Institute of Oceanology, Chinese Academy of Sciences
Article Information
- Elena V. ANUFRIIEVA
- How can saline and hypersaline lakes contribute to aquaculture development? A review
- Chinese Journal of Oceanology and Limnology, 36(6): 2002-2009
- http://dx.doi.org/10.1007/s00343-018-7306-3
Article History
- Received Nov. 1, 2017
- accepted in principle Jan. 23, 2018
- accepted for publication Jun. 13, 2018
The current world population of 7.6 billion is expected to reach 8.6 billion in 2030, 9.8 billion in 2050 and 11.2 billion in 2100 (UN, 2017). The growing population faces many challenges. Currently, a considerable part of the world's population is experiencing scarcity of freshwater and nutrition and these problems are among the most important challenges of the developing humanity (FAO et al., 2015; WWAP, 2015). Currently agriculture supplies about 97.5% of the total food mass production, wild fisheries more than 1.5%, and aquaculture about 1% (FAO et al., 2015). Usage of fresh water by agriculture, municipalities and industries has risen from less than 580 km3 in 1900 to more than 3 900 km3 in 2010, and agriculture consumes approximately 70% of the total freshwater consumption in the world (FAO, 2015). Human population growth leads to an increase in the demand for food and irrigation water for agriculture by 60% to 65%. In 1995–2015, the production growth rate for crops was 2.4% per year, for livestock it was 2.6%, and for aquaculture it was 13.7% (FAO, 2009, 2014, 2016). Total aquaculture production increased from about 20 million tonnes in 1995 to 73.8 million tonnes in 2014.
To meet the food demand of humankind there is only one way: to increase aquaculture production (Duarte et al., 2009; FAO, 2016). On a global scale, over the past decade, there has been no significant increase in marine aquaculture production (FAO, 2016). World aquaculture production in inland waters increased from 24.5 million tonnes in 2004 to 47.1 million tonnes in 2014 (Fig. 1), and its share in total aquaculture production increased from 58% to 64% (FAO, 2009, 2014, 2016). Inland aquaculture has the fastest growth in fresh waters, and this contributes to the eutrophication of freshwater bodies (Findlay et al., 2009; FAO, 2016), thus reducing the reserve for drinking water and other human needs. The question must be asked whether it will be possible for humanity to meet the food demands of its growing population by aquaculture development without damage to fresh water resources. The increase in freshwater aquaculture impacts on the problem of the increasing demand for fresh water. A way to overcome this is to develop aquaculture in saline lakes and cultivation of salt-tolerant crops around them without compromising the supply of drinking water (Rozema and Flowers, 2008; Shadrin and Anufriieva, 2016).
The use of aquaculture in inland saline waters may have great potential to be explored by the development of aquaculture trials in different inland saline waters. The first stage of such aquaculture development should be based on "learning by doing" (Holling, 1978). Conducting such experiments requires the integration of efforts by stakeholders who would include not only scientists, but also the users of lakes, aquaculture specialists, and environmental managers. The main goal of this paper is to stimulate cooperation between those stakeholders to explore the potential of saline and hypersaline lakes to contribute to aquaculture development.
2 THE WORLD DIVERSITY OF SALINE LAKES AND THEIR AQUACULTURAL POTENTIALContinental waters consist of both fresh and saline waters (defined as containing > 3.0–3.5 g/L salt) (Williams, 1996; Zheng, 2014); their total global volumes are similar with 126 thousand km3 of freshwater lakes and rivers and 104 thousand km3 saline lakes (Hammer, 1986; Williams, 1996). Salt lakes are more varied in physicochemical features than freshwater lakes including ion composition. Some of them are closer to marine (thalassic) waters and others have very different dominant salts, for example soda and sulfate lakes (Zheng, 2014). In the lakes of varying salt composition there is often different faunal structure (Belmonte et al., 2012). Environmental filtering by salt composition, biotic interactions and stochastic factors together determine such faunal differences (Poff, 1997; Tolonen et al., 2018). Often it is difficult to separate the role of a single factor. Data from the long-term study (2000– 2017) of the Crimean salt lakes shows that abundant species such as the anostracan Artemia spp. (Shadrin and Anufriieva, 2017), copepod Arctodiaptomus salinus (Daday, 1885) (Anufriieva and Shadrin, 2014a) and chironomid larvae Baeotendipes noctivagus (Kieffer, 1911) (Shadrin et al., 2017), which are possibilities for aquaculture, may reach high abundance both in marine and sulfate lakes under salinity range 30–250 g/L. Salt composition is an important factor but there is not enough published data in literature to comment on the role of salt composition in this paper. Saline lakes are widespread on all continents (Zheng, 2014); they played and still play an important role in the history of civilization (Adshead, 1992; Kurlansky, 2002). Salt is one of the essential products for humans; its extraction from salt lakes began more than 5 000 years ago and was widespread in the ancient civilizations of Asia, Europe, Africa and America with the oldest human settlements growing beside those lakes and becoming villages then towns (Lovejoy, 1986; Adshead, 1992; Williams, 1999; Kurlansky, 2002). The use of the therapeutic and cosmetic capabilities of the mud of salt lakes was known in the ancient world and currently lake muds are widely used for therapeutics and cosmetics in different countries (Ma'or et al., 1996; Du et al., 2006; Baschini et al., 2012). Aquaculture is a new potential way to use saline lakes which can generate profit in addition to salt production and mud use. There are good examples of such integrated sustainable use of saline lakes (Zheng, 2014; Shadrin et al., 2016; Shaalan et al., 2018). Most saline lakes are in arid areas (Zheng, 2014) and often people inhabiting those areas have very low incomes; aquaculture development in those areas may improve their economic situation (Jia et al., 2015a; Kavembe et al., 2016).
Due to global climate change and anthropogenic activity, salinity is increasing in natural and artificial water bodies in various regions of the world (Williams, 2001; Shadrin et al., 2015). There is also a high potential for aquaculture development using saline ground water (Shearer et al., 1997). Both freshwater and saline waters are used in continental aquaculture with successful examples of aquaculture in saline lakes in different countries (Jain et al., 2003; Kolkovski, 2011; De Los Rios-Escalante and Salgado, 2012; Jia et al., 2015a, b; Shaalan et al., 2018). However, saline waters are still used for this purpose in a lesser degree than freshwaters. This is not due to the low biological productivity of saline and hypersaline waters, since many of them are among the most productive aquatic ecosystems on the planet (Hammer, 1986; Shadrin et al., 2015). The reason is mainly the inertia of our thinking and activity patterns, traditions and habits. We need to change current aquaculture development priorities. Development of aquaculture in saline lakes must be among our main priorities (Shadrin and Anufriieva, 2016). Freshwater usage conflicts are common in arid countries or places where freshwater is pumped from groundwater or aquifers (WWAP, 2015). A way to overcome this is to develop aquaculture in saline lakes without compromising the supply of drinking water. Saline lake aquaculture may be one of the key elements of a new approach to environmental management of arid/ semi-arid zones (Zheng, 2014; Jia et al., 2015a, b; Shadrin and Anufriieva, 2016).
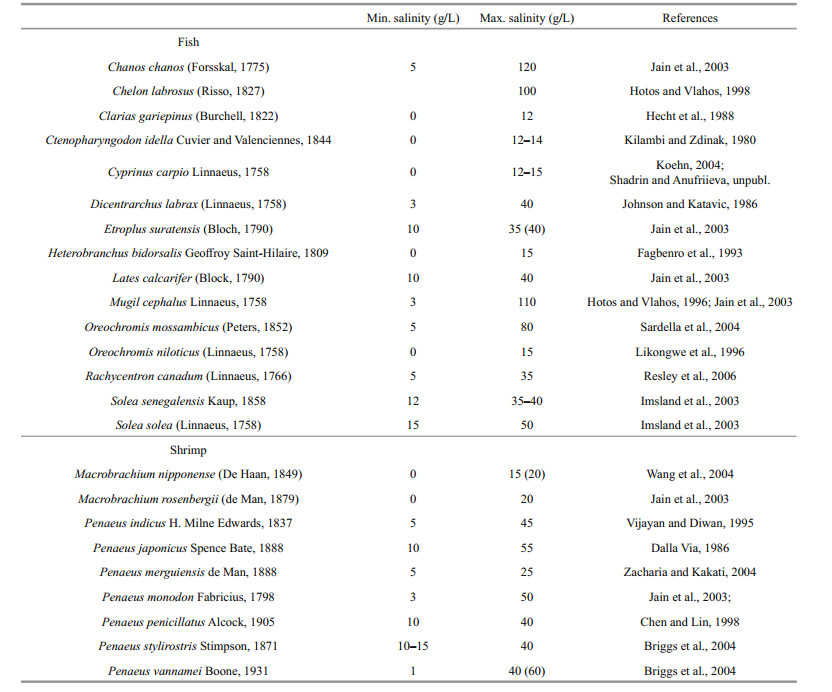
For the development of aquaculture in saline lakes we must take into account the diversity of such lakes (Zheng, 2014). This diversity could provide different opportunities for aquaculture, which need to be considered. The conditions for the existence of organisms and the potential for aquaculture development in water bodies are determined by a complex interlacing of factors (total salinity, salt composition, pH, oxygen regimes, mixing regimes, temperature regimes, etc.). Classification of saline lakes according to their chemical composition (proportions between main ions) has been described many times (Kurnakov et al., 1936; Zheng, 2014; Schagerl and Renaut, 2016). For example, in Crimea, there are two chemical types of saline lakes—chloride and sulphate-chloride (Kurnakov et al., 1936) and there are differences in their biotic composition (Belmonte et al., 2012). The salinity regime of a lake is one of most important factors determining what kind of aquaculture can be developed in it (Williams, 1998; Khlebovich and Aladin, 2010; Shadrin, 2017). There are several classifications of water bodies based on salinity, which have minor differences (Williams, 1996; Zheng, 2014) and they will not be discussed here. Averaging these classification systems, all saline waters may be conditionally divided into hyposaline/ brakish water (3–17 g/L), mesosaline/marine salinity (17–35 g/L) and hypersaline water/brine (> 35 g/L). The yearly average salinity and also seasonal and interannual salinity fluctuations need to be taken into account to plan a strategy of aquaculture development in a lake. Every species is adapted to live in a certain salinity range (Khlebovich and Aladin, 2010). The halotolerance of the potential aquaculture fauna needs to be known to develop a strategy and technology for culture. Halotolerance of hydrobionts was mostly studied in chloride (chloride-sulphate) waters. Many commercially valuable fish and shrimp species can be successfully cultivated in saline / hypersaline lakes. The ranges of their halotolerance in chloride (chloridesulphate) waters are given in Table 1. Currently only a small number of these are used in inland aquaculture. Not all species suitable for cultivation in saline lakes are listed in the table. Currently it is impossible to compile a full list because the aquaculture potential and halotolerance range for most species have not been yet studied. Based on the analysis of the literature and own field studies the author concludes that many species of fish and shrimp that are considered as freshwater organisms can be successfully cultivated in the salinity range 3–17 g/L. A salinity range of 17– 35 g/L is a good prospect for cultivation of many species of marine origin. Inland Lake Qarun (Egypt) is a good example of this (Shadrin et al., 2016). However, there is a scarcity of studies in this direction. Only few shrimp and fish species can grow at salinity higher than 50 g/L; more such species may be discovered during further experiments. As an example, 71 fish species are found in the East Africa soda lakes, six of these species live at salinity up to 40 g/L, and three live at salinity up to 100 g/L (Kavembe et al., 2016).
|
Monoculture of fish and shrimps negatively impacts the natural environment and biodiversity (Xie and Yu, 2007; FAO, 2016). Polyculture of organisms of different trophic levels is a way to overcome or mitigate this problem. Now 'Integrated Multi-Trophic Aquaculture' (IMTA) is recognized as one of the main topics in aquaculture development (Alexander et al., 2015; Guerra-García et al., 2017). The IMTAapproach allows simultaneously cultivating different species of two or more trophic levels in same pond / lake while the waste of one species is consumed as food by other species. Growth of cultivated animals can be co-limited by the supply of different biochemical essential components (PUFA, vitamins, etc.); a biochemically diverse food organism composition should promote their consumer growth (Marzetz et al., 2017). As an example, consumption of different osmolytes synthesized by different bacteria and algae can significantly increase halotolerance of animals-osmoconformers (Anufriieva, 2015; Shadrin et al., 2017). In addition to fish and shrimps many eukaryotic organisms of different taxa can be cultivated in saline/hypersaline lakes/ponds to be used in agriculture and aquaculture, different industries and as food supplements, etc. In the wide salinity range up to more than 50–200 g/L, they include different species of filamentous green algae, Crustacea (Amphipoda, Isopoda, Mysida), and larvae of Diptera. For example, among amphipods there are halotolerant species which can live at high salinity (Bayly, 1972; Britton and Johnson, 1987; the author's own data): Gammarus aequicauda Martynov, 1931 at more than 150 g/L, G. mucronatus Say, 1818 at 50 g/L, Parhyale inyacka K. H. Barnard, 1916 at 90 g/L, Grandidierella bonnieroides Stephensen, 1947 and Ericthonius punctatus Bate, 1857 at 80 g/L. A review on a possible use of halophilic bacteria was published previously (Oren, 2010).
3 LARVICULTURE OF COMMERCIAL FISH AND SHRIMP AND A PROBLEM OF LIVE FOOD ORGANISMSSuccessful development of aquaculture depends on healthy cultured stock of commercial fish and shrimps. A sustainable healthy stock of fish/shrimp can be only maintained while using live food for the cultured fish larvae, fry and fingerlings (Evjemo et al., 2003; Das et al., 2012). Artificial feed solely cannot meet all the elements required for their normal development and growth. Food organisms can be cultivated in a wide salinity range from 0 to 250 g/L (He et al., 2001; Kolkovski, 2011; Anufriieva, 2015; Shadrin et al., 2017). The focus of this paper is mostly on the highest salinity where commercial fish and shrimp cannot be grown, in hypersaline waters with salinity higher than 100 g/L.
When talking about food organisms, people generally remember only Artemia spp. (Anostraca), which are a key element of our current aquaculture practice (Das et al., 2012). There is a large potential for development of Artemia cultivation in lakes and ponds in arid areas (Kolkovski, 2011; De los Rios-Escalante and Salgado, 2012; Jia et al., 2015a, b). As well as for Artemia spp. there are many other crustacean species that can be grown in hypersaline waters. At least 26 copepod species around the world can live at salinity above 100 g/L; 12 species at salinity higher than 200 g/L (Anufriieva, 2015). The most halotolerant among them are: Cletocamptus retrogressus Shmankevitch, 1875 (Harpacticoida) at salinity 350 g/L, Arctodiaptomus salinus (Calanoida) at 300 g/L, and Meridiecyclops baylyi Fiers, 2001 (Cyclopoida) at 240 g/L (Anufriieva, 2015). Copepods have excellent nutritional value; they are rich in highly unsaturated fatty acids (HUFA) with a high omega-3 fatty acid (docosahexaenoid acid; DHA) content (Evjemo et al., 2003; Das et al., 2012). A. salinus also has a high content of the valuable carotenoid astaxanthin and is now harvested in some saline lakes (Anufriieva and Shadrin, 2014b). Some copepod species can reach a high abundance in hypersaline waters and produce thermo- and halotolerant resting eggs that can easily be cultivated (Jiménez-Melero et al., 2013; Anufriieva and Shadrin, 2014a; Annabi-Trabelsi et al., 2018). Among the Cladocera, Moina salina Daday 1888 is the most widespread halotolerant species that can live at salinity up to 220 g/L (Amarouayache et al., 2012). Currently some copepod, cladoceran and rotifera species are successfully cultivated in saline waters (He at al., 2001; Evjemo et al., 2003; Das et al., 2012; Reyes et al., 2017).
Crustacean groups of larger size such as Amphipoda, Mysida and Isopoda also have a potential for cultivation in saline waters as food organisms for fish and shrimp; they are cultivated in some countries (Herrera et al., 2009; Schmalenbach et al., 2009; Guerra-García et al., 2017). Chironomidae larvae are valuable food for fry, fingerlings and adults of fish and shrimps; cultivation of chironomid larvae was started in the USSR in 1940–1950 (Ivleva, 1969), and today it is developing in several countries and gives high profit (Shaw and Mark, 1980; Sahandi, 2011). The widespread Baeotendipes noctivagus, occurring in waters with salinity up to 280 g/L, and Australian Tanytarsus barbitarsis Freeman, 1961, reaching high abundance at salinity up to 177 g/L, are the most halotolerant chironomids in the world (Shadrin et al., 2017).
All the above relates to chloride and/or chloridesulphate waters. Animal species richness in soda lakes is lower than in other saline lakes but animal abundance may reach the highest values (Mengistou, 2016; Schagerl and Burian, 2016). Some invertebrate species may be successfully cultivated as food organisms. For example, animals occurring in the East Africa soda lakes are: 1. Rotifera (Brachionus plicatilis (Müller, 1786), Brachionus dimidiatus Bryce, 1931) at salinity up to 100 and 70 g/L respectively; 2. Copepoda (Lovenula africana (Daday, 1910), Afrocyclops gibsoni (Brady, 1904), Eucyclops serrulatus (Fischer, 1851), Mesocyclops sp.) at salinity up to 40–50 g/L; 3. Cladocera (Diaphanosoma excisum G.O. Sars, 1885, Moina spp.) at salinity up to 40 g/L; 4. Chironomidae larvae (12 species) at salinity above 40–60 g/L; 5. Diptera (Ephydra spp.) at salinity up to 160 g/L (Mengistou, 2016; Schagerl and Burian, 2016).
4 CONCLUSIONTo develop aquaculture in inland saline waters we need to take into account that there is no one best strategy, design and technology. The optimum strategy and design should be based on the environmentally and economic conditions prevailing in the locality. While learning from experiments a sustainable, environmental friendly and ecosystembased aquaculture must be developed. Sustainable and ecosystem-based aquaculture is independent of the biomass produced by natural ecosystems and includes representatives of all trophic levels in reasonable proportions. This means that aquaculture farms / production facilities must be diverse and include algae, small invertebrates, shrimp and fish.
Every lake / water body has its own individuality, with its abiotic and ecosystem peculiarities. To develop a sustainable strategy of aquaculture development we need to take the lake's individuality into account. Lakes are dynamic, and can transit from one state to another, shifting from one aquacultural potential to a different one (Shadrin, 2014). This must be taken into account for the long-term strategic development of an aquaculture farm/production facility in every lake. To do this we need deeper and wider knowledge of saline lakes, their ecosystems and objects of cultivation. The keystone for successful development of aquaculture in saline inland waters must be a strong social order and effective cooperation between science and lake user stakeholders.
5 ACKNOWLEDGMENTThe author thanks three anonymous reviewers for valuable comments and Bindy Datson (Australia) for her help to improve English.
Adshead S A M. 1992. Salt and Civilization. Macmillan, London. 417p.
|
Alexander K A, Potts T P, Freeman S, Israel D, Johansen J, Kletou D, Meland M, Pecorino D, Rebours C, Shorten M, Angel D L. 2015. The implications of aquaculture policy and regulation for the development of integrated multitrophic aquaculture in Europe. Aquaculture, 443: 16-23.
DOI:10.1016/j.aquaculture.2015.03.005 |
Amarouayache M, Derbal F, Kara M H. 2012. Note on the carcinological fauna associated with Artemia salina (Branchiopoda, Anostraca) from Sebkha Ez-Zemoul (northeast Algeria). Crustaceana, 85(2): 129-137.
DOI:10.1163/156854012X623728 |
Annabi-Trabelsi N, Rebai R K, Ali M, Subrahmanyam M N V, Belmonte G, Ayadi H. 2018. Egg production and hatching success of Paracartia grani (Copepoda, Calanoida, Acartiidae) in two hypersaline ponds of a Tunisian Solar Saltern. J. Sea Res., 134: 1-9.
DOI:10.1016/j.seares.2017.12.002 |
Anufriieva E V, Shadrin N V. 2014a. Factors determining the average body size of geographically separated Arctodiaptomus salinus (Daday, 1885) populations. Zool. Res., 35(2): 132-141.
|
Anufriieva E V, Shadrin N V. 2014b. Arctodiaptomus salinus (Daday, 1885) (Calanoida, Copepoda) in saline water bodies of the Crimea. Morskoi Ecologicheskii Zhurnal, 13(3): 5-11.
|
Anufriieva E V. 2015. Do copepods inhabit hypersaline waters worldwide? A short review and discussion. Chin. J. Oceanol. Limnol., 33(6): 1 354-1 361.
DOI:10.1007/s00343-014-4385-7 |
Baschini M, Piovano E L, López-Galindo A, Dietrich D, Setti M. 2012. Muds and salts from Laguna Mar Chiquita (or Mar de Ansenuza), Córdoba, Argentina: natural materials with potential therapeutic uses. Anales de Hidrología Médica, 5(2): 123-129.
|
Bayly I A E. 1972. Salinity tolerance and osmotic behavior of animals in athalassic saline and marine hypersaline waters. Ann. Rev. Ecol. Syst., 3(1): 233-268.
DOI:10.1146/annurev.es.03.110172.001313 |
Belmonte G, Moscatello S, Batogova E A, Pavlovskaya T, Shadrin N V, Litvinchuk L F. 2012. Fauna of hypersaline lakes of the Crimea (Ukraine). Thalassia Salentina, 34: 11-24.
|
Briggs M, Funge-Smith S, Subasinghe R, Phillips M. 2004. Introductions and movement of Penaeus vannamei and Penaeus stylirostris in Asia and the Pacific. Food and Agriculture Organization of the United Nations, Regional Office for Asia and the Pacific. RAP Publication, Bangkok.
|
Britton R H, Johnson A R. 1987. An ecological account of a Mediterranean salina: the Salin de Giraud, Camargue (S. France). Biol. Conserv., 42(3): 185-230.
DOI:10.1016/0006-3207(87)90133-9 |
Chen J C, Lin J N. 1998. Osmotic concentration and tissue water of Penaeus chinensis juveniles reared at different salinity and temperature levels. Aquaculture, 164(1-4): 173-181.
DOI:10.1016/S0044-8486(98)00185-9 |
Dalla Via G J. 1986. Salinity responses of the juvenile penaeid shrimp Penaeus japonicus: Ⅰ. oxygen consumption and estimations of productivity. Aquaculture, 55(4): 297-306.
DOI:10.1016/0044-8486(86)90170-5 |
Das P, Mandal S C, Bhagabati S K, Akhtar M S, Singh S K. 2012. Important live food organisms and their role in aquaculture. Front. Aquacult., 5: 69-86.
|
De Los Rios-Escalante P, Salgado I. 2012. Artemia (Crustacea, Anostraca) in Chile: a review of basic and applied biology. Lat. Am. J. Aquat. Res., 40(3): 487-496.
DOI:10.3856/vol40-issue3-fulltext-1 |
Du S Y, Sun P X, Li J, Huo W H. 2006. Study on mineral black mud from Shanxi Yuncheng salt lake. Detergent & Cosmetics, 29(3): 18-21.
(in Chinese with English abstract) |
Duarte C M, Holmer M, Olsen Y, Soto D, Marba N, Guiu J, Black K, Karakassis I. 2009. Will the oceans help feed humanity?. BioScience, 59(11): 967-976.
DOI:10.1525/bio.2009.59.11.8 |
Evjemo J O, Reitan K I, Olsen Y. 2003. Copepods as live food organisms in the larval rearing of halibut larvae (Hippoglossus hippoglossus L.) with special emphasis on the nutritional value. Aquaculture, 227(1-4): 191-210.
DOI:10.1016/S0044-8486(03)00503-9 |
Fagbenro O A, Adedire C O, Owoseeni E A, Ayotunde E O. 1993. Studies on the biology and aquaculture potential of feral catfish Heterobranchus bidorsalis (Geoffroy St. Hilaire 1809) (Clariidae). Trop. Zool., 6(1): 67-79.
DOI:10.1080/03946975.1993.10539209 |
FAO, IFAD, WFP. 2015. The state of food insecurity in the world 2015. Meeting the 2015 International Hunger Targets: Taking Stock of Uneven Progress. Food and Agricultural Organization of the United Nations, Rome. 62p.
|
FAO. 2009. The state of world fisheries and aquaculture 2008. FAO Fisheries and Aquaculture Department. Food and Agricultural Organization of the United Nations, Rome. 196p.
|
FAO. 2014. The state of world fisheries and aquaculture 2014. Opportunities and challenges. Food and Agricultural Organization of the United Nations, Rome. 243p.
|
FAO. 2015. FAO statistical pocketbook 2015: World food and agriculture. Food and Agriculture Organization of the United Nations, Rome. 236p.
|
FAO. 2016. The state of world fisheries and aquaculture 2016. Contributing to food security and nutrition for all. Food and Agricultural Organization of the United Nations, Rome. 190p.
|
Findlay D L, Podemski C L, Kasian S E M. 2009. Aquaculture impacts on the algal and bacterial communities in a small boreal forest lake. Can. J. Fish. Aquat. Sci., 66(11): 1 936-1 948.
DOI:10.1139/F09-121 |
Guerra-García J M, Baeza-Rojano E, Jiménez-Prada P, Calero-Cano S, Cervera J L. 2017. Trends in aquaculture today. Marine amphipods as alternative resource. Biodivers. J., 8(2): 395-398.
|
Hammer U T. 1986. Saline Lake Ecosystems of the World. Springer, Dordrecht. 616p.
|
He Z H, Qin J G, Wang Y, Jiang H, Wen Z. 2001. Biology of Moina mongolica (Moinidae, Cladocera) and perspective as live food for marine fish larvae: review. Hydrobiologia, 457(1-3): 25-37.
|
Hecht T, Vys W, Britz P J. 1988. The culture of the African sharptooth catfish (Clarias gariepinus) in Southern Africa. South African National Scientific Programmes. Report No. 153. Available at: http://www.nda.agric.za/doaDev/fisheries/03_areasofwork/Aquaculture/AquaAndEnvironment/C%20%201gariepinus%20final%20BRBA.pdf. Accessed on 2017-09-19.
|
Herrera A, Gómez M, Molina L, Otero F, Packard T. 2009. Advances in rearing techniques and analysis of nutritional quality of two mysids species present in Gran Canaria. Eur. Aquacult. Soc. Spec. Publ., 38: 171-174.
|
Holling C S. 1978. Adaptive Environmental Assessment and Management. Wiley, London. 377p.
|
Hotos G N, Vlahos N. 1998. Salinity tolerance of Mugil cephalus and Chelon labrosus (Pisces: Mugilidae) fry in experimental conditions. Aquaculture, 167(3-4): 329-338.
DOI:10.1016/S0044-8486(98)00314-7 |
Imsland A K, Foss A, Conceiçao L E C, Dinis M T, Delbare D, Schram E, Rema P, White P. 2003. A review of the culture potential of Solea solea and S. senegalensis. Rev. Fish Biol. Fish., 13(4): 379-408.
DOI:10.1007/s11160-004-1632-6 |
Ivleva I V. 1969. Mass Cultivation of Invertebrates: Biology and Methods. Keter Press, Jerusalem. 158p.
|
Jain AK, Mukherjee S C, Ayyappan S. 2003. Inland Salinewater Aquaculture: Research and Development. Indian Council of Agricultural Research, Mumbai.. 58p.
|
Jia Q X, Anufriieva E, Liu X F, Kong F J, Shadrin N. 2015a. Intentional introduction of Artemia sinica (Anostraca) in the high-altitude Tibetan Lake Dangxiong Co: the new population and consequences for the environment and for humans. Chin. J. Oceanol. Limnol., 33(6): 1 451-1 460.
DOI:10.1007/s00343-015-4371-8 |
Jia Q X, Liu S S, Lv G J, Liu X F, Zhang Y S, Zheng M P. 2015b. Artemia population characteristics in different conditions, and environmental carrying capacity in small saline lakes in the Inner Mongolian desert. Acta Ecol. Sin., 35(10): 3 364-3 375.
|
Jiménez-Melero R, Gilbert J D, Guerrero F. 2013. Secondary production of Arctodiaptomus salinus in a shallow saline pond: comparison of methods. Mar. Ecol. Prog. Ser., 483: 103-116.
DOI:10.3354/meps10377 |
Johnson D W, Katavic I. 1986. Survival and growth of sea bass (Dicentrarchus labrax) larvae as influenced by temperature, salinity, and delayed initial feeding. Aquaculture, 52(1): 11-19.
DOI:10.1016/0044-8486(86)90102-X |
Kavembe G D, Meyer A, Wood C M. 2016. Fish populations in East African saline lakes. In: Schagerl M ed. Soda La kes of East Africa. Springer, Cham. p.227-257.
|
Khlebovich V V, Aladin N V. 2010. The salinity factor in animal life. Herald Russ. Acad. Sci., 80(3): 299-304.
DOI:10.1134/S1019331610030172 |
Kilambi R V, Zdinak A. 1980. The effects of acclimation on the salinity tolerance of grass carp, Ctenopharyngodon idella (Cuv. and Val.). J. Fish Biol., 16(2): 171-175.
DOI:10.1111/jfb.1980.16.issue-2 |
Koehn J D. 2004. Carp (Cyprinus carpio) as a powerful invader in Australian waterways. Freshwater Biol., 49(7): 882-894.
DOI:10.1111/fwb.2004.49.issue-7 |
Kolkovski S. 2011. An overview on desert aquaculture in Australia. In: Crespi V, Lovatelli A eds. Aquaculture in Desert and Arid Lands: Development Constraints and Opportunities. FAO, Rome. p.39-60.
|
Kurlansky M. 2002. Salt: A World History. Walker, New York. 484p..
|
Kurnakov N S, Kuznetsov V G, Dzens-Lytovsky A I, Ravich M I. 1936. The Crimean salt lakes. AN USSR Publ., Moscow. 278p. (in Russian).
|
Likongwe J S, Stecko T D, Stauffer J R, Carline R F. 1996. Combined effects of water temperature and salinity on growth and feed utilization of juvenile Nile tilapia Oreochromis niloticus (Linneaus). Aquaculture, 146(1-2): 37-46.
DOI:10.1016/S0044-8486(96)01360-9 |
Lovejoy P E. 1986. Salt of the Desert Sun: A History of Salt Production and Trade in the Central Sudan. Cambridge University Press, Cambridge. 368p.
|
Ma'or Z E, Magdassi S, Efron D, Yehuda S. 1996. Dead Sea mineral-based cosmetics-facts and illusions. Isr. J. Med. Sci., 32Suppl: S28-S35.
|
Marzetz V, Koussoroplis A-M, Martin-Creuzburg D, Striebel M, Wacker A. 2017. Linking primary producer diversity and food quality effects on herbivores: A biochemical perspective. Sci. Rep., 7: 11 035.
DOI:10.1038/s41598-017-11183-3 |
Mengistou S. 2016. Invertebrates of East African soda lakes. In: Schagerl M ed. Soda lakes of East Africa. Springer, Cham. p.205-226.
|
Oren A. 2010. Industrial and environmental applications of halophilic microorganisms. Environ. Technol., 31(8-9): 825-834.
DOI:10.1080/09593330903370026 |
Poff N L. 1997. Landscape filters and species traits: towards mechanistic understanding and prediction in stream ecology. J. North Am. Benthol. Soc., 16(2): 391-409.
DOI:10.2307/1468026 |
Resley M J, Webb K A, Holt G J. 2006. Growth and survival of juvenile cobia, Rachycentron canadum, at different salinities in a recirculating aquaculture system. Aquaculture, 253(1-4): 398-407.
DOI:10.1016/j.aquaculture.2005.08.023 |
Reyes J C R, Monteón C J L, Urreta H C, Dosta M D C M, de Oca G A R M. 2017. Population growth and protein and energy content of Proales similis (Rotifera: Monogononta) reared at different salinities. Turk. J. Fish. Aquat. Sci., 17(4): 767-775.
|
Rozema J, Flowers T. 2008. Crops for a salinized world. Science, 322(5907): 1 478-1 480.
DOI:10.1126/science.1168572 |
Sahandi J. 2011. Natural food production for aquaculture: cultivation and nutrition of Chironomid larvae (Insecta, Diptera). AES Bioflux, 3(3): 268-271.
|
Sardella B A, Matey V, Cooper J, Gonzalez R J, Brauner C J. 2004. Physiological, biochemical and morphological indicators of osmoregulatory stress in 'California' Mozambique tilapia (Oreochromis mossambicus × O. urolepis hornorum) exposed to hypersaline water. J. Exp. Biol., 207(8): 1 399-1 413.
DOI:10.1242/jeb.00895 |
Schagerl M, Burian A. 2016. The ecology of African soda lakes: driven by variable and extreme conditions. In: Schagerl M ed. Soda Lakes of East Africa. Springer, Cham. p.295-320.
|
Schagerl M, Renaut R W. 2016. Dipping into the soda lakes of East Africa. In: Schagerl M ed. Soda Lakes of East Africa. Springer, Cham. p.3-24.
|
Schmalenbach I, Buchholz F, Franke H D, Saborowski R. 2009. Improvement of rearing conditions for juvenile lobsters (Homarus gammarus) by co-culturing with juvenile isopods (Idotea emarginata). Aquaculture, 289(3-4): 297-303.
DOI:10.1016/j.aquaculture.2009.01.017 |
Shaalan M, El-Mahdy M, Saleh M, El-Matbouli M. 2018. Aquaculture in Egypt: insights on the current trends and future perspectives for sustainable development. Rev. Fish. Sci. Aquacult., 26(1): 99-110.
DOI:10.1080/23308249.2017.1358696 |
Shadrin N V, Anufriieva E V, Belyakov V P, Bazhora A I. 2017. Chironomidae larvae in hypersaline waters of the Crimea: diversity, distribution, abundance and production. The European Zoological Journal, 84(1): 61-72.
DOI:10.1080/11250003.2016.1273974 |
Shadrin N V, Anufriieva E V. 2016. Why do we need to pay more attention to study the saline lakes?. SILNews, 61: 10-11.
|
Shadrin N V, Anufriieva E V. 2017. Size polymorphism and fluctuating asymmetry of Artemia (Branchiopoda: Anostraca) populations from the Crimea. Journal of Siberian Federal University Biology, 10(1): 114-126.
DOI:10.17516/1997-1389-0012 |
Shadrin N V, El-Shabrawy G M, Anufriieva E V, Goher M E, Ragab E. 2016. Long-term changes of physicochemical parameters and benthos in Lake Qarun (Egypt): can we make a correct forecast of ecosystem future?. Knowl. Manag. Aquat. Ecosyst., 417: 18.
DOI:10.1051/kmae/2016005 |
Shadrin N V. 2017. Peculiarities of structure, functioning and dynamics of the salt lake. In: Zheng M, Deng T, Oren A eds. Introduction to Salt Lake Sciences. Science Press, Beijing. p.179-186.
|
Shadrin N, Zheng M P, Oren A. 2015. Past, present and future of saline lakes: research for global sustainable development. Chin. J. Oceanol. Limnol., 33(6): 1 349-1 353.
DOI:10.1007/s00343-015-5157-8 |
Shadrin N. 2014. Alternative states of saline lake ecosystems and development of salinology. Acta Geol. Sin. (English Edition), 88(S1): 434-435.
DOI:10.1111/1755-6724.12280_9 |
Shaw P C, Mark K K. 1980. Chironomid farming—a means of recycling farm manure and potentially reducing water pollution in Hong Kong. Aquaculture, 21(2): 155-163.
DOI:10.1016/0044-8486(80)90024-1 |
Shearer T R, Wagstaff S J, Calow R, Stewart J A, Muir J F, Haylor G S, Brooks A C. 1997. The potential for aquaculture using saline groundwater. BGS Technical Report WC/97/58. British Geological Survey, Keyworth, Nottingham. 235p.
|
Tolonen K E, Leinonen K, Erkinaro J, Heino J. 2018. Ecological uniqueness of macroinvertebrate communities in high-latitude streams is a consequence of deterministic environmental filtering processes. Aquat. Ecol., 52(1): 17-33.
DOI:10.1007/s10452-017-9642-3 |
United Nations, Department of Economic and Social Affairs, Population Division. 2017. World Population Prospects: The 2017 Revision-Key Findings and Advance Tables. Working Paper No. ESA/P/WP/248. 53p.
|
Vijayan K K, Diwan A D. 1995. Influence of temperature, salinity, pH and light on molting and growth in the Indian white prawn Penaeus indicus (Crustacea: Decapoda: Penaeidae) under laboratory conditions. Asian Fish. Sci., 8: 63-72.
|
Wang W N, Wang A L, Bao L, Wang J P, Liu Y, Sun R Y. 2004. Changes of protein-bound and free amino acids in the muscle of the freshwater prawn Macrobrachium nipponense in different salinities. Aquaculture, 233(1-4): 561-571.
DOI:10.1016/j.aquaculture.2003.09.042 |
Williams E. 1999. The ethnoarchaeology of salt production at lake Cuitzeo, Michoacán, México. Latin American Antiquity, 10(4): 400-414.
DOI:10.2307/971964 |
Williams W D. 1996. The largest, highest and lowest lakes of the world: saline lakes. Internationale Vereinigung für Theoretische und Angewandte Limnologie: Verhandlungen, 26(1): 61-79.
|
Williams W D. 1998. Salinity as a determinant of the structure of biological communities in salt lakes. Hydrobiologia, 381(1-3): 191-201.
|
Williams W D. 2001. Anthropogenic salinisation of inland waters. Hydrobiologia, 466(1-3): 329-337.
|
WWAP (United Nations World Water Assessment Programme). 2015. The United Nations World Water Development Report 2015: Water for a Sustainable World. Paris, UNESCO. 123p. http://unesdoc.unesco.org/images/0023/002318/231823E.pdf.
|
Xie B, Yu K J. 2007. Shrimp farming in China: operating characteristics, environmental impact and perspectives. Ocean Coast. Manag., 50(7): 538-550.
DOI:10.1016/j.ocecoaman.2007.02.006 |
Zacharia S, Kakati V S. 2004. Optimal salinity and temperature for early developmental stages of Penaeus merguiensis De man. Aquaculture, 232(1-4): 373-382.
DOI:10.1016/S0044-8486(03)00538-6 |
Zheng M. 2014. Saline Lakes and Salt Basin Deposits in China. Science Press, Beijing. 321p.
|
 2018, Vol. 36
2018, Vol. 36



