Institute of Oceanology, Chinese Academy of Sciences
Article Information
- Igor G. PROKOPKIN, Egor S. ZADEREEV
- A model study of the effect of weather forcing on the ecology of a meromictic Siberian lake
- Chinese Journal of Oceanology and Limnology, 36(6): 2018-2032
- http://dx.doi.org/10.1007/s00343-018-7329-9
Article History
- Received Nov. 20, 2017
- accepted in principle Jan. 10, 2018
- accepted for publication Jan. 26, 2018
2 Siberian Federal University, 660079, Svobodnii av. 79, Krasnoyarsk, Russia
The effects of weather and climate on the thermal and mixing regimes and biological properties of lake ecosystems (e.g. population size, community structure, food-web dynamics, etc.) have been demonstrated in a number of studies (e.g. Straile, 2000; Jasser and Arvola, 2003; Arvola et al., 2009; Butcher at al., 2015). These effects are very often dramatic. For example, shifted dates of onset and breakdown of stratification (Liu et al., 2014), changes in the duration of the ice-free period due to the effect of elevated temperature (e.g. Weyhenmeyer et al., 1999) or the enhanced growth of cyanobacteria in warm, calm summers (e.g. Jöhnk et al., 2008; Paerl and Huisman, 2008) cause water quality problems and lead to the loss of aesthetic and recreational value of lakes.
The list of major weather parameters includes the air temperature, wind direction and strength, cloud cover, humidity and precipitation, and pressure. While different parameters have different effects on the physical, chemical, and biological properties of lakes, the major focus of many studies is on the effect of the air temperature. However, even for the temperature to forecast the effect of climate change on ecosystems and interactions among species is not an easy task because different species show unique responses to changes in environmental temperatures (Winder and Schindler, 2004).
To understand and predict responses of ecosystems to variable weather and climate, numerical lake models are often used to simulate the combined effect of different weather parameters on aquatic ecosystems (e.g. Jones et al., 2010). There are numerous examples where complex models were used to understand the effect of weather and climate on lakes. For example, for small lakes, it was shown by mathematical modelling and then observed in natural ecosystems that climate change will increase the probability of a shift from a clear to a turbid state with higher summer chlorophyll a concentration, a stronger dominance of cyanobacteria during summer, and reduced zooplankton abundance (Mooij et al., 2007). Hadley et al. (2014) used a dynamic reservoir simulation model to understand the thermal stability of Harp Lake (Canada). A one-dimensional dynamic thermal simulation model was applied to assess lake response to different climate scenarios relevant for the U.S. (Butcher et al., 2015). These and other similar studies usually focus on freshwater lakes that experience stratification during some season (e.g., summer thermal stratification).
Saline meromictic lakes differ from seasonally stratified lakes both in terms of stability of the water column and food web structure. According to a recent definition, "a meromictic lake is a lake in which a chemically different deep water (monimolimnion) remains partly or wholly unmixed with the main water mass (mixolimnion) at the circulation periods" (Zadereev et al., 2017a). Because the monimolimnion is excluded from gas exchange for years, it is usually anoxic. The change of water properties from oxic to anoxic happens within a thin water layer—the oxicanoxic interface (Boehrer and Schultze, 2008). For many meromictic lakes, seasonal thermal stratification of mixolimnion is also typical. Thus, different gradients divide the water column in a meromictic lake into several spatially separated habitats, which affect the vertical distribution of different species (Degermendzhy et al., 2010). A specific feature of meromictic lakes is the complex structure of the food web with dense layers of bacteria in the oxic-anoxic interface and autotrophic and chemotrophic primary production at different depths of the water column (Zadereev et al., 2017b).
Meromictic lakes are sensitive to weather and climate changes. Several examples are known where the stability of stratification was lost and lake mixing occurred due to the effect of weather or climate (Miller et al., 1993; Rogozin et al., 2017). Neither the complex effects of climate and weather on the stratification of meromictic lakes nor the effect of meteorological forcing on the complicated food web in meromictic lakes were explicitly studied by numerical modelling. Previously we developed a complex simulation model of the saline meromictic Lake Shira and verified it against field data (Prokopkin et al., 2010). The model was created taking into account the practices in aquatic food web modelling worked out by PCLake Model developers (Janse, 2005). The Lake Shira model was used to describe the dynamics of the lake food web under different loads of nutrients and to test several hypotheses about the effects of different factors on the vertical distribution of phytoplankton (Prokopkin et al., 2010, 2014).
In this research, we used this numerical model to estimate the response of a meromictic lake—Lake Shira—to variations in weather parameters. The aim of this research was to estimate the relative importance of weather parameters for the thermal stratification and spatial and temporal dynamics of the biomasses of the food web components compared to other factors; to reveal how different weather parameters affect stratification and key components of the food web in a meromictic lake; and to understand the causes of the observed effects.
2 MATERIAL AND METHOD 2.1 Brief description of Lake ShiraLake Shira (54°30′N, 90°12′E) is a large saline lake with a surface area of 35.9 km2 (length: 9.3 km; width: 5.3 km) and maximal and average depths of 24 m and 11.2 m, respectively. The lake was shown to be meromictic during regular observations (from 1999 to 2014) with autumnal overturn restricted to the mixolimnion (Zadereev et al., 2014). Lake Shira is covered with ice from November to mid-May. In summer, the mixolimnion is thermally stratified with the thermocline at a variable depth of 4–8 m. Salinity in summer is about 14–15 g/L in the epilimnion and about 18 g/L in the monimolimnion. During the observation period of 1999–2014, the depth of the oxic-anoxic interface in Lake Shira varied between 11 and 16 m depending on weather conditions (Zadereev et al., 2014). The oxic-anoxic interface of the lake is inhabited by bacterial community consisting of purple sulfur and heterotrophic bacteria (Degermendzhy et al., 2010).
In summer, the thermally stratified mixolimnion creates different habitats for various species. The phytoplankton diversity in Lake Shira is relatively low. From late June to September a deep-water chlorophyll maximum (up to 23 mg/L) is detected at the lower part of the thermocline at a depth of 8–12 m. This peak mainly consists of vertically segregated peaks of the cyanobacteria Lyngbya contorta and Microcystis pulverea and the green algae Dictyosphaerium tetrachotomum and Oocystis submarina var. schiriensis (Gaevsky et al., 2002).
The zooplankton is dominated by copepods (Arctodiaptomus salinus); other abundant zooplankton species are ciliates Cyclidium sp. and Strombidium sp., rotifers Brachionus plicatilis and Hexarthra sp., and amphipod Gammarus lacustris (Zotina et al., 1999). The lake is fishless in the pelagic part. Physicochemical and biological properties of the lake were described in more detail in several papers (Kopylov et al., 2002; Zadereev and Tolomeyev, 2007; Degermendzhy et al., 2010; Rogozin et al., 2017).
2.2 General information on the Lake Shira modelWe used a biochemical-hydrophysical model that had been previously developed to simulate the dynamics of the ecosystem in the pelagic part of Lake Shira during the summer season.
The hydrophysical part of the model is onedimensional and describes the hydrodynamic and thermal structure of Lake Shira. The temperature regime of the lake is determined by wind-induced mixing, solar heating, and heat exchange with the atmosphere. The hydrophysical process of turbulent mixing of water masses is based on the formula of Prandtl-Obuchov and the approximated solution for wind-forced flow (Belolipetsky and Genova, 1998). The hydrophysical algorithm uses meteorological data, including cloud cover, air temperature, vapor pressure, and wind speed and direction. To simulate the distribution of solar radiation in the water column, the model takes into account the attenuation, in accordance with the Lambert-Beer law, and light absorbing properties of water, detritus, and biological components.
The hydrophysial part of the model enables calculating the vertical profiles of temperature, salinity, density of water, and all biochemical components during the period of open water. A detailed description of the hydrophysical part of the model was presented in Belolipetsky et al. (2010).
The biochemical part of the model comprises the main components of the pelagic part of the ecosystem—dominant species of phyto- and zooplankton, bacteria, nutrients, oxygen, and hydrogen sulfide.
The biomass-dominant green algae and cyanobacteria (Zotina et al., 1999) represent phytoplankton in the model. The growth of algae and cyanobacteria is determined by the multiplicative effect of light, temperature, and nutrients. Zooplankton are represented by the calanoid copepod Arctodiaptomus salinus, a biomass-dominant species of pelagic zooplankton (Zadereev and Tolomeyev, 2007). The zooplankton in the model is divided into two age classes: young (nauplii and juvenile copepodites) and old (late copepodites and adult animals) zooplankton. Copepods consume phytoplankton and detritus (Tolomeyev, 2002). Two groups of microorganisms carry out transformation of sulfur in the model: light-dependent purple sulfur bacteria oxidize hydrogen sulfide (H2S) and sulfatereducing bacteria decompose detritus in the water column.
The following processes are calculated for biological components of the model: growth and mortality, consumption by other components (if any) and, optionally, respiration (excretion). For the phytoplankton and purple sulfur bacteria, calculations of the sedimentation rate are based on the literature data and the authors' results.
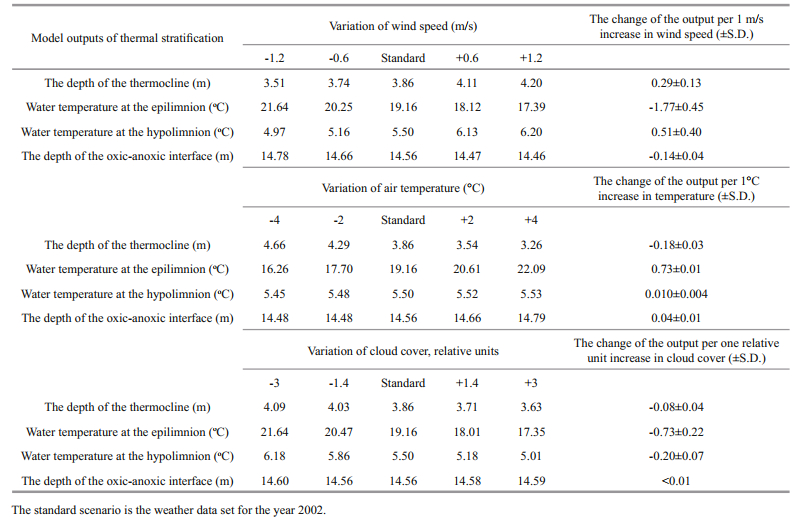
The model simulates the material cycling in the water column in units of dry weight, phosphorus, and nitrogen. Diffusion and transformation of the chemical components are also calculated. The calibration and validation of the model was performed by using field data and described in Prokopkin et al. (2010). The model demonstrates good predictions of the main thermal and mixing regimes and biomasses of food web components during the summer season (Fig. 1).
|
| Fig.1 The typical seasonal dynamics of the depth of the thermocline (white squares) and the oxic-anoxic interface (dark squares) (upper panel) and the biomasses of seston (white diamonds) (dry weight) and A. salinus (dark diamonds) (wet weight) (lower panel) Dots: field data for the parameters denoted at the graph, measured during the annual monitoring during 2007-2010. Grey areas: intervals of values for the parameters denoted at the graph, calculated with different weather sets (2002, 2007-2010) and initial conditions (deep and low oxic-anoxic interface; high and low seston biomass, high and low zooplankton biomass). |
The model was developed taking into account general principles and approaches that were used in developing similar imitation instruments and based on extensive model experience (Mooij et al., 2010). Several investigations were successfully carried out with this model (Degermendzhy et al., 2010; Prokopkin et al., 2014). The comparison of this model with other lake models was reported by Mooij et al. (2010).
2.3 Sensitivity analysis of the Lake Shira modelSensitivity analysis, which is the method to define input parameters that have the greatest influence on the model outputs (Saltelli et al., 2004), was used to compare the strength of the effect of weather forcing on the lake ecosystem with other external and internal factors affecting the ecosystem. In this study, we used the method of Morris (1991), which is the most widely used and the simplest technique of sensitivity analysis applied to models of large dimensionality. The Morris method requires a relatively low computational effort, and its result is easily interpreted, as it yields qualitative rather than exactly quantifying estimations of the relationships between inputs and outputs in the model.
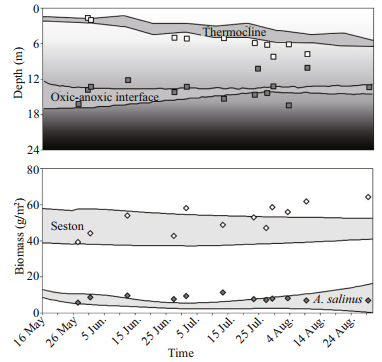
We selected 12 input parameters to perform sensitivity analysis. Input parameters were divided into three groups: 1) weather, 2) nutrient loading and 3) the initial "spring" state of the ecosystem, which depends on the preceding winter and on the previous year.
In the biochemical-hydrophysical model that we used, the hydrodynamic and thermal structures of the lake were calculated using meteorological data (Belolipetsky et al., 2010). The meteorological dataset for 2002 (hereinafter "standard year") had been previously used for model calibration (Prokopkin et al., 2010) and was chosen as the reference dataset for subsequent variations of the values of weather input time series. Each weather parameter in the dataset used for the model calculations was described by the daily dataset (from May 15 (beginning of calculation) to August 31 (end of calculation)) with several (4-6) values at regular time intervals for each 24-hour period. We varied the values of the air temperature (dTemp), cloud cover (dCloud), and wind speed (dWind) during the summer season to simulate weather changes.
Boundary conditions at the lake surface for differential equations describing the dynamics of mineral phosphorus and ammonia can be used for simulation of nutrient loading to the ecosystem. Thus, the second group of parameters consists of values of phosphorus (flowP) and nitrogen (flowN) incoming to the lake. The reference values of phosphorus and nitrogen flows used in the model are based on the field data (unpublished).
The third group of parameters determines the initial state of the ecosystem. There are seven parameters in this group describing the biomass (g/m2) of green algae (nGren), cyanobacteria (nCyano), old and young classes of zooplankton (nZooOld and nZooYoung), and detritus (nDet); the amount (g/m2) of mineral phosphorus (nP) and nitrogen (nN). The reference values for these parameters were determined from the field data (Zadereev et al., 2014).
The variation range of weather parameters was determined as follows. We carried out the analysis of weather data series in the vicinity of Lake Shira for the period of 2003-2010. The weather data series were taken from the state weather monitoring station located 5 km south-west of Lake Shira; data are available at http://meteo.infospace.ru/. The air temperature and wind speed in the weather data series were expressed in standard units of ℃ and m/s, respectively, the cloud cover was measured in relative units from 0 (clear sky) to 10 (maximal cloud cover). The cloud cover denotes daytime cloud cover, which influences the amount of solar radiation (higher cloud cover means less radiation). For each month (May, June, July and August) and each year, the average monthly values of air temperature, wind speed, and cloud cover were calculated. Next, the minimal, maximal, and average values of air temperature, wind speed, and cloud cover were determined for each month. After that, we determined the difference between the average and minimal and the average and maximal values for each month. The average value of the differences obtained was used as the range of variation. Thus, the variation ranges of weather conditions used in the model corresponded to the natural variability of these factors for the period between 2003 and 2010.
The variation ranges for phosphorus and nitrogen concentrations were selected to cover diverse possible scenarios of the nutrient load. The variation ranges for values of parameters describing the initial state of the ecosystem were set within ±50% of the reference values. With this approach, values of initial conditions did not fall outside the range of values of these parameters measured during the long-term observations at the lake (Zadereev et al., 2014). The reference values and variation ranges for all input parameters are presented in Table 1.
|
The following model outputs were chosen for the sensitivity analysis: the sum of daily (at 12:00) biomasses of cyanobacteria, green algae, two age classes of zooplankton, and purple and sulfatereducing bacteria (g dry weight/m2) and the sum of daily content of detritus (g dry weight/m2) and hydrogen sulfide (g/m2) in the water column over the whole period of calculations (from May 15 to August 31).
Sensitivity analysis calculations and processing of calculation results were based on a plan worked out in SIMLAB v.2.2 software (Simlab, 2011). The software, in accordance with the Morris method, generated the sensitivity analysis plan, in which every input parameter took several different values equally divided within its variation range. The combinations of these values, worked out by Morris's algorithm, created unique sets of values of all input parameters. As a result, the plan of calculations consisted of 104 unique sets, and each set was used to perform a model run.
Based on the sensitivity analysis for each model output for each input parameter, two sensitivity measures were determined—the mean (μ) and standard deviation (σ) of the finite distribution of elementary effects associated with each input parameter. A low value of μ means that the effect of the selected input parameter on the selected model output is low. A value of σ near zero means that the effect of the input parameter is mainly linear and is not considerably affected by other inputs. The strength of the effect of the input parameter on the output value, which was used to rank the input parameters, was determined by the length of the vector on the plane of parameters μ and σ (Janse, 2005).
2.4 Analysis of the effects of weather parameters on thermal and mixing regimes of the water columnTo analyze changes in the thermal and mixing regimes of the water column, we compared the output of the reference model run with the outputs of model runs performed with altered values of weather parameters. The reference model run was done using the meteorological data set for 2002. In other model runs, wind speed, cloud cover or air temperature in the standard year weather data set were decreased or increased by the value equal to the range of variation of a corresponding parameter (Table 1) and to its doubled value.
The following outputs were used as the characteristics of the thermal and mixing regimes of the water column: a) the depth of the thermocline calculated as the depth at which the maximal drop of temperature per incremental step (0.2 m) was observed; b) the temperature of water in the lake surface layer (epilimnion), which was determined as the average temperature in the layer from the surface to the depth of the thermocline; c) the temperature of water at a depth of 10 m (hypolimnion); d) the depth of the oxic-anoxic interface calculated as the depth at which the minimal concentrations of oxygen and hydrogen sulfide were recorded at the same time. To analyze changes in thermal and mixing regimes, the values of these characteristics were calculated as of June 1, June 15, July 1, July 15, August 1, August 15, and August 31, and then their average values were obtained.
2.5 Analysis of the effects of weather parameters on the seasonal dynamics of the food web components and on the integrated characteristics of the mixolimnion and monimolimnionTo study the effects of weather parameters on the lake ecosystem, the model was run with different weather scenarios. The standard model run was performed using the meteorological data set for 2002. In other model runs, the values of wind speed, cloud cover, and air temperature were modified as described in the previous section.
To estimate the effect of weather changes on the integrated characteristics of the mixolimnion and monimolimnion and to reveal the correlations between different components of the lake ecosystem, the following outputs were used: total (the sum of daily values from May 15 to August 31) biomass (g dry weight/m2) of phytoplankton (the sum of the biomass of cyanobacteria and green algae), zooplankton (the sum of biomasses of two zooplankton age groups), purple sulfur bacteria and detritus in the water column; total content of hydrogen sulfide in the monimolimnion. The significance of correlations between different ecosystem components was estimated using the STATISTICA 7.0 software.
3 RESULT 3.1 Sensitivity analysisInput parameters were ranked according to the degree of their influence on the values of model outputs—the main food web components of the Lake Shira ecosystem (Table 2).

|
At least one of the weather parameters was among the most influential parameters for all model outputs, both mixolimnetic and monimolimnetic ones. At the same time, it was difficult to select the weather parameter that was the most important for the ecosystem development. The strength of the effect of the same parameter on different model outputs was variable.
The ecosystem of the meromictic lake was also relatively sensitive to the initial conditions. Cyanobacteria and zooplankton were strongly influenced by the initial biomass. For all the monimolimnetic parameters, the initial biomass of detritus was the most influential parameter. However, monimolimnetic components were also sensitive to the variability of weather parameters. Even though the monimolimnion components had no direct contact with the surface layers and atmosphere, they responded to weather forcing.
The ecosystem components were not very sensitive to the effect of nutrients. The inflow of phosphorus was in the top three influential parameters only for cyanobacteria.
As this research is focused on the effects of weather parameters, the main results of the sensitivity analysis are as follows: a) within the natural range of variability of various external and internal forcing parameters, weather is an important forcing for the entire water column of the meromictic lake and b) out of the three selected weather parameters, none can be selected as the most influential one.
3.2 The effects of weather parameters on the thermal and mixing regimes of the water columnChanges of weather parameters had a strong effect on the temperature of the epilimnion but a less pronounced effect on the hypolimnion (Table 3). An increase in wind speed and cloud cover led to a pronounced decrease in water temperature in the epilimnion. An increase in air temperature led to the opposite effect - an increase in the temperature of the epilimnion. The temperature of the hypolimnion slightly increased with an increase in wind speed and decreased with an increase in cloud cover. Variations in air temperature did not affect the temperature in the hypolimnion.
Changes of weather factors also affected the depths of the thermocline and oxic-anoxic interface. The depth of the thermocline increased and the depth of the oxic-anoxic interface decreased with the increase in the wind speed. Increasing the air temperature decreased the depths of the thermocline but increased the depth of the oxic-anoxic interface. Increasing cloud cover decreased the depth of the thermocline while it had little effect on the depth of the oxicanoxic interface (Table 3).
Finally, based on our calculations, we estimated changes in the mixing regime and the temperature in the epi- and hypolimnion per 1 m/s, 1℃, or 1 unit of cloud cover increase in wind speed, air temperature, or cloud cover, respectively (Table 3).
3.3 The effect of weather parameters on the ecosystemIn order to demonstrate the interconnection between the mixing regime of the water column and food web components, we used several correlation graphs. Several correlations linked the stratification and food web components in the mixolimnion (Fig. 2) and monimolimnion (Fig. 3).
|
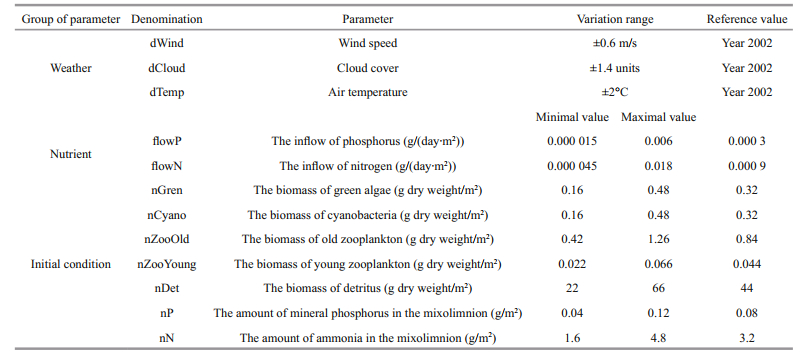
| Fig.2 Correlations between thermocline depth, water temperature in the epilimnion, and mixolimnetic food-web components |
|
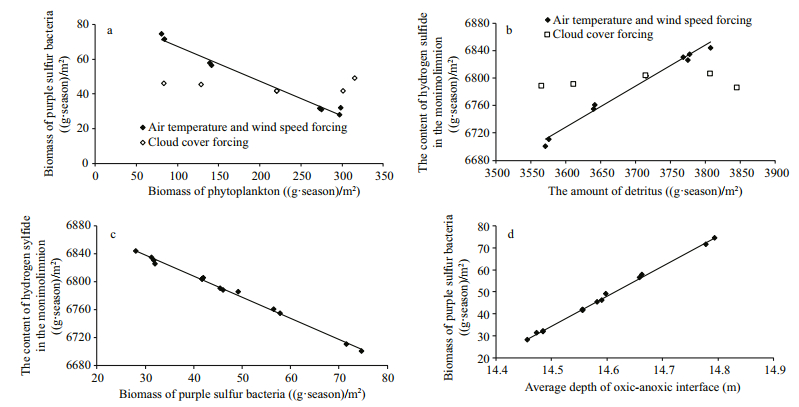
| Fig.3 Correlations between light-dependent food-web components (phytoplankton and purple sulfur bacteria), monimolimnetic food-web components, and the depth of oxic-anoxic interface |
Regardless of the weather parameter affecting the average depth of the thermocline, a significant positive correlation was observed between the depth of the thermocline and total seasonal biomass of phytoplankton (R=0.92; P < 0.001) (Fig. 2a). Correlation between the temperature of the epilimnion and total seasonal biomass of phytoplankton depended on the weather factor. When the temperature of the epilimnion was increased due to the effect of air temperature or wind speed, the total seasonal biomass of phytoplankton decreased. When the temperature of the epilimnion increased due to the effect of cloud cover, the total seasonal biomass of phytoplankton increased (Fig. 2b).
There also was a significant positive correlation between the total seasonal biomasses of phyto- and zooplankton (R=0.94; P < 0.001) (Fig. 2c). Significant positive correlations were found between the total seasonal biomasses of phyto- or zooplankton and the total seasonal biomass of detritus (R=0.99; P < 0.001 and R=0.94; P < 0.001, respectively) (Fig. 2d).
Several correlations also demonstrated how the components of the mixolimnion and monimolimnion were coupled. The principal link was between the groups of light-dependent components of the food web: the total biomass of phytoplankton in the mixolimnion and the total biomass of purple sulfur bacteria in the oxic-anoxic interface (Fig. 3a). When the air temperature and wind speed were varied, there was a significant negative correlation between the biomasses of phytoplankton and purple sulfur bacteria (R=-0.99; P < 0.001). When the cloud cover was varied, there was no correlation between the biomasses of phytoplankton and purple sulfur bacteria.
The effects of mixolimnetic and monimolimnetic food web components on the content of hydrogen sulfide in the monimolimnion were expressed by two correlations: a) a significant positive correlation between the content of detritus in the water column and the content of hydrogen sulfide in the monimolimnion (R=0.74; P=0.001) (Fig. 3b); b) a significant negative correlation between the biomass of purple sulfur bacteria and the content of hydrogen sulfide in the monimolimnion (R=-0.99; P < 0.001) (Fig. 3c). Finally, there was a significant positive correlation between the average depth of the oxicanoxic interface and the biomass of purple sulfur bacteria (R=0.99; P < 0.001) (Fig. 3d).
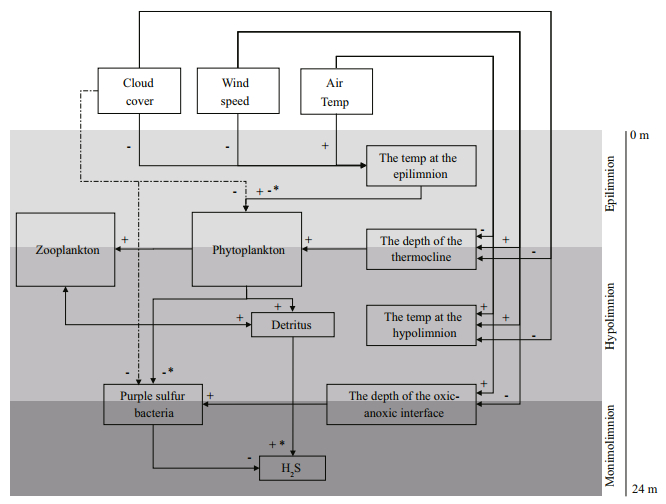
The results can be summarized as the scheme of the weather-induced interactions and changes in the lake ecosystem of Lake Shira (Fig. 4). This scheme demonstrates how changes in weather parameters affected the mixing regime of the lake and how these changes shaped the food web in the mixolimnion and monimolimnion.
|
| Fig.4 The effect of weather forcing on the stratification of the water column and the food web in Lake Shira "+" denotes the positive correlation between components; "–": negative correlation; +*: the correlation is positive only for the values calculated with the variations in air temperature and wind speed; -*: the correlation is negative only for the values calculated with the variations in air temperature and wind speed; +-*: the correlation is positive for the values calculated with the variations in cloud cover and negative for the values calculated with the variations in air temperature and wind speed. Significances of correlations between different components are presented in the Results. |
We started our study by conducting the sensitivity analysis, which was done to compare the effects of external and internal forcing on the food web of a meromictic lake. The sensitivity analysis demonstrated that weather parameters were among the most influential ones, both for mixolimnetic phyto- and zooplankton and for the components of the monimolimnion (bacteria and hydrogen sulfide). Previously, using the field data, we demonstrated the effect of air temperature on the depth of the oxicanoxic interface of Lake Shira, the temperature profiles during summer stratification, and the biomass of seston (Zadereev et al., 2014). However, it is important to note that according to sensitivity analysis, the air temperature was not among the most influential weather parameters. Responses of the components of both the mixolimnion and the monimolimnion to variations in cloud cover and wind speed were comparable in strength to their responses to variations in air temperature. Indeed, a recent study (Rogozin et al., 2017) showed that the effects of wind and temperature were responsible for the mixing event in Lake Shira in 2015. Many studies addressing climate change effects on lakes consider air temperature as the main external forcing (e.g. Livingstone, 2003). However, other climate and weather parameters may well compensate for the effect of temperature change. Thus, climatic scenarios should take into account complex interactions of weather parameters.
In this study, we simulated the effect of the selected parameters on food web components only during the summer season. For stratified and meromictic lakes, long-term and seasonal effects of weather and climate on the ecosystem are important too (e.g. Ito and Momii, 2015). For example, if a lake freezes every winter, the climate and weather variations influence the time of ice clearance and ice formation, which may change the time of stratification (spring) and destratification (autumn) (Arvola et al., 2009). A change in the time of ice clearance is a critical event for the development of the spring phytoplankton and zooplankton and subsequent timing of the clearwater phase (Gerten and Adrian, 2000). To imitate similar effects, we varied the initial spring biomasses of different food web components. Indeed, the initial spring biomasses of some of the food web components were among the most influential parameters. Monimolimnetic components were mostly controlled by the spring biomass of detritus in the water column. Our analysis confirms the importance of initial spring conditions for the development of the lake ecosystem. However, initial spring conditions in stratified lakes are also controlled by climate (Weyhenmeyer et al., 1999). Thus, our study is mostly valid for the assessment of short-term effects of variable weather on the ecosystem of a meromictic lake while longterm multiannual effects should have stronger impact (Ito and Momii, 2015; Rogozin et al., 2017).
Surprisingly, the ecosystem was not very sensitive either to the effect of spring nutrient concentrations or to nutrient inflows during the season. Usually, lake models are quite sensitive to the effects of nutrients (e.g. Arhonditsis and Brett, 2005; Elliott et al., 2006). At the same time, internal nutrient loading may be more important than external nutrient forcing in the case of frequent mixing events (Burger et al., 2008). In the meromictic lake, nutrients are effectively deposited in the monimolimnion (Zadereev et al., 2014). Thus, neither internal nor external nutrient loadings will probably be among the driving forces of ecosystem development in a meromictic lake because of nutrient deposition into the deep waters.
4.2 The effects of weather parameters on thermal and mixing regimes of the water column of the meromictic lakeWe observed that weather forcing had a strong effect on the temperature of the epilimnion, while for the hypolimnion this effect was less pronounced. We estimated that water temperature in the epilimnion would increase by 0.73℃ and in the hypolimnion by 0.01℃ per 1℃ increase in air temperature, which is in good agreement with similar published estimates (e.g. Robertson and Ragotzkie, 1990; Hondzo and Stefan, 1993). Our calculations support observations demonstrating that an increase in the air temperature warms up the epilimnion but does not affect the hypolimnion (Liu et al., 2014). To increase hypolimnetic temperatures in thermally stratified lakes, winds should be strong enough to stimulate mixing (Arvola et al., 2009).
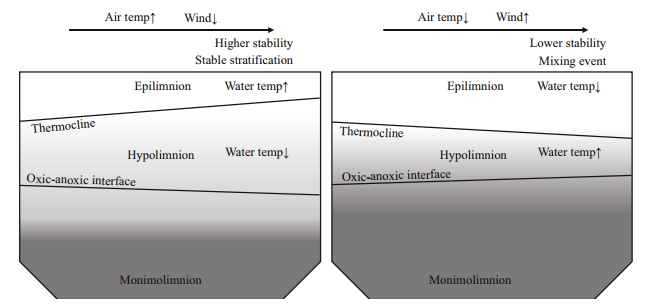
The greatest variations in the depth of the thermocline and the oxic-anoxic interface in our calculations were caused by variations in the wind speed and air temperature. Other models of stratified lakes demonstrated similar results for the depth of the thermocline. For example, simulating water temperatures and stratification of a pre‐alpine lake with a hydrodynamic model, Bueche and Vetter (2014) demonstrated that the model was most sensitive to air temperature and wind speed. Our results suggest that these two parameters also affect the depth of the oxic-anoxic interface. We can predict two scenarios in response to opposite variations in the values of the wind speed and air temperature. First, when the air temperature increases and the wind speed decreases, the water temperature in the epilimnion and the depth of the thermocline will increase while the depth of the oxic-anoxic interface will decrease. Second, when the air temperature decreases and the wind speed increases, the water temperature of the epilimnion and the thermocline depth will decrease while the depth of the oxic-anoxic interface will increase. It is usually considered that the first scenario will promote a higher degree of thermal stability of the water column (e.g. Jankowski et al., 2006). It is important for a meromictic lake that the second scenario predicts less stable stratification and higher probability of lake overturn (Fig. 5). For example, it was demonstrated that in Lake Lugano cold and windy weather destabilized the water column and led to a mixing event (Holzner et al., 2009).
|
| Fig.5 Hypothetical scenarios of interactions of weather parameters that can change the stability of the stratification of the water column Left panel scenario: higher air temperatures and lower wind speed; right panel scenario: lower air temperature and higher wind speed. |
Salinity and water level are two additional important parameters that are linked to weather and climate variability. Multiannual dynamics of these parameters are critical for the stability of stratification of a saline meromictic lake (e.g. Rogozin et al., 2017). Depending on the direction, changes in the water level and related changes in salinity can stimulate a lake to switch from meromixis to holomixis or vice versa (e.g. Melack et al., 2017). Our model analysis was not focused on multiannual dynamics of a meromictic lake. The water level and salinity of the lake were fixed. Multiannual and interannual (seasonal) weather and climatic effects are most likely more important for the control of meromixis stability through water level and salinity fluctuations. Still, we consider the results of our model analysis as an important contribution to the understanding of complex effects of weather parameters on meromictic lake ecosystems.
4.3 The effects of weather parameters on the ecosystemThe effects of weather parameters on the ecosystem are complex. Air temperature and wind speed control the water temperature and mixing depth, while cloud cover mostly affects the amount of light, which is important both for the water temperature and for the photosynthesizing species.
Our calculations demonstrated that any changes in weather parameters that increase the mixing depth also increase the biomass of phytoplankton. This prediction is in close agreement with the previous theoretical calculations and field observations that demonstrated an increase in the standing stock of algal biomass with an increase in mixing depth of between 2 and 10 m (Berger et al., 2006).
The effect of the temperature of the epilimnion on the biomass of phytoplankton is more complex. Obviously, it also depends on the mixing depth of the water column. Under the effect of the air temperature or the wind speed, the increase in the water temperature in the epilimnion coincides with the decrease in the mixing depth. Hence, the biomass of phytoplankton decreases with an increase in the water temperature when it is stimulated by air temperature or wind speed.
As to the effect of clouds, less cloud cover results in more light and deeper and warmer epilimnion. Thus, in our calculations the biomass of phytoplankton increased with the increase in the water temperature in the epilimnion when this increase was induced by variations in cloud cover. Rinke et al. (2010) previously described complex interactions between light extinction, thermal stratification, and phytoplankton. Indeed, these factors are closely interrelated with light climate, depending on the light extinction coefficient, which is affected by phytoplankton biomass (Rinke et al., 2010). Not only phytoplankton biomass but also species composition will be affected by the complex interactions between light, physical stability, and nutrient availability (e.g. Madgwick et al., 2006), but this issue is beyond the scope of our study.
Another positive correlation linked primary producers and zooplankton. It was not surprising that we observed a positive correlation between the total biomasses of phyto- and zooplankton, as the food web in our model was truncated, missing fish or other zooplankton predators. Most probably, this is an example of bottom-up control, when the biomass of primary producers controls the abundance of higher trophic levels (McCauley and Kalff, 1981).
There are two major links between mixolimnetic and monimolimnetic food-web components that depend on weather conditions. One is related to the positive correlation between the biomasses of phytoand zooplankton and detritus. The amount of detritus is important for the production of hydrogen sulfide in the deep waters of a meromictic lake; detritus is involved in the sulfur cycle.
When weather forcing stimulated the increase in the biomass of food-web components in the mixolimnion, it produced a cascading effect, resulting in the higher content of the detritus in the water column and the higher content of hydrogen sulfide in the monimolimnion. This cascading effect was complicated by the interaction of light-dependent photosynthetic species at the different depths: phytoplankton in the mixolimnion and purple sulfur bacteria at the oxic-anoxic interface. A strong negative correlation between these two spatially separated light-dependent components was observed when changes in the biomass were induced by air temperature or wind speed. Most probably, a decrease in phytoplankton biomass leads to an increase in water transparency, more favorable light conditions, and an increase in the biomass of purple sulfur bacteria. Previously, Rogozin et al. (2010) showed that the concentrations of purple sulfur bacteria in the oxic-anoxic interfaces of lakes Shira and Shunet depended on weather conditions and on the transparency of water in the lake. It was demonstrated that the maximal biomass of bacteria was observed during those years when water transparency was the highest. When an increase in the biomass of phytoplankton was stimulated by a decrease in the cloud cover, the biomass of purple sulfur bacteria was constant. Again, the effect of the cloud cover changes both the thermal regime and light climate.
Purple sulfur bacteria at the oxic-anoxic interface oxidize hydrogen sulfide, which is the main substrate for their growth (Rogozin et al., 2012). Consequently, the content of hydrogen sulfide in the monimolimnion decreases with an increase in the biomass of PSB. In turn, the biomass of PSB positively correlates with the depth of the oxic-anoxic interface. We should note that these complex interactions between physical, chemical, and biological parameters at the depth of oxic-anoxic interface are difficult to interpret at this simplified level of analysis. For example, purple sulfur bacteria are strongly controlled by light. Deeper oxic-anoxic interface implies poorer light conditions for photosynthetic bacteria. Thus, correlations that we discuss are valid only for the specific range of depths and boundary conditions that we use.
5 CONCLUSIONWe demonstrated how weather-related changes in the water temperature and mixing depth affected both mixolimnetic and monimolimnetic food-web components and the depth of the oxic-anoxic interface during the growing season. We can summarize that weather conditions that stimulate a higher phytoplankton biomass will lead to higher detritus content and lower biomass of purple sulfur bacteria, a higher content of hydrogen sulfide, and a less deep oxic-anoxic interface. The same weather conditions (stronger wind, thinner cloud cover, and lower temperature) favor less stable thermal stratification. Thus, interaction of these abiotic and biotic changes will be associated with a higher probability of lake overturn.
Variations in the depth of the oxic-anoxic interface induced by weather variability during growing season should not be overestimated. Our calculations demonstrated that under different weather scenarios, the variability of the average depth of the oxic-anoxic interface was in the range of 14.4–14.8 m (2%–3% variability). At the same time, the naturally observed variability of the depth of the oxic-anoxic interface in Lake Shira for the period between 2007 and 2011 was 11–17 m (40%–50%) (Zadereev et al., 2014). Thus, the control of the depth of oxic-anoxic interface by other factors is stronger. For example, we did not use the depth of the oxic-anoxic interface in spring as a forcing function. However, it was demonstrated before that the depth of the oxic-anoxic interface in spring was largely determined by the mixing depth of the mixolimnion in the autumn of the previous year (Zadereev et al., 2014). Thus, multiannual model analysis of response of the meromictic lake ecosystem to weather and climate forcing is needed.
We have already mentioned that water level and salinity are important factors that control the stability of stratification of a saline meromictic lake. The maximal depth of Lake Shira in the last 120 years has changed from 16 to 24 m (Rogozin et al., 2010). Recent analysis demonstrated that interannual change of the water level of Lake Shira is strongly positively related to the annual sum of precipitation. The water level of Lake Shira also has strong positive long-term trend, probably driven by the continual groundwater inflow (Babushkina et al., 2017). Changes in maximal depth, salinity, and lake area influence the stability of the permanent stratification. Several examples are known in which the mixing patterns of lakes have changed because of the changes in their depth and salinity (Miller et al., 1993; Kaden et al., 2010). For Lake Shira, the stabilization of the water level coincided with consistent multiannual weather patterns, which destabilized meromixis in 2015 (Rogozin et al., 2017). Thus, the response of the meromictic lake to external and internal forcing is more complex. Most probably, there is a hierarchy of seasonal, annual, and interannual effects that control the ecosystem development and the stability of meromixis. Our research was focused on seasonal effects. More sophisticated modeling software is required to analyze the broader context.
Despite the limitations mentioned above, we consider the obtained results as quite valuable. As emphasized by Shimoda et al. (2011), it is essential to examine the heterogeneity of in-lake processes that are affected by climate change in different water bodies. Previously, complex effects of weather forcing on stratification and food-web interactions in meromictic lakes were not explicitly studied using numerical modelling. For the first time we demonstrate that, even in the case of considerable variation in the spring biomasses of food web components and nutrient flows, the ecosystem of a stratified meromictic lake is very sensitive to variations in weather parameters. Also, the present study characterized a detailed network of weather-induced physical and biogeochemical interactions between mixolimnetic and monimolimnetic food-web components in a meromictic lake.
6 DATA AVAILABILITY STATEMENTThe data that support the findings of this study are available from the corresponding author upon reasonable request.
7 ACKNOWLEDGMENTWe are grateful to all colleagues from the Institute of Biophysics SB RAS and other academic institutes who have been engaged in long-term research at Lake Shira, providing the field data to support modeling. Special thanks to Prof. Aharon Oren, Elena Krasova and Sofia Shishatskaya for linguistic improvements. Two anonymous reviewers are acknowledged for helpful comments and suggestions.
Simlab. 2011. Software package for uncertainty and sensitivity analysis. Joint Research Centre of the European Commission. http://simlab.jrc.ec.europa.eu.
|
Arhonditsis G B, Brett M T. 2005. Eutrophication model for Lake Washington (USA):Part Ⅰ. model description and sensitivity analysis. Ecol. Modell., 187(2-3): 140-178.
DOI:10.1016/j.ecolmodel.2005.01.040 |
Arvola L, George G, Livingstone D M, Järvinen M, Blenckner T, Dokulil M T, Jennings E, Aonghusa C N, Nõges P, Nõges T, Weyhenmeyer G A. 2009. The impact of the changing climate on the thermal characteristics of lakes.In: George G ed. The Impact of Climate Change on European Lakes. Aquatic Ecology Series, vol. 4. Springer, Dordrecht. p.85-101. https://link.springer.com/chapter/10.1007%2F978-90-481-2945-4_6
|
Babushkina E A, Belokopytova L V, Grachev A M, Meko D M, Vaganov E A. 2017. Variation of the hydrological regime of Bele-Shira closed basin in Southern Siberia and its reflection in the radial growth of Larix sibirica. Reg.Environ. Change, 17(6): 1725-1737.
DOI:10.1007/s10113-017-1137-1 |
Belolipetsky P V, Belolipetskii V M, Genova S N, Mooij W M. 2010. Numerical modeling of vertical stratification of Lake Shira in summer. Aquat. Ecol., 44(3): 561-570.
DOI:10.1007/s10452-010-9330-z |
Belolipetsky V M, Genova S N. 1998. Investigation of hydrothermal and ice regimes in hydropower station bays. Int. J. Comput. Fluid. Dyn., 10(2): 151-158.
DOI:10.1080/10618569808961681 |
Berger S A, Diehl S, Kunz T J, Albrecht D, Oucible A M, Ritzer S. 2006. Light supply, plankton biomass, and seston stoichiometry in a gradient of lake mixing depths. Limnol. Oceanogr., 51(4): 1898-1905.
DOI:10.4319/lo.2006.51.4.1898 |
Boehrer B, Schultze M. 2008. Stratification of lakes. Rev.Geophys., 46(2): RG2005.
|
Bueche T, Vetter M. 2014. Simulating water temperatures and stratification of a pre-alpine lake with a hydrodynamic model:calibration and sensitivity analysis of climatic input parameters. Hydrol. Proc., 28(3): 1450-1464.
DOI:10.1002/hyp.v28.3 |
Burger D F, Hamilton D P, Pilditch C A. 2008. Modelling the relative importance of internal and external nutrient loads on water column nutrient concentrations and phytoplankton biomass in a shallow polymictic lake. Ecol. Modell., 211(3-4): 411-423.
DOI:10.1016/j.ecolmodel.2007.09.028 |
Butcher J B, Nover D, Johnson T E, Clark C M. 2015. Sensitivity of lake thermal and mixing dynamics to climate change. Clim. Change, 129(1-2): 295-305.
DOI:10.1007/s10584-015-1326-1 |
Degermendzhy A G, Zadereev Y S, Rogozin D Y, Prokopkin I G, Barkhatov Y V, Tolomeev A P, Khromechek E B, Janse J H, Mooij W M, Gulati R D. 2010. Vertical stratification of physical, chemical and biological components in two saline lakes Shira and Shunet (South Siberia, Russia). Aquat. Ecol., 44(3): 619-632.
DOI:10.1007/s10452-010-9336-6 |
Elliott J A, Jones I D, Thackeray S J. 2006. Testing the sensitivity of phytoplankton communities to changes in water temperature and nutrient load, in a temperate lake. Hydrobiologia, 559(1): 401-411.
DOI:10.1007/s10750-005-1233-y |
Gaevsky N A, Zotina T A, Gorbaneva T B. 2002. Vertical structure and photosynthetic activity of Lake Shira phytoplankton. Aquat. Ecol., 36(2): 165-178.
DOI:10.1023/A:1015609011227 |
Gerten D, Adrian R. 2000. Climate-driven changes in spring plankton dynamics and the sensitivity of shallow polymictic lakes to the North Atlantic Oscillation. Limnol.Oceanogr., 45(5): 1058-1066.
DOI:10.4319/lo.2000.45.5.1058 |
Hadley K R, Paterson A M, Stainsby E A, Michelutti N, Yao H, Rusak J A, Ingram R, McConnell C, Smol J P. 2014. Climate warming alters thermal stability but not stratification phenology in a small north-temperate lake. Hydrol. Proc., 28(26): 6309-6319.
DOI:10.1002/hyp.v28.26 |
Holzner C P, Aeschbach-Hertig W, Simona M, Veronesi M, Imboden D M, Kipfer R. 2009. Exceptional mixing events in meromictic Lake Lugano (Switzerland/Italy), studied using environmental tracers. Limnol. Oceanogr., 54(4): 1113-1124.
DOI:10.4319/lo.2009.54.4.1113 |
Hondzo M, Stefan H G. 1993. Regional water temperature characteristics of lakes subjected to climate change. Clim.Change, 24(3): 187-211.
DOI:10.1007/BF01091829 |
Ito Y, Momii K. 2015. Impacts of regional warming on long-term hypolimnetic anoxia and dissolved oxygen concentration in a deep lake. Hydrol. Proc., 29(9): 2232-2242.
DOI:10.1002/hyp.v29.9 |
Jankowski T, Livingstone D M, Bührer H, Forster R, Niederhauser P. 2006. Consequences of the 2003 European heat wave for lake temperature profiles, thermal stability, and hypolimnetic oxygen depletion:implications for a warmer world. Limnol. Oceanogr., 51(2): 815-819.
DOI:10.4319/lo.2006.51.2.0815 |
Janse J H. 2005. Model Studies on the Eutrophication of Shallow Lakes and Ditches. Wageningen University, Wageningen, The Netherlands. 378p.
|
Jasser I, Arvola L. 2003. Potential effects of abiotic factors on the abundance of autotrophic picoplankton in four boreal lakes. J. Plankton Res., 25(8): 873-883.
DOI:10.1093/plankt/25.8.873 |
Jöhnk K D, Huisman J, Sharples J, Sommeijer B, Visser P M, Stroom J M. 2008. Summer heatwaves promote blooms of harmful cyanobacteria. Glob. Change Biol., 14(3): 495-512.
DOI:10.1111/gcb.2008.14.issue-3 |
Jones I, Sahlberg J, Persson I. 2010. Modelling the impact of climate change on the thermal characteristics of lakes. In: George G ed. The Impact of Climate Change on European Lakes. Aquatic Ecology Series, vol. 4. Springer, Dordrecht. p.103-120. https://link.springer.com/chapter/10.1007%2F978-90-481-2945-4_7
|
Kaden H, Peeters F, Lorke A, Kipfer R, Tomonaga Y, Karabiyikoglu M. 2010. Impact of lake level change on deep-water renewal and oxic conditions in deep saline Lake Van, Turkey. Water Resour. Res., 46(11): W11508.
|
Kopylov A I, Kosolapov D B, Romanenko A V, Degermendzhy A G. 2002. Structure of planktonic microbial food web in a brackish stratified Siberian lake. Aquat. Ecol., 36(2): 179-204.
DOI:10.1023/A:1015678918611 |
Liu W T, Bocaniov S A, Lamb K G, Smith R E H. 2014. Three dimensional modeling of the effects of changes in meteorological forcing on the thermal structure of Lake Erie. J. Great Lakes Res., 40(4): 827-840.
DOI:10.1016/j.jglr.2014.08.002 |
Livingstone D M. 2003. Impact of secular climate change on the thermal structure of a large temperate central European lake. Clim. Change, 57(1-2): 205-225.
|
Madgwick G, Jones I D, Thackeray S J, Elliott J A, Miller H J. 2006. Phytoplankton communities and antecedent conditions:high resolution sampling in Esthwaite Water. Freshw. Biol., 51(10): 1798-1810.
DOI:10.1111/fwb.2006.51.issue-10 |
McCauley E, Kalff J. 1981. Empirical relationships between phytoplankton and zooplankton biomass in lakes. Can. J.Fish. Aquat. Sci., 38(4): 458-463.
DOI:10.1139/f81-063 |
Melack J M, Jellison R, MacIntyre S, Hollibaugh J T. 2017.Mono Lake: plankton dynamics over three decades of meromixis or monomixis. In: Gulati R D, Zadereev E S, Degermendzhi A G eds. Ecology of Meromictic Lakes.Springer, Cham. p.325-351. https://link.springer.com/chapter/10.1007/978-3-319-49143-1_11
|
Miller L G, Jellison R, Oremland R S, Culbertson C W. 1993. Meromixis in hypersaline Mono Lake, California. 3. biogeochemical response to stratification and overturn.Limnol. Oceanogr., 38(5): 1040-1051.
|
Mooij W M, Janse J H, De Senerpont Domis L N, Hülsmann S, Ibelings B W. 2007. Predicting the effect of climate change on temperate shallow lakes with the ecosystem model PCLake. Hydrobiologia, 584(1): 443-454.
DOI:10.1007/s10750-007-0600-2 |
Mooij W M, Trolle D, Jeppesen E, Arhonditsis G, Belolipetsky P V, Chitamwebwa D B R, Degermendzhy A G, DeAngelis D L, De Senerpont Domis L N, Downing A S, Elliott J A, Fragoso C R Jr, Gaedke U, Genova S N, Gulati R D, Håkanson L, Hamilton D P, Hipsey M R, Hoen J, Hülsmann S, Los F H, Makler-Pick V, Petzoldt T, Prokopkin I G, Rinke K, Schep S A, Tominaga K, Van Dam A A, Van Nes E H, Wells S A, Janse J H. 2010. Challenges and opportunities for integrating lake ecosystem modelling approaches. Aquat. Ecol., 44(3): 633-667.
DOI:10.1007/s10452-010-9339-3 |
Morris M D. 1991. Factorial sampling plans for preliminary computational experiments. Technometrics, 33(2): 161-174.
DOI:10.1080/00401706.1991.10484804 |
Paerl H W, Huisman J. 2008. Blooms like it hot. Science, 320(5872): 57-58.
DOI:10.1126/science.1155398 |
Prokopkin I G, Barkhatov Y V, Khromechek E B. 2014. A onedimensional model for phytoflagellate distribution in the meromictic lake. Ecol. Modell., 288: 1-8.
DOI:10.1016/j.ecolmodel.2014.05.011 |
Prokopkin I G, Mooij W M, Janse J H, Degermendzhy A G. 2010. A general one-dimensional vertical ecosystem model of Lake Shira (Russia, Khakasia):description, parametrization and analysis. Aquat. Ecol., 44(3): 585-618.
DOI:10.1007/s10452-010-9326-8 |
Rinke K, Yeates P, Rothhaupt K O. 2010. A simulation study of the feedback of phytoplankton on thermal structure via light extinction. Freshw. Biol., 55(8): 1674-1693.
|
Robertson D M, Ragotzkie R A. 1990. Changes in the thermal structure of moderate to large sized lakes in response to changes in air temperature. Aquat. Sci., 52(4): 360-380.
DOI:10.1007/BF00879763 |
Rogozin D Y, Genova S N, Gulati R D, Degermendzhy A G. 2010. Some generalizations based on stratification and vertical mixing in meromictic Lake Shira, Russia, in the period 2002-2009. Aquat. Ecol., 44(3): 485-496.
DOI:10.1007/s10452-010-9328-6 |
Rogozin D Y, Tarnovsky M O, Belolipetskii V M, Zykov V V, Zadereev E S, Tolomeev A P, Drobotov A V, Barkhatov Y V, Gaevsky N A, Gorbaneva T B, Kolmakova A A, Degermendzhi A G. 2017. Disturbance of meromixis in saline Lake Shira (Siberia, Russia):possible reasons and ecosystem response. Limnologica Limnol. Ecol. Manage.Inland Waters, 66: 12-23.
|
Rogozin D Y, Zykov V V, Degermendzhi A G. 2012. Ecology of purple sulfur bacteria in the highly stratified meromictic Lake Shunet (Siberia, Khakassia) in 2002-2009. Microbiology, 81(6): 727-735.
DOI:10.1134/S0026261712060148 |
Saltelli A, Tarantola S, Campolongo F, Ratto M. 2004. Sensitivity Analysis in Practice:A Guide to Assessing Scientific Models. John Wiley & Sons, Ltd., Chichester. 217p.
|
Shimoda Y, Azim M E, Perhar G, Ramin M, Kenney M A, Sadraddini S, Gudimov A, Arhonditsis G B. 2011. Our current understanding of lake ecosystem response to climate change:what have we really learned from the north temperate deep lakes? J. Great Lakes Res., 37(1): 173-193.
DOI:10.1016/j.jglr.2010.10.004 |
Straile D. 2000. Meteorological forcing of plankton dynamics in a large and deep continental European lake. Oecologia, 122(1): 44-50.
DOI:10.1007/PL00008834 |
Tolomeyev A P. 2002. Phytoplankton diet of Arctodiaptomus salinus (Copepoda, Calanoida) in lake Shira (Khakasia). Aquat. Ecol., 36(2): 229-234.
DOI:10.1023/A:1015618104520 |
Weyhenmeyer G A, Blenckner T, Pettersson K. 1999. Changes of the plankton spring outburst related to the North Atlantic Oscillation. Limnol. Oceanogr., 44(7): 1788-1792.
DOI:10.4319/lo.1999.44.7.1788 |
Winder M, Schindler D E. 2004. Climate change uncouples trophic interactions in an aquatic ecosystem. Ecology, 85(8): 2100-2106.
DOI:10.1890/04-0151 |
Zadereev E S, Boehrer B, Gulati R D. 2017a. Introduction: meromictic lakes, their terminology and geographic distribution. In: Gulati R D, Zadereev E S, Degermendzhi A G eds. Ecology of Meromictic Lakes. Springer, Cham.p.1-11. https://link.springer.com/chapter/10.1007%2F978-3-319-49143-1_1
|
Zadereev E S, Gulati R D, Camacho A. 2017b. Biological and ecological features, trophic structure and energy flow in meromictic lakes. In: Gulati R D, Zadereev E S, Degermendzhi A G eds. Ecology of Meromictic Lakes.Springer, Cham. p.61-86. https://link.springer.com/chapter/10.1007%2F978-3-319-49143-1_4
|
Zadereev E S, Tolomeev A P, Drobotov A V, Kolmakova A A. 2014. Impact of weather variability on spatial and seasonal dynamics of dissolved and suspended nutrients in water column of meromictic Lake Shira. Contemp. Probl. Ecol., 7(4): 384-396.
DOI:10.1134/S199542551404012X |
Zadereev Y S, Tolomeyev A P. 2007. The vertical distribution of zooplankton in brackish meromictic lake with deepwater chlorophyll maximum. Hydrobiologia, 576(1): 69-82.
DOI:10.1007/s10750-006-0294-x |
Zotina T A, Tolomeyev A P, Degermendzhy N N. 1999. Lake Shira, a Siberian salt lake:ecosystem structure and function:1. Major physico-chemical and biological features. Int. J. Salt Lake Res., 8(3): 211-232.
|
 2018, Vol. 36
2018, Vol. 36