Institute of Oceanology, Chinese Academy of Sciences
Article Information
- Rania S. MABROKE, Osama M. EL-HUSSEINY, Abd El-Naem F. A. ZIDAN, Al-Azab TAHOUN, Ashraf SULOMA
- Floc meal as potential substitute for soybean meal in tilapia diets under biofloc system conditions
- Journal of Oceanology and Limnology, 37(1): 313-320
- http://dx.doi.org/10.1007/s00343-019-7222-1
Article History
- Received Aug. 24, 2017
- accepted in principle Jan. 22, 2018
- accepted for publication Mar. 7, 2018
2 Aquaculture Department, Faculty of Fish Resources, Suez University, Egypt
The more rise in world population; necessitate developing the aquaculture industry to partly satisfy the global demand for animal protein sources. In order to support the development of aquaculture industry, developing economical diets by using unconventional feedstuffs is required since feed costs represent around 50% of the aquaculture production expenses (FAO, 2017). In addition, elevation of soybean price is shown every day as animal and human compete on soy products (Suloma et al., 2014). Therefore, it becomes critical to discover sustainable alternative protein sources not only for fish meal, but also for soybean meal.
Biofloc technology (BFT) has been widely studied and applied in the cultivation of shrimp and tilapia. BFT has beneficial effects, including the recycling of the solids e.g. feces and uneaten feed" and nutrients (e.g. nitrogen and phosphorus), into microbial flocs and avoiding environmental detrimental effects. The system management depends on the elevation of C:N ratio in water body up to 20 to activate the growth of heterotrophic bacteria which have the ability to utilize the ammonia-nitrogen released from these wastes (Avnimelech, 1999). Biofloc community consists of microorganisms, as filamentous cyanobacteria, protozoa, nematode, and copepods (Emerenciano et al., 2013b). This mixture represents a beneficial protein source for tilapia when grazing directly in the water column. It was reported by Moreno-Aria et al. (2017) that the extracellular of floc organisms may contain enzymes that help to improve digestion process inside fish gut. Meanwhile it contains other beneficial components such as carotenoids, amino sugars and vitamins that may positively improve digestion and utilization (Ju et al., 2008). Thus, biofloc system provides suitable condition for more inclusion of vegetable ingredient into fish diets. In the same context, the elevation of total suspended solids above desirable levels may be avoided by precipitation of flocs periodically (Hatem et al., 2013; Khalil et al., 2016; Zidan et al., 2017). The dried floc meal may be used as a beneficial ingredient in fish diet as it contains many nutrients and components such as protein which ranged between 24%–50% (Ju et al., 2008; Kuhn et al., 2010b; Maicá et al., 2012; López-Elías et al., 2015; Dantas et al., 2016; Gaona et al., 2016) and lipids ranged between 0.5%–3.5% (Azim and Little, 2008; Kuhn et al., 2009; Maicá et al., 2012; Emerenciano et al., 2013a). Adoption of biofloc system to generate unconventional feedstuff "microbial flocs" is another prospective for improving its sustainability. Moreover, some authors suggested the ability to replace soybean by flocs derived from treating fish effluent in shrimp diets (Kuhn et al., 2009, 2010a, b). No available data on replacing dietary soybean meal with floc meal in tilapia diets were found in the literature. The aim of our work was to examine the potential substitution of soybean meal in tilapia diets by flocs meal under biofloc system condition to improve its sustainability.
2 MATERIAL AND METHOD 2.1 Fish and experimental conditionsThe present study was carried out at the Fish Nutrition Laboratory (FNL), Department of Animal Production, Faculty of Agriculture, Cairo University, Egypt. Nile tilapia were obtained from a commercial hatchery located in Kafr El-Sheikh Governorate, Egypt, and adapted to experimental conditions for 2 weeks. Tilapia juvenile with average weight of 12.14 g±0.03 were randomly distributed in the experimental tanks that were cited in the wet-lab. The system included nine circular plastic tanks (85 L capacity filled to 55 L) to form three experimental groups. Each tank was stocked by 12 fish corresponding to the density of 3 kg tilapia/m3. The experiment was conducted for 56 days. Experimental tanks were supplied with well water and 8 L of previously matured biofloc with TSS ˃300 mg/L to initiate the system. The matured biofloc was collected from tilapia (Oreochromis niloticus) tanks. Aeration was continuously provided using an air pump (RS electrical model: RS-348A) and porous stones (Qubic and cylindrical-shape with length of 2 and 4 cm, respectively) to maintain the oxygen supply above 5 mg/L. The experiment was conducted under room temperature and normal photoperiod condition (12 h light:12 h dark).
2.2 Experimental designThe experimental design consisted of three treatments: control diet (C; absence of LPFM and diet contained soybean meal as main protein source); 25% soybean meal protein was substituted by LPFM (FM25); and 50% soybean meal protein was substituted by LPFM (FM50) (Tables 1, 2 and 3).

|
|
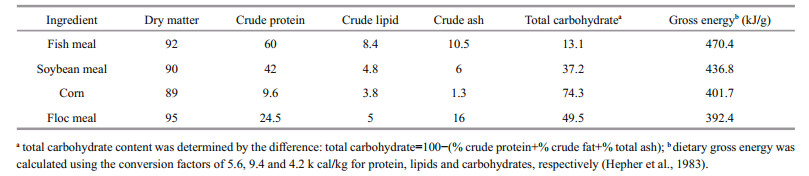
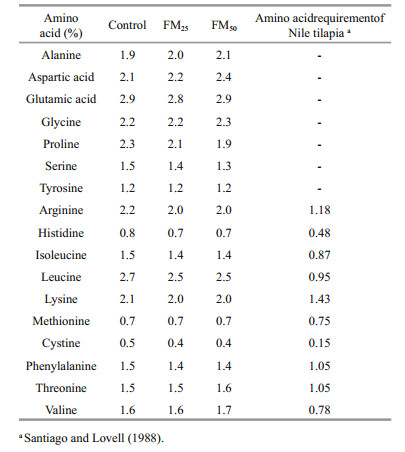
LPFM was collected from biofloc tanks from previous experiments when the TSS reached 300 mg/L. Eighty percent of the tanks water was settled in another tank for 15 min and floc bulk was then collected and sieved by 0.045 mm-mesh to dewater the suspended solid (biofloc). The collected wet biofloc was sun dried, mixed and analyzed. Chemical composition of the floc meal are presented in Table 1. The experimental diets were balanced using Excel package (2007) and were prepared by blending the ingredients into a homogeneous mixture; then the mixture was passed through the laboratory pellet mill. The pellets were then dried overnight under forced air at room temperature and stored at 4℃ until used. Amino acids profile of the experimental diets (Table 3) were calculated according NRC (2011) and Aboseif (2017) for the amino acid profile of LPFM (24% CP).Fish were fed twice daily (9:00 h and 17:00 h) on 4% of their body weight. Feed ration was adjusted biweekly according to fish biomass changes.
2.4 Water qualityStarch was used as a carbon source to maintain the biofloc system condition at a carbon: nitrogen ratio around 10:1 and the starch quantity was determent according to the method of Crab et al. (2012). Water temperature and dissolved oxygen were measured using Senso DirectOxi 200, while pH was measured using Milwaukee-PH600 Digital pH metertwice weekly. Ammonia-N, nitrite, nitrate values were detected using water analysis photometer (MultiDirectLovibond) once weekly. Biofloc volume was measured using Imhoff cone and the volume of floc on the bottom of the cone was observed after 15 min of sedimentation once weekly (Avnimelech, 2009).Alkalinity was monitored biweekly by titration with sulfuric acid till PH point of 4.5 (APHA, 1998). Total suspended solids (TSS) were monitored weekly by water analysis photometer (MultiDirectLovibond).
Zooplankton samples were collected from different biofloc system using zooplankton net (55 μm, 25 cm diameter and 80 cm length). The water was agitated well and 5 L of water sample was filtered by zooplankton net. After filtration, the samples were fixed immediately using formaldehyde solution (4%– 7%). Two milliliters of Rose Bengal stain (0.5%) was added after fixation. The filtrated samples were examined with Optic Research Microscope. Three sub-samples (one mL for each) of the homogenized plankton samples were transferred to a counting cell and the different plankters were counted. Zooplankton population was then calculated as the number of individuals of the different species per cubic meter.
The organisms were identified and counted on the counting try with magnification varying from 100× to 400×. Planktonic organisms were identified according to description and keys constructed by Edmondson (1959); Ruttner-Kolisko (1974); Pennak (1978) and Shehata et al. (1998) in addition to keys by Fernando et al. (1987) and Jeje (1988).
Zooplankton was calculated after examination for all the recorded species in each sample and expressed for Organisms/litters depending on the following equation according to (APHA, 2005):
number of organisms (L)=N×D/S×C,
where N=number of organisms for the calculated species; D=volume of sample after filtration; S=number of subsamples; C=total volume of the collected water sample.
2.5 Growth parametersFish (n=12) of different treatment groups were weighed biweekly and assessed for growth parameters such as weight gain (%), feed conversion ratio (FCR), and specific growth rate (%) (SGR) as follows:
weight gain (%)=(FW–IW),
FCR=feed given (DW)/body weight gain (WW),
SGR (%)=[ln(FW)–ln(IW)/N] ×100,
where FW=final weight, IW=initial weight, DW=dry weight, WW=wet weight, ln=natural log and N=number of culture days.
2.6 Proximate compositionThe proximate composition of fish, diets and floc meal samples generated from the experimental tanks were determined after completion of the experiment according to AOAC (1995). The moisture content was determined by drying the samples at 105℃ (Binder oven, E series 28, Germany) to a constant weight, and the difference in weight of the sample indicated the moisture content. Nitrogen content was estimated by P3L39: Kjeldah method and crude protein content was calculated by multiplying the nitrogen percentage by 6.25. Crude lipid was determined by soxhlet extraction with ether (boiling point, 40–60℃) as a solvent. Ash content was determined by incinerating the samples in a muffle furnace at 600℃ for 3 h. Total carbohydrate content was determined by the difference (total carbohydrate=100−(% crude protein+% crude fat+% total ash+% moisture).
2.7 Mineral analysisCalcium, sodium, potassium, magnesium, phosphorus, and selenium concentrations in fish carcass and diets were determined according to AOAC (2012) using ICP-AES (Perkin Elmer, Optima 2000DV). The same elements were analyzed in precipitated floc from different experimental treatments except for the control where only one replicate was analyzed.
2.8 Statistical analysisAll statistical analyses were performed using SPSS 17.0 software (SPSS, Chicago, IL, USA). Data were analyzed by one-way ANOVA. Odd replicate value was omitted during statistical analysis to save data integrality. Duncan's multiple range tests was used to identify differences among experimental groups at a significant difference of P≤0.05.
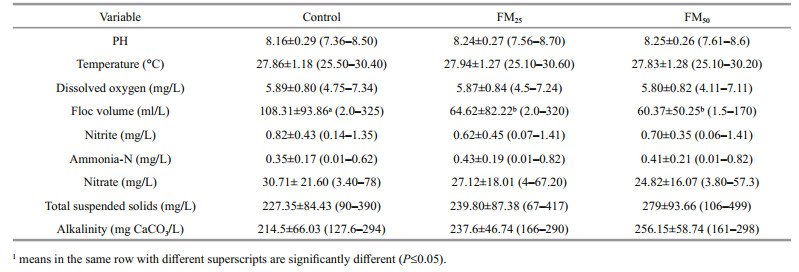
3 RESULT AND DISCUSSION 3.1 Water qualityThe overall mean, standard deviation, and range of water temperature, dissolved oxygen and pH of the different treatments are presented in (Table 4).Water quality parameters were within the optimal range for the culture of tilapia. No significant differences in water quality values were observed among treatments. Fish fed (FM25) recorded the highest Floc volume during the whole experimental period especially after the fifth week. Water temperature values were optimal for fish culture and ranged from 25–30℃. Dissolved oxygen remained within the recommended range for growth of tilapia. The DO average was kept above 5 mg/L, which are within the recommended levels of DO as reported by Azim et al. (2008). Ammonia-N, NO2 and NO3 values recorded for all treatments were within normal ranges (average of 0.39, 0.71 and 27.5 mg/L, respectively). Total suspended solids and alkalinity were numerically higher in floc meal treatments compared to the control and increased with the increasing floc meal in the diet.
|
The potential substitution of soybean meal by flocs meal in tilapia diet under biofloc system was examined and fish growth performance are presented in Table 5. No significant differences in tilapia growth performance between the control diet and FM25 was noticed. The worst growth performance values were recorded for fish fed FM50 which indicate that, more inclusion of floc meal in tilapia diet negatively affects growth performance.
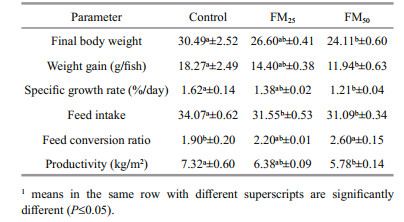
|
The harvest of floc meal from zero-water exchange tanks and its examination as alternative feed ingredient has been previously reported for shrimp (Kuhn et al., 2009, 2010b). However, in the present study, floc meal was collected and evaluated as replacement ingredient for soybean meal protein in tilapia diet. Floc meal, which is typically a waste product, could be considered as high value ingredient. In the present study, protein content of the tested LPFM was 24% CP. The same level was recorded by other authors (López-Elías et al., 2015; Dantas et al., 2016; Gaona et al., 2016) while this level is considered low protein floc meal compared to other reported values; less than 30% (Ekasari et al., 2010), 38% (Kuhn et al., 2016) while higher than 50% was recorded by (Kuhn et al., 2009).
The tested floc meal contained satisfactory protein level for replacing up to 25% of soybean protein. The replacement had no great reflect on the amino acid profile and ash content of the diet subsequently growth performance. Meanwhile, sufficient contribution from microbial biofloc supported the fish growth. This may suggest that microbial biofloc may have the ability to recover the deficiency in amino acids of the diets up to a specific limit. The same results were suggested by other authors, where biofloc did not compromise the growth performance when floc meal inclusion was less than 50% of the substituted ingredient. Floc meal replaced 10%, 20%, and 30% of soybean in shrimp diet without negative effect on growth (Valle et al., 2015; Kuhn et al., 2016). In the same context, Dantas et al. (2016) suggested that 30% fish meal could be replaced by folc meal in shrimp diet without growth suppression. Moreover, Kuhn et al. (2009) found significant differences between treatments groups, in terms of increased weight gain, and SGR when soybean was replaced with microbial flocs at 7.8% and 15.6% inclusion level on a dry matter basis.
The amino acid content of different experimental diets seems to cover the requirement of tilapia even when the floc meal reached 50% (Table 3). The suppression effect of floc meal observed in FM50% diet, could be attributed to: 1. the elevated of dietary ash content as increasing the inclusion of the floc meal in the experimental diet as elevation in dietary Ca:P ratio was observed in FM25 and FM50. It was suggested that optimum Ca:P ratio for tilapia ranged between 0.5–0.9 (Yao et al., 2012). Elevation of dietary Ca may resulted in low absorption of other minerals as phosphorus, iron, zinc and manganese that may negatively affect biological function subsequently growth performance (Lall, 2002; Yao et al., 2012; Hossain and Yoshimatsu, 2014); 2. the low availability of some amino acids in LPFM (Table 3).
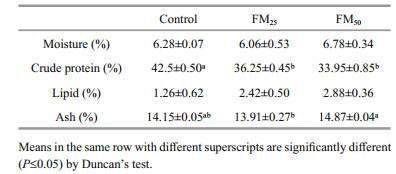
The whole body chemical composition of tilapia are summarized in Table 6. Tilapia protein content showed significantly higher value in fish fed the control diet, while there was numerical decrease in the carcass fat with more inclusion of floc meal in tilapia diet. This may reflect the increase of energy pardon in tilapia fed more floc meal. Despite floc meal had high content of selenium (Table 6), more inclusion of floc meal in tilapia diet was not reflected in fish carcass content of selenium. Meanwhile, selenium was not detected in biofloc precipitated from different experimental treatments. These results may reflect the high utilization of selenium in biological mechanisms during metabolism caused by more inclusion of floc meal in fish diets. Subsequently, the need of supplementing fish diets with more selenium is suggested in the presence of more inclusion of floc meal in fish diet.
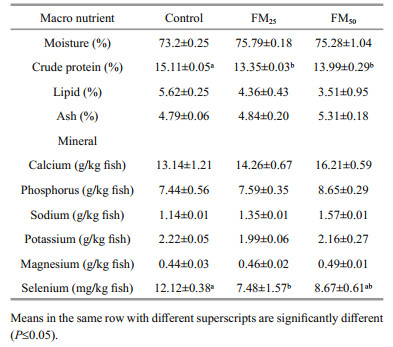
|
The calcium content of different experimental diets was not reflected in calcium content in fish carcass but showed its reflection on calcium level of flocs precipitated from different treatments. It was noticed that the more inclusion of floc meal in fish diet, the more calcium content in precipitated flocs was observed by the end of the experiment. This may be due to: 1. the low bioavailability of floc meal calcium or 2. the excess levels of calcium than required by fish. The last hypothesis could be supported by the carcass Ca:P ratio that showed constant values (1.8) among different treatment. No significant differences were recorded regarding the content of other minerals in fish carcass.
Biofloc quality in terms of fatty acid profile and protein content seems to be affected by the system inputs as sources of carbon (Ekasari et al., 2010; Dantas et al., 2016) meanwhile, dietary composition affected biofloc chemical composition in the present study. Chemical analysis of precipitated folc samples showed slight increase in ash content P≤0.05 with more inclusion of floc meal in tilapia diet (Table 7). Accumulation of minerals may be the cause behind the high ash content (Dantas et al., 2016). The average ash content of both floc meal and precipated floc (16% and 14%, respectively, were within the range reported in previous works being 7%–47% (Azim et al., 2008; Emerenciano et al., 2013b). Protein content of biofloc generated from the control treatment had significantly higher value compared to other treatments. The control treatment recorded the highest zooplankton number among other treatments which may explain its superiority in protein content (Fig. 1). The same was suggested Emerenciano et al. (2013b), who corroborated the variation of floc protein content to the concentration of species such as copepods and nematodes The superiority of control in zooplankton count may be a reflect of fish low grazing activity especially, in the presence of higher feed intake value under condition of no soybean substitution in comparison with substitution condition. The mineral content of precipitated floc varied numerically among different treatments (Table 8) but the values were within the range of previous studies except for sodium and potassium as fresh water supply were used in our experiment. Mineral content of floc meal in previous studies differed depending on the method of producing biofloc, where calcium, phosphorus, sodium, potassium and magnesium values ranged 12.8–26.7; 12.9–22.6; 12.7–24.4; 7.5–13.7; 4.1–18.1 g/kg, respectively (Kuhn et al., 2009, 2010a, 2016). Elevation in Ca:P ratios were noticed in precipitated biofloc in parallel with more inclusion of floc meal in tilapia diets. In the same context a constant Ca:P ratio recorded for tilapia carcass (1.8) in different treatments which support the hypothesis of that excess dietary calcium was excreted into the tank water. Excess calcium may reflect the absorption of other mineral as phosphorus, zinc and iron from the intestine (Lall, 2002; Yao et al., 2012; Hossain and Yoshimatsu, 2014).

|
| Fig.1 Total zooplankton count in different experimental treatments |
Under the condition of our experiment, floc meal substituted soybean up to 25% without negative impact on fish performance. The more inclusion of floc meal into tilapia diet resulted in growth depression. Ash content of floc meal may be the limiting factor behind the growth suppression. Further studies are required to detect the effect of floc meal mineral composition on tilapia performance. According to our work it may increase the energy pardon in tilapia fed diets with more floc meal.
5 DATA AVAILABILITY STATEMENTThe data that support the findings of this study are available from the corresponding author upon reasonable request
Aboseif A M. 2017. Development and Validation of Economic and Eco-Friendly Feed for Tilapia (Oreochromis niloticus)and African Catfish (Clarias gariepinus). Faculty of Agriculture, Cairo University, Giza, Egypt.
|
AOAC. 1995. Official Methods of Analysis. Association of Official Analytical Chemists. 16th Ed. Arlington, VA.
|
AOAC. 2012. Official Methods of Analysis. 19th ed. Association of Official Analytical Chemists, Washington, DC, USA.
|
APHA. 1998. Standard Methods for the Examination of Water and Wastewater. 20th ed. American Public Health Association, Washington, DC, USA.
|
APHA. 2005. Standard Methods for the Examination of Water and Wastewater. 21st ed. Standard Methods is a joint publication of the American Public Health Association(APHA), the American Water Works Association(AWWA), and the Water Environment Federation (WEF), Washington, DC, USA.
|
Avnimelech Y. 1999. Carbon/nitrogen ratio as a control element in aquaculture systems. Aquaculture, 176(3-4): 227-235.
DOI:10.1016/S0044-8486(99)00085-X |
Avnimelech Y. 2009. Biofloc Technology-A Practical Guide Book. The World Aquaculture Society, Baton Rouge.
|
Azim M E, Little D C, Bron J E. 2008. Microbial protein production in activated suspension tanks manipulating C:N ratio in feed and the implications for fish culture. Bioresource Technology, 99(9): 3 590-3 599.
DOI:10.1016/j.biortech.2007.07.063 |
Azim M E, Little D C. 2008. The biofloc technology (BFT) in indoor tanks:water quality, biofloc composition, and growth and welfare of Nile tilapia (Oreochromis niloticus). Aquaculture, 283(1-4): 29-35.
DOI:10.1016/j.aquaculture.2008.06.036 |
Crab R, Defoirdt T, Bossier P, Verstraete W. 2012. Biofloc technology in aquaculture:beneficial effects and future challenges. Aquaculture, 356: 351-356.
|
Dantas Jr E M, Valle B C S, Brito C M S, Calazans N K F, Peixoto S R M, Soares R B. 2016. Partial replacement of fishmeal with biofloc meal in the diet of postlarvae of the Pacific white shrimp Litopenaeus vannamei. Aquaculture Nutrition, 22(2): 335-342.
|
Edmondson W T. 1959. Freshwater Biology. 2nd ed. John Wiley and Sons, Inc, New York. 12248p.
|
Ekasari J, Crab R, Verstraete W. 2010. Primary nutritional content of bio-flocs cultured with different organic carbon sources and salinity. HAYATI Journal of Biosciences, 17(3): 125-130.
DOI:10.4308/hjb.17.3.125 |
Emerenciano M, Cuzon G, Arévalo M, Miquelajauregui M M, Gaxiola G. 2013a. Effect of short-term fresh food supplementation on reproductive performance, biochemical composition, and fatty acid profile of Litopenaeus vannamei (Boone) reared under biofloc conditions. Aquaculture International, 21(5): 987-1 007.
DOI:10.1007/s10499-012-9607-4 |
Emerenciano M, Cuzon G, Paredes A, Gaxiola G. 2013b. Evaluation of biofloc technology in pink shrimp Farfantepenaeus duorarum culture:growth performance, water quality, microorganisms profile and proximate analysis of biofloc. Aquaculture International, 21(6): 1 381-1 394.
DOI:10.1007/s10499-013-9640-y |
Fernando C H, Paggi J C, Rajapaksa R. 1987. Daphnia in tropical lowlands. In: Peters R H, de Bernardi R eds.Daphnia. Memorie dell'Istituto Italiano di Idrobiologia, Hyderabad, Italy, 45: 107-141.
|
Food and agriculture organization of the united nation. 2017.FAO Aquaculture Newsletter (FAN) 56, March 2017.http://www.fao.org/documents/card/en/c/cbeb55d3-cf9c-428f-9450-3c5f0f3191d3/
|
Gaona C A P, da Paz Serra F, Furtado P S, Poersch L H, Wasielesky Jr W. 2016. Biofloc management with different flow rates for solids removal in the Litopenaeus vannamei BFT culture system. Aquaculture International, 24(5): 1 263-1 275.
DOI:10.1007/s10499-016-9983-2 |
Hatem M H, Abdelhay Y B, Alayafi A H, Suloma A. 2013. Application of new strategies to reduce suspended solids in zero-exchange system:I.Histological alterations in the gills of Nile tilapia. Journal of Applied Sciences Research, 9(2): 1 186-1 192.
|
Hepher B, Liao I C, Cheng S H, Hsieh C S. 1983. Food utilization by red tilapia-effects of diet composition, feeding level and temperature on utilization efficiencies for maintenance and growth. Aquaculture, 32(3-4): 255-275.
DOI:10.1016/0044-8486(83)90223-5 |
Hossain M A, Yoshimatsu T. 2014. Dietary calcium requirement in fishes. Aquaculture Nutrition, 20(1): 1-11.
|
Jeje C Y. 1988. A revision of the Nigerian species of the genera Mesocyclops Sars, 1914 and Thermocyclops Kiefer, 1927(Copepoda:Cyclopoida). Hydrobiologia, 164(2): 171-184.
DOI:10.1007/BF00008457 |
Ju Z Y, Forster I, Conquest L, Dominy W, Kuo W C, Horgen F D. 2008. Determination of microbial community structures of shrimp floc cultures by biomarkers and analysis of floc amino acid profiles. Aquaculture Research, 39(2): 118-133.
|
Khalil M T, Mohamed R A, El-Deeb R, Suloma A. 2016. Ecofriendly cultivation of Keeled mullet (Liza carinata) in biofloc system. Egyptian Journal of Aquatic Biology and Fisheries, 20(2): 23-35.
DOI:10.21608/ejabf.2016.2291 |
Kuhn D D, Boardman G D, Flick G J. 2010b. Production of microbial flocs using laboratory-scale sequencing batch reactors and tilapia wastewaters. International Journal of Recirculating Aquaculture, 11: 37-54.
|
Kuhn D D, Boardman G D, Lawrence A L, Marsh L, Flick Jr G J. 2009. Microbial floc meal as a replacement ingredient for fish meal and soybean protein in shrimp feed. Aquaculture, 296(1-2): 51-57.
DOI:10.1016/j.aquaculture.2009.07.025 |
Kuhn D D, Lawrence A L, Boardman G D, Patnaik S, Marsh L, Flick Jr G J. 2010a. Evaluation of two types of bioflocs derived from biological treatment of fish effluent as feed ingredients for Pacific white shrimp, Litopenaeus vannamei. Aquaculture, 303(1): 28-33.
|
Kuhn D D, Lawrence A L, Crockett J, Taylor D. 2016. Evaluation of bioflocs derived from confectionary food effluent water as a replacement feed ingredient for fishmeal or soy meal for shrimp. Aquaculture, 454: 66-71.
DOI:10.1016/j.aquaculture.2015.12.009 |
Lall S P. 2002. The minerals. In: Halver J E, Hardy R W eds.Fish Nutrition. 3rd edn. Academic Press Inc., The San Diego, CA. p.259-308.
|
López-Elías J A, Moreno-Arias A, Miranda-Baeza A, MartínezCórdova L R, Rivas-Vega M E, Márquez-Ríos E. 2015. Proximate composition of bioflocs in culture systems containing hybrid red tilapia fed diets with varying levels of vegetable meal inclusion. North American Journal of Aquaculture, 77(1): 102-109.
DOI:10.1080/15222055.2014.963767 |
Maicá P F, de Borba M R, Wasielesky Jr W. 2012. Effect of low salinity on microbial floc composition and performance of Litopenaeus vannamei (Boone) juveniles reared in a zero-water-exchange super-intensive system. Aquaculture Research, 3(3): 361-370.
|
Moreno-Arias A, López-Elías J A, Miranda-Baeza A, Rivas-Vega M E, Martínez-Córdova L R, Ramírez-Suárez J C. 2017. Replacement of fishmeal by vegetable meal mix in the diets of Litopenaeus vannamei reared in low-salinity biofloc system:effect on digestive enzymatic activity. Aquaculture Nutrition, 23(2): 236-245.
DOI:10.1111/anu.12384 |
NRC. 2011. Nutrient Requirements of Fish and Shrimp. National Academies Press, Washington DC, USA.
|
Pennak R W. 1978. Freshwater Invertebrates of the United States. 2nd ed. John Wiley & Sons, New York. 803p.
|
Ruttner-Kolisko A. 1974. Planktonic rotifers:biology and taxonomy. Die Binnergewässer, 26(S1): 146.
|
Santiago C B, Lovell R T. 1988. Amino acid requirements for growth of Nile tilapia. The Journal of Nutrition, 118(12): 1 540-1 546.
DOI:10.1093/jn/118.12.1540 |
Shehata S M, Shehata K K, Hussien M M, Mageed A A. 1998. Taxonomical and ecological studies on some Zooplankton species:ciliata, rhizopoda, turbellaria and crustacea of the high Dam Lake. Egyptian Journal of Aquatic Biology and Fisheries, 2: 37-63.
|
Suloma A, El-Husseiny O M, Hassane M I, Mabroke R S, ElHaroun E R. 2014. Complementary responses between hydrolyzed feather meal, fish meal and soybean meal without amino acid supplementation in Nile tilapia Oreochromis niloticus diets. Aquaculture International, 22(4): 1 377-1 390.
DOI:10.1007/s10499-014-9753-y |
Valle B C S, Dantas Jr E M, Silva J F X, Bezerra R S, Correia E S, Peixoto S R M, Soares R B. 2015. Replacement of fishmeal by fish protein hydrolysate and biofloc in the diets of Litopenaeus vannamei postlarvae. Aquaculture Nutrition, 21(1): 105-112.
DOI:10.1111/anu.2015.21.issue-1 |
Yao Y F, Wen H, Jiang M, Wu F, Liu W, Tian J. 2012. Dietary calcium/phosphorus ratio requirement of GIFT tilapia fingerling, Oreochromis niloticus. Journal of Northwest A&F University (Natural Science Edition), 40(4): 38-46.
(in Chinese with English abstract) |
Zidan A E N F A, Mola H R A, El-Husseiny O M, Suloma A, Mabroke R S. 2017. Inclusion of biofloc meal in tilapia diets and its effect on the structure of zooplankton community under biofloc system condition. Journal Egypt Academy Society. Environment Development, 18(1): 47-57.
|
 2019, Vol. 37
2019, Vol. 37