indicate the involvement of bacteria in the process of mineral formation.
5 CONCLUSIONThe present study is the first attempt to determine the hydrochemical, mineralogical, and biological features of Baraba Lake Krasnovishnevoye and to compare them to the data obtained from saline neutral Kulunda Lake Malinovoe. The mineral associations and water chemistry in Lake Krasnovishnevoye resembled those in hypersaline lakes of Kulunda steppe (Malinovoe in particular). The terrigenous fraction represented basically by quartz, feldspars, mica was similar in both lakes while the evaporitic fraction with prevalence of halite slightly differs in complex salts. The aeolian components in Lake Krasnovishnevoye were more pronounced.
The studied hydrobiota in Lake Malinovoe was represented by primary producers (mainly unicellular algae, APB were also detected) and Artemia sp., that are characteristic also for other salt and soda lakes of Kulunda steppe. The culturable diversity of APB from Lake Krasnovishnevoye showed the same patterns as in the lakes of Kulunda, but their role in the process of primary production in Lake Krasnovishnevoye, as well as the studies of decomposers and microorganisms driving the cycles of nitrogen and sulfur is subject to further investigations. The absence of Artemia in Lake Krasnovishnevoye was obviously the result of the sharp fluctuations of the physicochemical parameters in the shallow water body.
6 DATA AVAILABILITY STATEMENTSequence data that support the findings of this study have been deposited in GenBank with the accession numbers MH537738 and MH537737.
7 ACKNOWLEDGEMENTAuthors thank L. M. Kipriyanova for sampling in 2003 and Prof. V. M. Gorlenko for fruitful discussion.
Biebl H, Pfennig N. 1981. Isolation of members of the family rhodospirillaceae. In: Starr M P, Stolp H, Trüper H G et al eds. The Prok aryotes. Springer, Berlin, Heidelberg. p.267-273. http://link.springer.com/10.1007/978-3-662-13187-9_14
|
Bryanskaya A V, Malup T K, Lazareva E V, Taran O P, Rozanov A S, Efimov V M, Peltek S E. 2016. The role of environmental factors for the composition of microbial communities of saline lakes in the Novosibirsk region (Russia). BMC Microbiol, 16: S4.
DOI:10.1186/s12866-015-0618-y |
Foti M J, Sorokin D Y, Zacharova E E, Pimenov N V, Kuenen J G, Muyzer G. 2008. Bacterial diversity and activity along a salinity gradient in soda lakes of the Kulunda Steppe (Altai, Russia). Extremophiles, 12(1): 133-145.
DOI:10.1007/s00792-007-0117-7 |
Gorlenko V M, Bryantseva I A, Rabold S, Tourova T P, Rubtsova D, Smirnova E, Thiel V, Imhoff J F. 2009. Ectothiorhodospira variabilis sp. nov., an alkaliphilic and halophilic purple sulfur bacterium from soda lakes. International Journal of Systematic and Evolutionary Microbiology, 59(Pt 4): 658-664.
|
Grant W D. 2006. Alkaline Environments and Biodiversity. EOLSS Publishers Oxford, UK. p.1-19.
|
Hammer U T. 1986. Saline Lake Ecosystems of the World (Monographiae Biologicae). Dr. W. Junk Publishers, Dordrecht. 616p.
|
Herbert R A. 1990. Methods for enumerating microorganisms and determining biomass in natural environments. In: Grigorova R, Norris J R eds. Methods in Microbiology. Academic Press, London. p.1-40. http://www.sciencedirect.com/science/article/pii/S0580951708702381
|
Imhoff J F. 2005. Order Chromatiales. In: Garrity G M, Editorin-Chief. Bergey's Manual of Systematic Bacteriology: The Gammaproteobacteria. 2nd edn. Springer-Verlag, New York, Berlin, Heidelberg. p.1-59.
|
Komárek J, Fott B. 1983. Chlorophyceae (grünalgen). Ordnung: chlorococcales. In: Huber-Pestalozzi G ed. Das Phytoplankton des Süßwassers. Systematik und Biologie. Schweizerbart'sche, Stuttgart.
|
Kompantseva E I, Komova A V, Rusanov I I, Pimenov N V, Sorokin D Y. 2009. Primary production of organic matter and phototrophic communities in the soda lakes of the Kulunda steppe (Altai krai). Microbiology, 78(5): 643-649.
DOI:10.1134/S002626170905018X |
Kompantseva E I, Komova A V, Sorokin D Y. 2010. Communities of anoxygenic phototrophic bacteria in the saline soda lakes of the Kulunda Steppe (Altai Krai). Microbiology, 79(1): 89-95.
DOI:10.1134/S0026261710010121 |
Kompantseva E I, Naimark E B, Boeva N M, Zhukhlistov A P, Novikov V M, Nikitina N S. 2013. Interaction of anoxygenic phototrophic bacteria Rhodopseudomonas sp. with kaolinite. Microbiology, 82(3): 316-326.
DOI:10.1134/S0026261713030077 |
Kompantseva E I, Naimark E B, Komova A V, Nikitina N S. 2011. Interaction of the haloalkaliphilic purple bacteria Rhodovulum steppense with aluminosilicate minerals. Microbiology, 80(5): 650-656.
DOI:10.1134/S0026261711050067 |
Landsat Look Viewer. https://landsatlook.usgs.gov/, accessed 2 May 2018.
|
Lepot K, Compère P, Gérard E, Namsaraev Z, Verleyen E, Tavernier I, Hodgson D A, Vyverman W, Gilbert B, Wilmotte A, Javaux E J. 2014. Organic and mineral imprints in fossil photosynthetic mats of an East Antarctic lake. Geobiology, 12(5): 424-450.
DOI:10.1111/gbi.2014.12.issue-5 |
Melack J M, Kilham P. 1974. Photosynthetic rates of phytoplankton in East African alkaline, saline lakes. Limnol. Oceanogr., 19(5): 743-755.
DOI:10.4319/lo.1974.19.5.0743 |
Moiseenko T I, Gashkina N A, Dinu M I, Kremleva T A, Khoroshavin V Y. 2013. Aquatic geochemistry of small lakes: effects of environment changes. Geochemistry International, 51(13): 1 031-1 148.
DOI:10.1134/S0016702913130028 |
Naimark E B, Kompantseva E I, Komova A V. 2009. Interaction between anoxygenic phototrophic bacteria of the genus Rhodovulum and volcanic ash. Microbiology, 78(6): 747-756.
DOI:10.1134/S0026261709060125 |
Namsaraev Z, Samylina O, Sukhacheva M, Borisenko G, Sorokin D Y, Tourova T. 2018. Effect of salinity on diazotrophic activity and microbial composition of phototrophic communities from Bitter-1 soda lake (Kulunda Steppe, Russia). Extremophiles, 22(4): 651-663.
DOI:10.1007/s00792-018-1026-7 |
Oren A. 2014. The ecology of Dunaliella in high-salt environments. Journal of Biological Research-Thessaloniki, 21: 23.
DOI:10.1186/s40709-014-0023-y |
Pfennig N, Lippert K D. 1966. über das Vitamin B12-Bedürfnis phototropher Schwefelbakterien. Archiv für Mikrobiologie, 55(3): 245-256.
DOI:10.1007/BF00410246 |
Popa R, Kinkle B K, Badescu A. 2004. Pyrite framboids as biomarkers for iron-sulfur systems. Geomicrobiology Journal, 21(3): 193-206.
DOI:10.1080/01490450490275497 |
Rivadeneyra M A, Delgado G, Ramos-Cormenzana A, Delgado R. 1998. Biomineralization of carbonates by Halomonas eurihalina in solid and liquid media with different salinities: crystal formation sequence. Research in Microbiology, 149(4): 277-287.
DOI:10.1016/S0923-2508(98)80303-3 |
Samylina O S, Sapozhnikov F V, Gainanova O Y, Ryabova A V, Nikitin M A, Sorokin D Y. 2014. Algo-bacterial communities of the Kulunda steppe (Altai region, Russia) soda lakes. Microbiology, 83(6): 849-860.
DOI:10.1134/S0026261714060162 |
Savchenko N V. 1997. The Lakes of Southern Plains of Western Siberia. Publishers SBRAS, Novosibirsk. 297p.
|
Shadrin N, Anufriieva E. 2012. Review of the biogeography of Artemia Leach, 1819 (Crustacea: Anostraca) in Russia. International Journal of Artemia Biology, 2(1): 51-61.
|
Sorokin D Y, Abbas B, Geleijnse M, Pimenov N V, Sukhacheva M V, Van Loosdrecht M C. 2015. Methanogenesis at extremely haloalkaline conditions in the soda lakes of Kulunda Steppe (Altai, Russia). FEMS Microbiology Ecology, 91(4): fiv016.
|
Sorokin D Y, Kuenen J G, Muyzer G. 2011. The microbial sulfur cycle at extremely haloalkaline conditions of soda lakes. Frontiers Microbiology, 2: 44.
|
Sorokin D Y, Rusanov I I, Pimenov N V, Tourova T P, Abbas B, Muyzer G. 2010. Sulfidogenesis under extremely haloalkaline conditions in soda lakes of Kulunda Steppe (Altai, Russia). FEMS Microbiology Ecology, 73(2): 278-290.
|
State Standard PND F 14.2.99-97. 1997. Quantitative chemical analysis of water. A titrimetric method of measurement of hydrocarbonates mass concentration in the natural samples. Moscow, State Committee of the Russian Federation for Environmental Protection. 20p. (in Russian)
|
Strakhovenko V D, Ovdina E A, Ermolaeva N I, Zarubina E Yu, Saltykov A V. 2017. The peculiarities of chemical composition of lake waters and forming there various sapropels. In: Proceedings of Russian Scientific Conference with Foreign Participation "Water Resources: The New Challenges and the Ways of Solution". Sochi, p.426-431. (in Russian)
|
Strakhovenko V D, Shcherbov B L, Malikova I N, Vosel Y S. 2010. The regularities of distribution of radionuclides and reare-earth elements in bottom sediments of Siberian lakes. Russian Geology and Geophysics, 51(11): 1167-1 178.
DOI:10.1016/j.rgg.2010.10.002 |
Strakhovenko V D. 2011. Geochemistry of Bottom Sediments in the Small Continental Lakes of Siberia. IGM SORAN, Novosibirsk. 32p. (in Russian)
|
Tatarinov A V, Yalovik L I, Namsaraev Z, Plyusnin A M, Konstantinova K K, Zhmodik S M. 2005. Role of bacterial mats in the formation of rocks and ore minerals in travertines of nitric hydrothermal springs in the Baikal Rift zone. Doklady Earth Sciences, 403(6): 939-942.
|
Tourova T P, Slobodova N V, Bumazhkin B K, Sukhacheva M V, Sorokin D Y. 2014. Diversity of diazotrophs in the sediments of saline and soda lakes analyzed with the use of the nifH gene as a molecular marker. Microbiology, 83(5): 634-647.
DOI:10.1134/S002626171404016X |
Tsalolikhin S Y. 1995. Key to Freshwater Invertebrates of Russia. Zoological Institute RAS, Saint-Petersburg.
|
Vesnina L V, Mitrofanova E Yu, Lisitsyna T O. 2005. Plankton of salted lakes of the territory of a closed runoff (the South of West Siberia, Russia). Sibirskiy Ekologicheskiy Zhurnal, 2: 221-233.
|
Woronichin N N. 1929. Materials towards study of algal vegetation in lakes of Kulunda Steppe. Izvestiya Glavnogo Botanicheskogo Sada SSSR, 28(1-2): 12-40.
|
Woronichin N N. 1934. Contributions to the knowledge of biology of saline water bodies from Kulunda Steppe. Trudy SOPS AN SSSR, Ser. Sibirskaya, 8: 177-183.
|
Zavarzin G A. 1993. Epicontinental soda lakes as probable relict biotopes of terrestrial biota formation. Mikrobiologiya, 62(6): 789-800.
|
 2018, Vol. 36
2018, Vol. 36