Institute of Oceanology, Chinese Academy of Sciences
Article Information
- Nickolai V. SHADRIN
- The alternative saline lake ecosystem states and adaptive environmental management
- Chinese Journal of Oceanology and Limnology, 36(6): 2010-2017
- http://dx.doi.org/10.1007/s00343-018-7307-2
Article History
- Received Nov. 1, 2017
- accepted in principle Jan. 4, 2018
- accepted for publication Jan. 29, 2018
Environmental risks for the life-supporting capacity of the biosphere are increasing, and the chance to meet the demands of the growing human population for food and other resources is decreasing as a result of landscape destruction and reduction of natural ecosystem resilience and productivity (Folke et al., 2005; Bindraban et al., 2013). A main reason for this is the insufficiently developed environmental management of human activities. Analyzing why people have such inappropriate environmental management humans need to recollect that we contemporaneously live and operate in two worlds—the real physical world and the virtual world of signs. The virtual world includes all our myths, traditions, motivations, goals, concepts, and models of the physical world. How people interpret the virtual world of signs determines their interaction with the physical world. Environmental management is the interface between the virtual world of signs and the environment of the physical world. Reasonableness of human activity in the world around us is determined at first by how adequately the physical world is reflected in the virtual world, or how close our concepts and paradigms are to the reality of the physical world in its variability. Integrated Water Resource Management and Integrated Lake—Watershed Management takes into account the connectivity of structures and processes and are now the main approaches to management of lakes. Nevertheless, these approaches may be based on different ecosystem concepts. Integrated management approaches must be based on adequate ecological concepts (Hilderbrand et al., 2005; Lyytimäki and Hildén, 2007; Cundill et al., 2012; Bindraban et al., 2013). The question arises: What concept is better to use for understanding and management of ecosystem dynamics? To catalyze a trans-sector discussion of this question is the aim of the paper.
In looking for answers, attention could be focused on saline lakes, which are an essential, integral and dynamic part of the biosphere and have considerable environmental, social and economic values (Hammer, 1986; Zheng, 2014; Shadrin et al., 2015a; Shadrin and Anufriieva, 2016). Saline lakes are found in every climatic zone on every continent including Antarctica. However, the majority of saline water bodies originated due to dry climates and are situated in arid and semiarid climatic zones that occupy about onethird of the Earth land (Zheng, 2001, 2014). As an example, in China (Xinjiang, Qinghai, Tibet, Ganzu, Inner Mongolia, Shanxi, Shaanxi, Ningxia, Jilin, etc.) there are more than 1 000 such lakes (Zheng, 2014). Many saline lakes are distributed in Russia from Crimea to East Siberia (Egorov and Kosmakov, 2010; Shadrin and Anufriieva, 2012; Rumyantsev et al., 2015). The total volume of all freshwater lakes on Earth is very close to same quantification of salt lakes—126 and 104 km3, respectively (Hammer, 1986). Why use salt lakes to compare ecological concepts? There are three reasons: 1. our own longterm experience in the study of saline/hypersaline lakes; 2. there are no conceptual differences in the organization and dynamics of both lake type ecosystems, although, of course, the ecosystems of saline lakes have their own characteristics (Lin et al., 2017; Shadrin, 2017). The total biodiversity and the diversity of individual taxa in salt lakes are less than in freshwater and demonstrate negative correlation with salinity and their community structure is simpler (Balushkina et al., 2009; Belmonte et al., 2012; Anufriieva, 2015); 3. multipurpose sustainable use of saline lakes is a high priority goal (Zheng, 2014; Shadrin and Anufriieva, 2016).
2 THE CONCEPT OF MULTIPLICITY OF ECOSYSTEM ALTERNATIVE STABLE STATESThe traditional assumption that every ecosystem is in a quasi-stable equilibrium fluctuating around a single point of a global equilibrium with balance of energy, matter, diversity and prevailing of smooth changes may be named the Conception of unicity of an ecosystem stable state (CUESS). In reality all ecosystems as well as every complicated system have several alternative stable states and may demonstrate a regime shift, which is a large, abrupt, and persistent change in the structure and functioning of an ecosystem (Holling, 2001; Beisner et al., 2003; Biggs et al., 2009). Existence of alternative stable states in complex systems inevitably follows from the general provisions of the theory of dissipative structures (Prigogine and Stengers, 1984) and synergetics (Haken, 1993).
Many researchers wrote that the idea that communities can be found in one of several possible alternative stable states was first proposed by R. C. Lewontin (Lewontin, 1969), and empirical evidences were found later (Beisner et al., 2003). Earlier in 1928 after the study of the Popov pond over several years, G. G. Winberg wrote that the pond community was in different alternative states in the years 1925 and 1926 (Winberg, 1928). In the 20th and 21st centuries, science accumulated data showing that communities, ecosystems and populations can exist in more than one stable state. This was shown for ecosystems of freshwater and saline lakes, rivers, forests, beaches, and seas (Dublin et al., 1990; Knowlton, 1992; Blindow et al., 1993; Scheffer, 2001; Adema et al., 2002; Dent et al., 2002; Davis et al., 2003; Zagorodnyaya et al., 2008; Petraitis et al., 2009; McGlathery et al., 2013; Shadrin, 2013). There is also the opposite opinion that observed temporal ecosystem dynamics represent variation along a continuum rather than categorically different states (Capon et al., 2015). Authors analyzed many cases, and the author supports some their particular conclusions, but not the general one. There are two main views on Nature—discrete and continuum, but the author doesn't plan to discuss these here.
Currently a new theoretical vision of an ecosystem was developed, which may be named The Concept of multiplicity of ecosystem alternative stable states (CMEASS) (Holling, 2001; Walker et al., 2004; Shadrin, 2012). In the evolution and dynamics of ecosystems there are coherent and incoherent stages (Krasilov, 1986). An ecosystem realizes a smooth adaptation to the changing environment within an existing norm of reaction during a coherent stage; destabilization of an ecosystem and its transformation through the tipping point leads into a new state during an incoherent stage. Each of the stages in turn includes two phases. The Canadian ecologist C. Holling(1973, 2001) described patterns and processes over time in ecosystem dynamics using a four-phase model—an adaptive cycle, in which an ecosystem alternates between long coherent stages of aggregation (r) and transformation (K) of resources and shorter incoherent stages that create opportunities for innovation (α and Ω) (Fig. 1). Holling wrote that the phase from Ω to α, which may be named "creative destruction", "is a period of rapid reorganization during which a novel recombination can unexpectedly seed experiments that lead to innovations in the next cycle". We need to remember that an adaptive cycle is an abstract generalization or a scientific metaphor; in reality, there is more complicated diversity of the alternations between possible states.
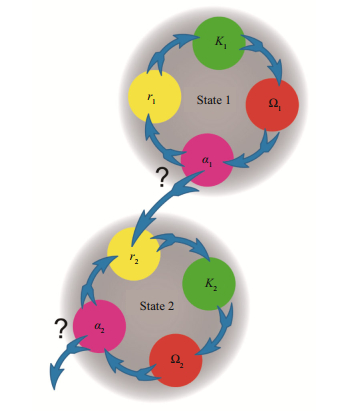
|
| Fig.1 The adaptive cycle with two stages 1. coherent, from r to K, is the slow, incremental phase of growth and accumulation, 2. incoherent, from Ω to α, is the rapid phase of reorganization leading to renewal. |
Salt lakes as all-natural complicated systems have multi-scale natures and their changes occur on different temporal and spatial scales. Adaptive cycles occur on all scales; cross-scalar dynamics couple those systems—the Principle of Panarchy (Gunderson and Holling, 2002). In the World of Hierarchy, a higher level determines the dynamics on the lower level. In the World of Panarchy systems of different levels (scales) are interlinked in determination of dynamics of each other.
The state shifts are often illustrated heuristically by the ball-in-cup model (Holling et al., 1995). A 'ball and cup' is a metaphor: an ecosystem is a ball which can move among several cups (locally stable attractors). Internal feedback mechanisms operate to keep the system in a particular cup (state). The depth of the cup demonstrates the power of these internal feedbacks. Internal causes (resource depletion, loss of adaptive capacity, microevolution events) or/and a change in an external driving factor, for example, such as climate fluctuations or human impact, can result in a sudden change of an ecosystem state when internal regulating mechanisms are overcome, and the ecosystem inevitably transits to a new state. In CUESS 'the ball' can move only inside one 'cup', but in CMEASS 'the ball' can also make jumps between 'cups'.
Every ecosystem has a critical (bifurcation) point—tipping point (TP) in its dynamics where a system may shift from one stable state to an alternative one. In a coherent stage we can use CUESS to analyze and understand the ecosystem dynamics, but in an incoherent stage we must use CMEASS to analyze system dynamics. In different stable states there are different structures of connectivity between the ecosystem elements and feedbacks; a regime shift may occur, for example, from top-down to bottom-up control (Schmitz, 2010). In ecosystem dynamics we observe a hysteresis, which is the dependence of a system output on the system history; it is not only a result of the current influence on it. Hysteresis arises because the history affects an internal system state. If a given input parameter alternately increases or decreases we observe different TP (Scheffer et al., 2001); a typical mark of hysteresis is that the output forms a loop as in Fig. 2. As an example, such hysteresis exists in saline lakes during a shift from submerged macrophytes to benthic microbial mats and in reverse (Davis et al., 2003). Ecologists and environmental managers must be interested in understanding hysteresis because it indicates that ecosystems might easily be pushed into some new unwanted configurations from which it may prove much more difficult to recover. That is why many environmental restoration projects do not reach the expected results (Suding et al., 2004; Boldgiv et al., 2005; Hilderbrand et al., 2005; Biggs et al., 2009). From a management perspective, it is critical to know when, where, and how a hysteresis may occur.
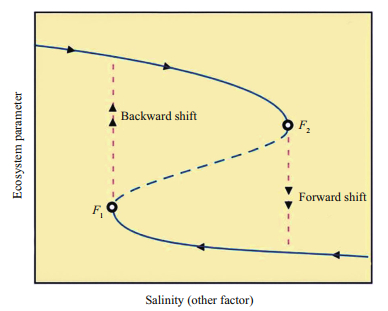
|
| Fig.2 Hysteresis loop with two different tipping points (F1 and F2) |
CMEASS seems fairly well developed and consistent now (Holling, 2001; Walker et al., 2004; Biggs et al., 2009). However, despite extensive interest in it, the effective implementation of adaptive management remains a challenge in the study of the dynamics of particular ecosystems and the practice of decision-making. What are the causes of this? The main reasons are: 1. the inertia of our thinking, and 2. the problems with identification of the discrete alternative stable states of the real aquatic ecosystems and prediction of the TPs. For identification of the alternative stable states in the dynamics of lake ecosystems different approaches may be used and criteria (correlation matrix, graphs, etc.). We need to develop effective tools for this.
3 ALTERNATIVE STATES OF SALINE LAKE ECOSYSTEMSEcosystems existing in an extreme and highly changeable environment are more responsive to climate changes and anthropogenic impacts; their alternative stable states may be designated more easily. Using data of our long-term study on the Crimean hypersaline lakes we may identify the alternative stable states in the dynamics of their ecosystems. The Crimea is the biggest peninsula in the Black Sea (nearly 26.5×103 km2). There are many saline lakes in the peninsula—fifty relatively large lakes and numerous small hypersaline water bodies (Shadrin, 2013, 2017). We conducted a multiannual integrated study of the Crimean hypersaline lakes (2000-2015); most of the long-term results are published in different papers (Prazukin et al., 2008; Balushkina et al., 2009; Belmonte et al., 2012; Anufriieva et al., 2017; Shadrin et al., 2017a, b). Our study showed that every lake can exist in different states (Zagorodnyaya et al., 2008; Shadrin, 2013, 2017; Shadrin and Anufriieva, 2013). To identify the alternative stable states, we took into account the differences in the structure of the energy input into the ecosystems.
Input of energy into the ecosystems can be caused by various groups of primary producers, which use the three mechanisms of phototrophy (bacteriorhodopsin pump, oxygenic and anoxygenic photosynthesis) and the different chemolithotrophic mechanisms. In the Crimean shallow salt lakes salinity varies widely. In some lakes during the period of our observations (2000-2015) it varied from 15 to 410 g/L. Accordingly, we observed a change in ecosystem state—transits to new states; we can allocate up to 7-9 of the observed main ecosystem stable states—different compositions of various groups of primary producers (Shadrin, 2013, 2017). More detailed information on these compositions in studied lakes was given in the published papers (Prazukin et al., 2008; Balushkina et al., 2009; Samylina et al., 2010; Shadrin, 2013; Anufriieva et al., 2017; Shadrin et al., 2017a). The most productive states are: 1. filamentous green algae Cladophora mats with primary production—up to 46 g C/(m2·d), 2. bottom biofilms: 4.1 g C/(m2·d), 3. oxygenic phytoplankton: 3.2 g C/(m2·d). Similar alternative states also exist in the Australian saline lakes (Davis et al., 2003). Alternative lake ecosystem states have different biological resources and possibilities for their use by humans. As an example, Fig. 3 shows satellite views on two different alternative states of Lake Bakalskoye, when 1. floating mats of the filamentous green alga Cladophora were the main primary producers (2000-2003), and 2. in 2004-2015 when phytoplankton played the most important role in primary production (Shadrin and Anufriieva, 2013). Changes in lake alternative states are imprinted in the bottom sediments of the Crimean hypersaline lakes (Fig. 4). Alternation of black and gray layers in the sediment cores reflects the repetitive changes of states in the past; oxidative—in years with phytoplankton dominance and reductive—under floating mats.

|
| Fig.3 Satellite views on Lake Bakalskoye in two different alternative states in June 2002: floating filamentous mats of green algae Cladophora were the main primary producers; 2013: phytoplankton played a most important role in primary production (from http://landsatlook.usgs.gov). |

|
| Fig.4 Alternation of black and gray layers in the sediment core from hypersaline lake koyashskoye (crimea) reflects the repetitive changes of states in the past: reductive (black), under floating mats and oxidative, in years with phytoplankton dominance |
From our data at first view we may conclude that salinity is the main or sole driving factor. But this is not the case. In different years in the same lake under the same salinity we found different stable states (Zagorodnyaya et al., 2008; Shadrin, 2013). No single driving factor can be identified; alien species also may push the saline lake ecosystems to transit into a new state (El-Shabrawy et al., 2015; Jia et al., 2015). A value of salinity as TP relates to the direction of salinity change (Shadrin, 2013). In different directions there are distinct ТР values (Fig. 2). Effect of hysteresis was observed; this means that ecosystems, in a certain sense, have a memory. Most species-inhabitants of hypersaline waters have resting stages (Belmonte et al., 2012; Anufriieva and Shadrin, 2014; Shadrin et al., 2015b). Such "sleeping" biodiversity is one element of the ecosystem memory. From all the above mentioned results we may conclude that the ecosystem memory and the Principle of Panarchy make it impossible to reliably forecast an ecosystem's future. If anyone thinks that he/she able to make a reliable and accurate prediction of the future ecosystem changes, he/she deceives himself, and not only himself but also those who need the forecasts to make the responsible management decisions.
4 ADAPTIVE MANAGEMENT AND DEVELOPMENT OF SALINOLOGYThe discussed theoretical issues may have a strong application in the development of salinology as "a branch of applied science focused on the study of the chemistry, physics and biology of saline lake systems. The basic task of salinology is to study and explore the features of saline lakes, to provide a scientific and technological basis for coordination between mankind and saline lakes, to promote the scientific management and rational utilization of saline lake, and to contribute to the sustainable development of saline lake agriculture, mining and tourism" (Zheng, 2001). In other words, salinology is the scientific basis of an integrated saline lake and its watershed management, a part of environmental sustainable management. Is salinology developed enough to be a scientific background of a multipurpose sustainable use of the saline lakes? It is sad to say but it makes only the first steps towards this. The main reasons are: 1. an underestimation of the integrated importance of diverse ecosystems resources and services and lack of tools to assess their integrated value (natural, social, economic); 2. lack of interdisciplinary understanding of peculiarities of saline/hypersaline unique water bodies and the connectivity of all their elements and processes, as well as absence of a well-developed theory of salinology; 3. lack of public understanding of the importance of saline lakes for sustainable development, and an obscure vision of perspectives and ways for a multipurpose sustainable use of the saline lakes in the World of Change; 4. insularity of decision making mechanisms and the weak public involvement in them; and 5. as a result of all above, the decision makers are interested only in some applied issues of salinology, not in its theoretical development; a strong social request to develop theoretical salinology is absent. The theoretical background of salinology must be sound and reflect the real peculiarities of the highly variable salt lake ecosystems; it must be based on CMEASS. CUESS and CMEASS give us the different views on possibilities, tasks and institutional organization of environmental management (Shadrin et al., 2012), including management of saline lakes. When accumulating available knowledge the 'trial-anderror' approach is used in traditional environmental management to develop a best guess 'one way' management strategy, which is changing as new information modifies the best guess. Adaptive environmental management, based on CMEASS, identifies uncertainties, and then comes up with a method to test hypotheses concerning those uncertainties (Holling, 1978; Habron, 2003). In CMEASS we use management as a tool not only to drive the system development, but as a tool to get more knowledge about the system. We need to know the cost of ignorance, while a traditional management emphasizes the need to preserve and the cost of knowledge (Holling, 1978; Habron, 2003). The main goal of traditional management based on CUESS: we need to strive to accurately forecast the responses of an ecosystem to anthropogenic impacts and to develop an optimal strategy for the management of the ecosystem, and use. However, an optimal single strategy for ecosystem management is a myth in the World of Change. The tasks of management based on CMEASS should be: to predict when an ecosystem will reach a TP, to evaluate the shift probabilities into one of the new alternative stable states, to identify the variety of new possible alternative states, and then to develop a set of possible socio: economic adaptive strategies in the new environment, and to use them flexibly (Shadrin et al., 2012). The overall goal of environmental management is to predict and, if possible, to prevent unwanted changes unless it is not possible to prevent, then be prepared for survival in new, certainly not predictable conditions. Therefore, a proper selection of strategy of management depends on a correct evaluation of the ecosystem resilience, distance to ТP, and speed of transition to it. Different stable states of the ecosystems carry out different resource potential and various possibilities for their human use. As example, in Lake Bakalskoye in one ecosystem state we may use filamentous green algae Cladophora and the crustacean Artemia, but in other years we may harvest amphipod crustaceans and grow fish (Shadrin and Anufriieva, 2013). Long-term sustainable use of salt lakes requires a variety of alternative strategies of environmental management; we must timely switch from one strategy to the alternative.
Adaptive management needs to at least maintain or create decision-making openness for new ideas and more wide involvement of different stakeholder sectors (Habron, 2003; Crépin et al., 2012; Cundill et al., 2012). Thus, adaptive environmental management should be a social as well as a scientific process and should focus to develop the new institutions and institutional strategies just as much as it should concentrate on knowledge management—scientific hypotheses and their experimental testing (Holling, 1978; Habron, 2003; Lin et al., 2015). Adaptive management is regarded as an experiment based on learning by doing. Adaptive management must be collaborative in which strategies include a dialogue between interested groups and stakeholders (on different levels from local to international), the development of complex and diverse institutions with a combination of different organizational types, designs and strategies that make easy experimentation and learning through change (Holling, 1978; Habron, 2003; Crépin et al., 2012; Costanza and Liu, 2014). A transit of an ecosystem into some new alternative state leads to the loss of some potential opportunities for human use, but is not only a loss for humans; it also creates new opportunities. As an example, in some Crimean salt lakes there are 'artemia' and 'amphipoda' years. Both crustaceans (Artemia and Amphipoda) may be harvested with high profit. People must not be afraid of the new unpredictable ecosystem states and gaining knowledge about new capabilities. Losses may be offset by new opportunities... but only if we want to see and use these new opportunities.
We need to develop the general concepts of salinology as an integrated multi-scale and multidisciplinary view on the saline lakes and their watersheds. CMEASS may serve as one of the key scientific elements in the basis of salinology, to do so we need not only to develop the theoretical issues CMEASS and its application to salt-lake management, but we must deepen our knowledge on alternative stable states of different salt lakes and the possibilities to use the resources and services for sustainable society development. Reducing and mitigating risks of catastrophic regime shift in salt-lake ecosystems as well as building adaptive capacity in social-ecological systems (salt lake-its watershed-humans) in such situations requires integration of natural and social sciences to better understand 'salt lake-its watershedhuman' system dynamics.
5 ACKNOWLEDGEMENTThe author is deeply grateful to all who assisted him in studying of salt lakes, as well as Mrs. Lilia Prazukina for the drawings made for this article and Dr. Bindy Datson (Australia) for her help to improve English of the manuscript.
Adema E B, Grootjans A P, Petersen J, Grijpstra J. 2002. Alternative stable states in a wet calcareous dune slack in the Netherlands. J. Veg. Sci., 13(1): 107-114.
DOI:10.1111/j.1654-1103.2002.tb02027.x |
Anufriieva E V, Shadrin N V, Shadrina S N. 2017. History of research on biodiversity in Crimean hypersaline waters. Arid Ecosyst., 7(1): 52-58.
DOI:10.1134/S2079096117010036 |
Anufriieva E V. 2015. Do copepods inhabit hypersaline waters worldwide? A short review and discussion. Chin. J.Oceanol. Limnol., 33(6): 1354-1361.
DOI:10.1007/s00343-014-4385-7 |
Anufriieva E, Shadrin N. 2014. Resting stages of crustaceans in the Crimean hypersaline lakes (Ukraine) and their ecological role. Acta Geol. Sin., 88(S1): 46-49.
DOI:10.1111/1755-6724.12266_3 |
Balushkina E V, Golubkov S M, Golubkov M S, Litvinchuk L F, Shadrin N V. 2009. Effect of abiotic and biotic factors on the structural and functional organization of the saline lake ecosystems in Crimea. Zh. Obshch. Biol., 70(6): 504-514.
|
Beisner B E, Haydon D T, Cuddington K. 2003. Alternative stable states in ecology. Front. Ecol. Environ., 1(7): 376-382.
DOI:10.1890/1540-9295(2003)001[0376:ASSIE]2.0.CO;2 |
Belmonte G, Moscatello S, Batogova E A, Pavlovskaya T, Shadrin N V, Litvinchuk L F. 2012. Fauna of hypersaline lakes of the Crimea (Ukraine). Thalass. Salent., 34: 11-24.
|
Biggs R, Carpenter S R, Brock W A. 2009. Turning back from the brink:detecting an impending regime shift in time to avert it. Proc. Natl. Acad. Sci. USA, 106(3): 826-831.
DOI:10.1073/pnas.0811729106 |
Bindraban P S, Brink B T, Bai Z G, Bakkenes M, Van Beek R, Van Den M, Muller C, Schaphoff S, Sonneveld B, Stoorvogel J, Temme A. 2013. Mapping global ecosystem degradation and its impacts. http://www.isric.org/sites/default/files/UNCCD_paper_SBSTA_2-2013_Bindraban_et_al_final_10-12-12.pd.
|
Blindow I, Andersson G, Hargeby A, Johansson S. 1993. Long-term pattern of alternative stable states in two shallow eutrophic lakes. Freshw. Biol., 30(1): 159-167.
DOI:10.1111/fwb.1993.30.issue-1 |
Boldgiv B, Bayartogtokh B, Bayarsaikhan U. 2005. Yellow dragon, green belt and alternative ecosystem states. Mong.J. Biol. Sci., 3(2): 49-58.
|
Capon S J, Lynch A J J, Bond N, Chessman B C, Davis J, Davidson N, Finlayson M, Gell P A, Hohnberg D, Humphrey C, Kingsford R T, Nielsen D, Thomson J R, Ward K, Nally R M. 2015. Regime shifts, thresholds and multiple stable states in freshwater ecosystems; a critical appraisal of the evidence. Sci. Total Environ., 534: 122-130.
DOI:10.1016/j.scitotenv.2015.02.045 |
Costanza R, Liu S. 2014. Ecosystem services and environmental governance:comparing China and the U. S. Asia Pac.Policy Stud., 1(1): 160-170.
DOI:10.1002/app5.v1-1 |
Crépin A S, Biggs R, Polasky S, Troell M, De Zeeuw A. 2012. Regime shifts and management. Ecol. Econ., 84: 15-22.
DOI:10.1016/j.ecolecon.2012.09.003 |
Cundill G, Cumming G S, Biggs D, Fabricius C. 2012. Soft systems thinking and social learning for adaptive management. Conserv. Biol., 26(1): 13-20.
DOI:10.1111/j.1523-1739.2011.01755.x |
Davis J A, McGuire M, Halse S A, Hamilton D, Horwitz P, McComb A J, Froend R H, Lyons M, Sim L. 2003. What happens when you add salt:predicting impacts of secondary salinisation on shallow aquatic ecosystems by using an alternative-states model. Aust. J. Bot., 51(6): 715-724.
DOI:10.1071/BT02117 |
Dent C L, Cumming G S, Carpenter S R. 2002. Multiple states in river and lake ecosystems. Philos. Trans. Roy. Soc. B, 357(1421): 635-645.
DOI:10.1098/rstb.2001.0991 |
Dublin H T, Sinclair A R E, McGlade J. 1990. Elephants and fire as causes of multiple stable states in the SerengetiMara woodlands. J. Anim. Ecol., 59(3): 1147-1164.
DOI:10.2307/5037 |
Egorov A N, Kosmakov I V. 2010. Geography and A Use of Salt Lakes. Nauka, Novosibirsk. 184p.
|
El-Shabrawy G M, Anufriieva E V, Germoush M O, Goher M E, Shadrin N V. 2015. Does salinity change determine zooplankton variability in the saline Qarun Lake (Egypt)? Chin. J. Oceanol. Limnol., 33(6): 1368-1377.
DOI:10.1007/s00343-015-4361-x |
Folke C, Fabricius C, Cundill G, Schultz L. 2005. Communities, ecosystems and livelihoods. In: Capistrano D, Samper C, Lee M, Raudsepp-Hearne C eds. Ecosystems and Human Well-being: Multiscale Assessment. Island Press, Washington. p.261-277. https://www.researchgate.net/publication/229076656_Communities_ecosystems_and_livelihoods
|
Gunderson L H, Holling C S. 2002. Panarchy:Understanding Transformations in Human and Natural Systems. Island Press, Washington. 507p.
|
Habron G. 2003. Role of adaptive management for watershed councils. Environ. Manage., 31(1): 29-41.
DOI:10.1007/s00267-002-2763-y |
Haken H. 1993. Advanced Synergetics:Instability Hierarchies of Self-Organizing Systems and Devices. SpringerVerlag, New York. 356p.
|
Hammer U T. 1986. Saline Lake Ecosystems of the World. Springer, Dordrecht. 616p.
|
Hilderbrand R H, Watts A C, Randle A M. 2005. The myths of restoration ecology. Ecol. Soc., 10(1): 19. http://www.ecologyandsociety.org/vol10/iss1/art19/.
|
Holling C S, Schindler D W, Walker B W, Roughgarden J. 1995. Biodiversity in the functioning of ecosystems: an ecological synthesis. In: Perrings C, Mäler K G, Folke C, Holling C S, Jansson B O eds. Biodiversity Loss: Economic and Ecological Issues. Cambridge University Press, Cambridge. p.44-83. http://citeseerx.ist.psu.edu/showciting?cid=1654652
|
Holling C S. 1973. Resilience and stability of ecological systems. Annu. Rev. Ecol. Syst., 4: 1-23.
DOI:10.1146/annurev.es.04.110173.000245 |
Holling C S. 1978. Adaptive Environmental Assessment and Management. Wiley, New York. 377p.
|
Holling C S. 2001. Understanding the complexity of economic, ecological, and social systems. Ecosystems, 4(5): 390-405.
DOI:10.1007/s10021-001-0101-5 |
Jia Q X, Anufriieva E, Liu X F, Kong F J, Shadrin N. 2015. Intentional introduction of Artemia sinica (Anostraca) in the high-altitude Tibetan Lake Dangxiong Co:the new population and consequences for the environment and for humans. Chin. J. Oceanol. Limnol., 33(6): 1451-1460.
DOI:10.1007/s00343-015-4371-8 |
Knowlton N. 1992. Thresholds and multiple stable states in coral reef community dynamics. Integr. Comp. Biol., 32(6): 674-682.
|
Krasilov V A. 1986. Unsolved Problems in the Theory of Evolution. DVNTs AN SSSR, Vladivostok. 139p.
|
Lewontin R C. 1969. The meaning of stability. In: Woodwell G M, Smith H H eds. Diversity and Stability in Ecological Systems, Brookhaven Symposia in Biology, No. 22.Brookhaven Laboratories, New York. p.13-24.
|
Lin H B, Thornton J A, Shadrin N. 2015. A watershed-based adaptive knowledge system for developing ecosystem stakeholder partnerships. Chin. J. Oceanol. Limnol., 33(6): 1476-1488.
DOI:10.1007/s00343-015-4366-5 |
Lin Q Q, Xu L, Hou J Z, Liu Z W, Jeppesen E, Han B P. 2017. Responses of trophic structure and zooplankton community to salinity and temperature in Tibetan lakes:implication for the effect of climate warming. Water Res., 124: 618-629.
DOI:10.1016/j.watres.2017.07.078 |
Lyytimäki J, Hildén M. 2007. Thresholds of sustainability:policy challenges of regime shifts in coastal areas. Sustainability:Sci. Pract. Policy, 3(2): 61-69.
DOI:10.1080/15487733.2007.11908007 |
McGlathery K J, Reidenbach M A, D'Odorico P, Fagherazzi S, Pace M L, Porter J H. 2013. Nonlinear dynamics and alternative stable states in shallow coastal systems. Oceanography, 26(3): 220-231.
DOI:10.5670/oceanog |
Petraitis P S, Methratta E T, Rhile E C, Vidargas N A, Dudgeon S R. 2009. Experimental confirmation of multiple community states in a marine ecosystem. Oecologia, 161(1): 139-148.
DOI:10.1007/s00442-009-1350-9 |
Prazukin A V, Bobkova A N, Evstigneeva I K, Tankovska I N, Shadrin N V. 2008. Structure and seasonal dynamics of the phytocomponent of the bioinert system marine hypersaline lake on cape of Chersonesus (Crimea). Morsk.Ekol. Zh., 7(1): 61-79.
|
Prigogine I, Stengers I. 1984. Order Out of Chaos. Heinemann, London. 349p.
|
Rumyantsev V A, Drabkova V G, Izmaylova A V. 2015. Lakes of the European part of Russia. Lena, St., Petersburg. 392p.
|
Samylina O S, Gerasimenko L M, Shadrin N V. 2010. Comparative characteristic of the phototroph communities from the mineral lakes of Crimea (Ukraine) and Altai region (Russia). Int. J. Algae, 12(2): 142-158.
DOI:10.1615/InterJAlgae.v12.i2 |
Scheffer M, Carpenter S, Foley J A, Folke C, Walker B. 2001. Catastrophic shifts in ecosystems. Nature, 413(6856): 591-596.
DOI:10.1038/35098000 |
Scheffer M. 2001. Alternative attractors of shallow lakes. Sci.World J., 1: 254-263.
DOI:10.1100/tsw.2001.62 |
Schmitz O J. 2010. Resolving Ecosystem Complexity. Princeton University Press, Princeton. 192p.
|
Shadrin N V, Anufriieva E V, Amat F, Eremin O Y. 2015b. Dormant stages of crustaceans as a mechanism of propagation in the extreme and unpredictable environment in the Crimean hypersaline lakes. Chin. J. Oceanol.Limnol., 33(6): 1362-1367.
DOI:10.1007/s00343-015-4363-8 |
Shadrin N V, Anufriieva E V, Belyakov V P, Bazhora A I. 2017b. Chironomidae larvae in hypersaline waters of the Crimea:diversity, distribution, abundance and production. Eur. Zool. J., 84(1): 61-72.
DOI:10.1080/11250003.2016.1273974 |
Shadrin N V, Anufriieva E V, Shadrina S N. 2017a. Brief review of phototrophs in the Crimean hypersaline lakes and lagoons:diversity, ecological role, the possibility of using. Mar. Biol. J., 2(2): 80-85.
DOI:10.21072/mbj.2017.02.2.08 |
Shadrin N V, Anufriieva E V. 2012. Review of the biogeography of the genus Artemia Leach, 1819 (Crustacea, Anostraca)in Russia. Int. J. Artemia Biol., 2(1): 51-61.
|
Shadrin N V, Anufriieva E V. 2013. Climate change impact on the marine lakes and their crustaceans:the case of marine hypersaline Lake Bakalskoye (Ukraine). Turk. J. Fish.Aquat. Sci., 13: 603-611.
|
Shadrin N V, Anufriieva E V. 2016. Why do we need to pay more attention to study the saline lakes?. SILNews, 61: 10-11.
|
Shadrin N V, Mironov S S, Ferat T A. 2012. Interrelations between the losses of sandy beaches and biodiversity in seas:case of the Bakalskaya Spit (Crimea, Ukraine, Black sea). Turk. J. Fish. Aquat. Sci., 12: 411-415.
|
Shadrin N V. 2012. Ecosystem dynamics and evolution:multiplicity of steady states and tipping points. Necessity of new understanding. Morsk. Ekol. Zh., 11(2): 85-95.
|
Shadrin N V. 2013. Alternative stable states of lake ecosystems and critical salinities:is there a rigid connection?. Proc.Zool. Inst., (3): 214-221.
|
Shadrin N V. 2017. Peculiarities of structure, functioning and dynamics of the salt lake ecosystems. In: Zheng M, Deng T, Oren A eds. Introduction to Salt lake Sciences. Science Press, Beijing. p.179-186.
|
Shadrin N, Zheng M P, Oren A. 2015a. Past, present and future of saline lakes:research for global sustainable development. Chin. J. Oceanol. Limnol., 33(6): 1349-1353.
DOI:10.1007/s00343-015-5157-8 |
Suding K N, Gross K L, Houseman G R. 2004. Alternative states and positive feedbacks in restoration ecology. Trends Ecol. Evol., 19(1): 46-53.
DOI:10.1016/j.tree.2003.10.005 |
Walker B, Holling C S, Carpenter S R, Kinzig A. 2004.Resilience, adaptability and transformability in socialecological systems. Ecol. Soc., 9(2): 5. http://www.ecologyandsociety.org/vol9/iss2/art5/.
|
Winberg G G. 1928. Planktonolgical and physico-chemical investigations of Pond Popov. In: Skadovskyi S N ed. A Use of Methods of Physical Chemistry to Study Biology of Freshwaters. Institute of National Health, Moscow.p.352-364. (in Russian).
|
Zagorodnyaya Y A, Batogova E A, Shadrin N V. 2008. Longterm transformation of zooplankton in the hypersaline lake Bakalskoe (Crimea) under salinity fluctuations. Mar.Ecol. J., 7(4): 41-50.
|
Zheng M P. 2001. On salinology. In: Melack J, Jellison R, Herbst D B eds. Saline Lakes. Springer, Dordrecht. p.339-347.
|
Zheng M P. 2014. Saline Lakes and Salt Basin Deposits in China-Selected Works of Zheng Mianping. Science Press, Beijing. 321p.
|
 2018, Vol. 36
2018, Vol. 36


