Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZHAN Yaoyao, ZHANG Weijie, GE Chen, LIN Kai, LI Guang, SONG Jian, CHANG Yaqing
- Relationships between body weight and other morphological traits in young sea cucumbers Apostichopus japonicas
- Journal of Oceanology and Limnology, 37(2): 759-766
- http://dx.doi.org/10.1007/s00343-019-7255-5
Article History
- Received Dec. 11, 2017
- accepted in principle Feb. 8, 2018
- accepted for publication May. 15, 2018
The commercially important Japanese sea cucumber, Apostichopus japonicus (Echinodermata: Holothuroidea: Aspidochirota: Stichopodidae), is endemic to the coasts of Russia, China, Japan, and South Korea (Chang et al., 2009). Due to the growing belief that sea cucumbers have nutritional and medicinal value (Chen et al., 2011), consumer demand for A. japonicus has increased dramatically in recent years; in China, for example, the annual aquaculture yield of A. japonicus increased from 170 kilotons in 2012 (Ministry of Agriculture of the People's Republic of China, 2013), to 193 kilotons in 2013 (Ministry of Agriculture of the People's Republic of China, 2014), to 200 kilotons in 2014 (Ministry of Agriculture of the People's Republic of China, 2015). As the scale of A. japonicus aquaculture has increased, breeding difficulties, including a degeneration of commercially important traits, an increase in individual heterogeneity, and a decrease in disease resistance, have become more prominent (Chang et al., 2006; Liu et al., 2010; Wang et al., 2015; Zhang et al., 2015; Shao et al., 2016). To combat these problems, several studies have focused on optimizing breeding programs and monitoring aquacultural practices (Jiang et al., 2009; Zhu et al., 2013; Hu et al., 2017; Liu et al., 2017).
A combination of correlation and regression analyses is considered an effective way to assess relationships among morphological traits, and to determine the contribution of various morphological traits to a given trait of interest, especially those that are economically important (Bahmani et al., 2012). Several previous studies have employed this combined method of statistical analysis to design and optimize selective breeding strategies to enhance production in aquatic organisms. For example, in the Chinese sea bass Lateolabrax maculates, body weight was most directly correlated with body width (Wang et al., 2016). In transgenic carp, body length and body height were the main predictors of body weight, while in non-transgenic carp, body weight was primarily predicted by body depth and tail length (Liu et al., 2011). In the manila clam Ruditapes philippinarum, shell height was correlated with both live body weight and edible tissue weight in 1-year old clams; in 2- to 3-year old clams, shell width was correlated with live body weight and shell length was correlated with edible tissue weight (Huo et al., 2010). In the marine gastropod Glossaulax reiniana, operculum height was correlated with body weight and soft-tissue weight (Zhao et al., 2014). In the bivalve Dosinia japonica, body mass was correlated with shell size (Zhang et al., 2013).
In A. japonicus, it has been shown that key growth traits (including body weight) are influenced by genetic factors, such as the paternal and maternal effects, and their interactive heritability (Zhang et al., 2015). However, the relationships among the morphological traits of A. japonicus at different developmental stages are unclear, as are the impacts of morphological variation on economically important traits (i.e. body weight) at different developmental phases.
We therefore aim (1) to explore the relationships among key morphological traits of A. japonicus in early growth stages; (2) to identify morphological traits that might effectively be used as selection indicators in A. japonicus breeding programs; and (3) to determine the morphological variable most reliably correlated with body weight in the early development of A. japonicus. To do this, we analyzed the correlations among four morphological traits, and performed a multiple regression analysis.
2 MATERIAL AND METHOD 2.1 Experimental animals and maintenanceA broodstock of A. japonicus was obtained from a cultured population in Zhuanghe, Dalian, China (122.97°E, 39.7°N) in April 2016. The broodstock was transported to the Key Laboratory of Mariculture & Stock Enhancement in the Ministry of Agriculture and Rural Affairs of the North China Sea at Dalian Ocean University, Dalian, China. All specimens were kept in 1 000-L recirculating seawater tanks (15 specimens per tank) at room temperature (15–16℃), with a flow rate of 4 500 L/h. All tanks were illuminated by less than 500 lx incandescent light, with a 10 h–12 h light: 12 h–14 h dark photoperiod. Specimens were fed 3%–5% of their total body weight with a nutrient mix (40% w/w dry sea mud, 30% w/w Sargassum thunbergii powder, and 30% w/w artificial sea cucumber feed (25% w/w Ulva lactuca powder, 10% w/w Laminaria japonica powder, 20% w/w oyster shell powder, 10% w/w anchovy fishmeal, 10% w/w corn gluten meal, and 25% w/w yeast powder; Shangdong Anyuan Aquatic Product Co. Ltd., China)), 1–2 times a day.
2.2 Spawning and fertilizationWe placed 50 healthy A. japonicus specimens (average mass ~300 g), all with well-developed gonads, in a 100-L plastic bucket (50.5 cm diameter×50.0 cm deep) filled with fresh filtered seawater (FSW; filtered at 28 μm) at 20℃. We then induced spawning in these specimens, following the methods of Song et al. (2009) and Chang et al. (2004). Briefly, all sea cucumbers were removed from the bucket, dried outdoors in the shade for 30 min (temperature 20℃; humidity approximately 50%–60%), and replaced in the recirculating tanks for 10–20 min. The water temperature in the tanks was then increased approximately 3℃.
Gametes from 5 males and 5 females were collected to ensure that gametes from multiple parents were mixed. Prior to mixing, we checked the quality of the gametes microscopically: eggs were checked for shape and appearance, and sperm were checked for motility.
Gametes were mixed in a 100-L FSW-filled tank at 20℃, with a ratio of five sperm to each visible ovum (Chang et al., 2004). FSW in the tank was stirred by hand for 1–2 min once per h for 12 h until the fertilized ova hatched into larvae. After 12 h, we gently removed the hatched larvae and transferred them to 100 L FSW-filled tanks at 20℃.
2.3 Egg hatching and larval rearingWe used the methods of Chang et al. (2004) to hatch the fertilized eggs and rear the larvae. The initial larval density was 0.5–1 larvae/mL. The FSW in the larval tank was maintained at 20.0±4℃ (mean±SD; n=30), with a salinity of 30.5±0.5 (n=30), a pH of 8.0±0 (n=30), and a dissolved oxygen concentration of 7.1±1.2 mg/L (n=30). The larval tank was illuminated by less than 500 lx in candescent light, with a 10 h–12 h light: 12 h–14 h dark photoperiod. Hatched larvae were fed (0.7–1.0)×104 cells/mL Chaetoceros muelleri three times a day, and FSW was completely replaced twice a day. Once larvae metamorphosed into juveniles, sea cumbers were fed 3%–5% of the total body weight of the above nutrient mix, 1–2 times a day. FSW was completely replaced once a day, and the plastic plates used as substratum were completely replaced once a month.
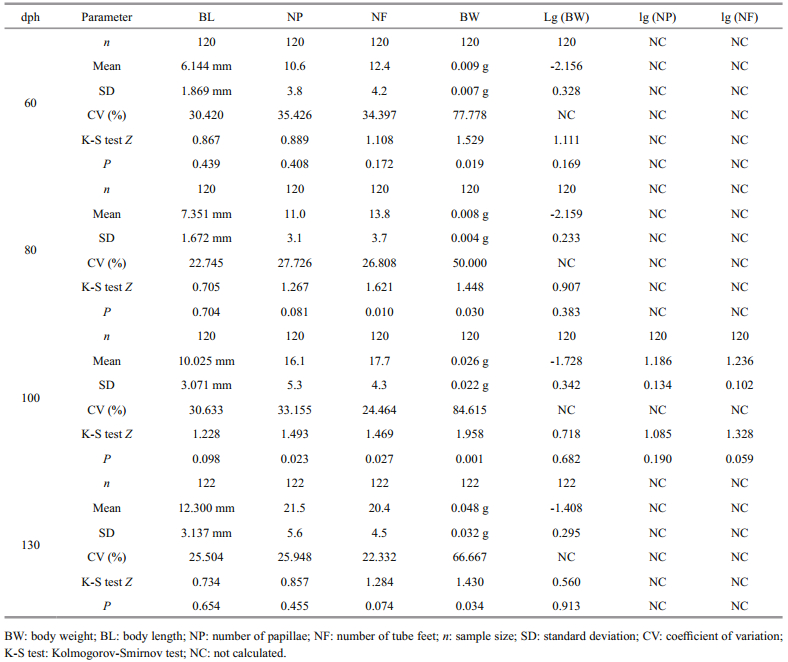
2.4 Trait measurementsWe randomly selectefed 120 A. japonicus specimens for trait measurement at 60, 80, and 100 days posthatching (dph); 122 specimens were randomly selected at 130 dph. Each specimen was placed in a separate, sterile Petri dish. We used vernier calipers (accuracy: 0.01 mm) to measure body length (BL) of each specimen following the methods of Wei et al. (2007). In brief, each specimen was starved for 24 h, and then soaked in 0.5 mol/L MgSO4 at room temperature for about 1.5 h until the specimen straightened naturally. Body length of the straightened specimens was measured. After measurement, specimens were allowed to recover in normal seawater for 5 to 8 h. We counted papillae (NP) and tube feet (NF) under a 5× magnifier. Finally, to measure live body weight (BW), specimens were dried on paper towels and weighed with an electronic balance (accuracy: 0.000 1 g). To avoid user bias, the same researcher measured all specimens.
2.5 Data analysisWe used Excel 2008 (Microsoft, USA) to calculate the mean, the standard deviation (SD), and the coefficient of variation (CV) of each measured trait across all specimens within each age group. Before pathway and regression analyses, we used the Kolmogorov-Smirnov (K-S) test to test whether our data were normally distributed. The K-S test indicated that BW was non-normally distributed across all age groups, as were NF at 80 dph, NF at 100 dph, and NP at 100 dph. We therefore converted BW, NF, and NP values to base-10 logarithms (lg). After lg conversion, all traits, with the exception of NF at 80 dph, were normally distributed.
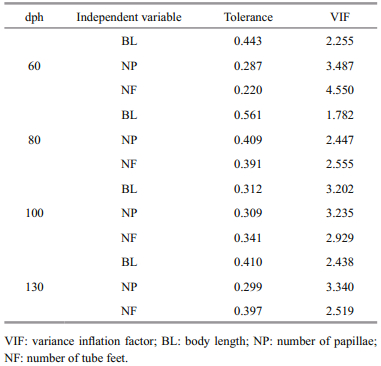
We next used correlation analysis to determine the correlations between BW and the other morphological traits. As there was no collinearity among the morphological traits we measured (Table 1), we used a multiple regression analysis to determine the path (standardized regression) coefficients between the measured morphological traits and BW. We then used stepwise multiple regression to identify the regression equations that best predicted BW, with BW as the dependent variable and each measured morphological trait as an independent variable. By gradually removing non-significant morphological variables, we generated multiple regression equations for BW.
|
We calculated the single-trait determination coefficients in SPSS for Windows v16 (SPSS Inc., USA) with the following equation:
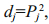
where Pj is the direct path coefficient of a single trait. We calculated the co-determination coefficient of both i and j on body weight in SPSS for Windows v16 (SPSS Inc., USA) with the following equation:
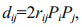
where rij is the correlation coefficient between i and j, Pi is the corresponding direct path coefficient of i, and Pj is the corresponding direct path coefficient of j. We considered P < 0.01 statistically significant.
3 RESULT 3.1 Variations in morphological traitsThe mean and SD of each measured trait increased with specimen age (Table 2). The CVs of BW were moderate to high across all four age groups (CV for all age groups ≥50%; Table 2). In addition, the CV for BW was greater than the CVs of all other morphological traits in each age group; NP had the next highest CV across all age groups. In the 60- and 80-dph groups, CV was lowest for BL, while in the 100- and 130-dph groups, CV was lowest for NF. For NP and NF, the 60-dph group had the highest CV, while for BL and BW, the 100-dph group had the highest CV.
|
The correlations between all pairs of phenotypic traits that we measured were significant (P < 0.01; Table 3). NP and NF were highly correlated in A. japonicus specimens of all ages (60 dph, R=0.844; 80 dph, R=0.749; 100 dph, R=0.769; 130 dph, R=0.767; Table 3); NF and BL were moderately well correlated across all age groups, while BL was most highly correlated with BW in specimens at 100 dph (R=0.923) and 130 dph (R=0.867; Table 3). The correlation between NP and NF did not vary substantially among the different age groups (Table 3).
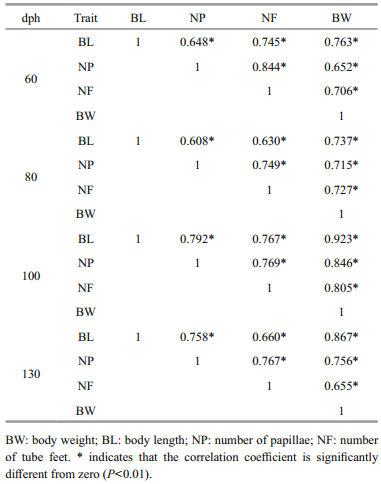
|
BL had the greatest effect on BW across all age groups of A. japonicus (Table 4). In the 60- and 80- dph groups, NF had a stronger influence than NP; in the 100- and 130-dhp groups, the reverse was true (Table 4). The correlation between the measured morphological traits and BW was high across all age groups (R2>0.6 for all age groups; Table 4).
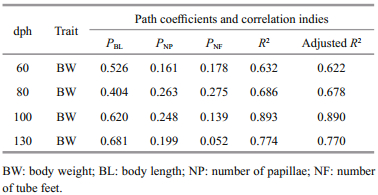
|
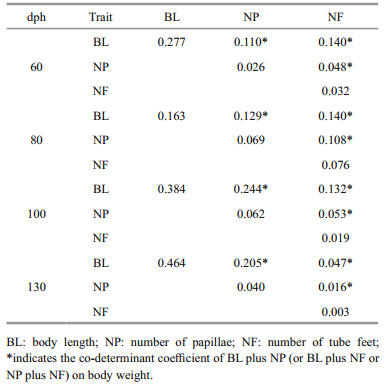
The correlation between morphological traits and BW (rij) was separated into the direct effects of a single trait (Pi) and the indirect effects of other traits (rijPj) (Table 5). The direct effect of BL was highly correlated with BW across all age groups, but the indirect effects of the measured morphological traits varied (Table 5). rijPj was greatest between BL and NF in specimens 60 dph (0.392) and 80 dph (0.255); rijPj was greatest between BL and NP in specimens 100 dph (0.491) and 130 dph (0.517).
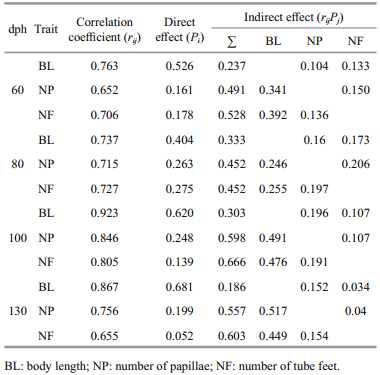
|
BL had the highest coefficient of determination across all age groups with respect to BW (R2>0.16; Table 6). In 60- and 80-dph specimens, the coefficient of determination with respect to BW was higher for NF than NP; in 100- and 130-dph specimens, the reverse was true (Table 6). Our stepwise regression analysis recovered the following equations describing BW at 60, 80, 100, and 130 dph:
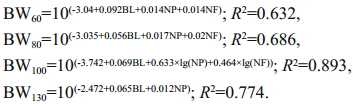
Herein, we present a novel analysis of the relationship between BW and other morphological traits in juvenile specimens of A. japonicus. Across the four juvenile age groups we measured (60, 80, 100, and 130 dph), the CV of BW was high (greater than 50%), indicating that BW might be useful for selection. The CVs for the three additional morphological traits we measured varied with juvenile age. That is, the CVs for NP and NF were highest in the 60-dph specimens, but the CV for BL was highest at 60 dph and 100 dph (Table 2). The CV for NF decreased steadily with increasing age, while the CVs for NP and BL fluctuated (Table 2). However, as the CVs for all measured traits remained high across all age groups, our results suggested that BL, NP, and NF were also potential target traits for the artificial selection of juvenile A. japonicus. In general, high correlations between morphological traits tend to be observed in marine invertebrates with relatively fixed body shapes, such as bivalves (Liu et al., 2002), crustaceans (Liu et al., 2004), and sea urchins (Zhang et al., 2010). However, a relatively low correlation between morphological measurements and BW was found in Octopus variabilis, a soft-bodied invertebrate (Song et al., 2012). This discrepancy between softand hard-bodied invertebrates might be because correlation analyses depend heavily on the accuracy of morphological trait measurements, and it is difficult to measure soft and easily deformed organisms, such as cephalopods and holothurians, exactly. Surprisingly, in contrast to the results reported for O. variabilis, we found high correlations among most of the morphological traits that we measured (Table 3). This suggests that our measurements of juvenile A. japonicus specimens were more accurate than those of adult O. variabilis specimens, probably because the elasticity and flexibility of adult O. variabilis is greater than that of juvenile A. japonicus. In addition, these high phenotypic correlations suggested that growth in the juvenile A. japonicus might follow certain predictable patterns, allowing for the development of efficient selective breeding programs.The high correlation between NP and NF that we observed (R=0.749–0.844; Table 3) was consistent with the external morphological characters of A. japonicus, because the pentamerous symmetry of A. japonicus is always recognized by the presence of five meridional ambulacra bearing podia (Purcell et al., 2012). In addition, although both tube feet and papillae are of ectodermic origin, tube feet are locomotory podia found on the ventral surface of A. japonicus, while papillae are papillate podia on the dorsal surface (Purcell et al., 2012). NP and NF were also well correlated with BL, probably because papillae and tube feet are evenly distributed along the entire length of A. japonicus, and thus their numbers must increase with body length.
|
Path analysis has been widely used in the development of breeding programs to identify the direct effects of one trait on another (Yücel et al., 2004; Zhang et al., 2011). When we compared single direct effects with multiple indirect effects on BW, we found that the direct effect of BL was greater than the combined indirect effects of NP and NF (Table 5). Indeed, the differences between the direct effects of BL and the indirect effects of NP or NF on BW were much greater than the differences among the corresponding correlation coefficients (Table 5). This might be because the correlation of NP and NF with BW also included the indirect effects of BL on BW.
The coefficient of determination of BL on BW was greater than the coefficient of determination of NP on BW or of NF on BW across all age groups, despite fluctuations in coefficient values depending on age group (Table 6). Indeed, in the 130-dph specimens, the effect of BL on BW was 12 times that of NP, and 155 times that of NF (Table 6). It may be that variations in individual body shape caused the substantial differences that we found in the effects of BL, NP, and NF on BW. Such heterogeneous development is common in sea cucumber cultivation (Liu et al., 2015).
The greater influence of NP and NF on BW in 80- dph specimens, as compared to older specimens (Table 6), might be because the papillae and the tube feet grow rapidly at younger developmental stages (Chang et al., 2004), and these traits might thus have had more influence on BW at younger ages. After 100 dph, the reduction in the influence of NP and NF (Table 6) might be because other morphological developments, such as the thickening of the body wall, the enlargement of the body cavity, and the increase in coelomic fluid, might be having more effect on BW than NP or NF.
NF had a greater influence on BW than NP at younger juvenile ages, while NP had a greater influence on BW at older juvenile ages (Tables 5 and 6). This result might be due to a weight difference between papillae and tube feet: it is possible that, in older juveniles, papillae weigh more than tube feet. This supposition is consistent with the external morphology of adult A. japonicus: in adults, papillae are larger than tube feet (Purcell et al., 2012).
It is generally believed that independent variables are significant only if the correlation index of the path analysis is high (R2≥0.85; Zhao et al., 2016). By this metric, the only significant variable with respect to BW in juvenile A. japonicus is BL at 100 dph (Table 4), indicating that BL at 100 dph may be a useful trait for selection. For 60-, 80-, and 130-dph A. japonicus specimens, although BL was relatively well correlated with BW, correlation indices were only moderate (R2 < 0.78), indicating that other factors besides BL might be associated with BW at those ages.
In the aquaculture of A. japonicus, juvenile weight measurements are often inconvenient or inaccurate (Liu et al., 2015). The optimal regression equations reported herein provide a new way to estimate the body weight of A. japonicus juveniles at different ages using more easily and accurately measured morphological traits.
5 CONCLUSIONWe investigated the relationships between body weight and various morphological traits of juvenile Japanese sea cucumbers (A. japonicus), using correlation, multiple regression, and path analyses. The four morphological traits we measured (BW, BL, NP, and NF) were highly correlated. In 100-dph A. japonicus, BL had a significant direct effect on BW (R2>0.89), indicating that, in specimens of this age, BL may be a useful trait for selection. In juvenile A. japonicus specimens at 60, 80, and 130 dph, other morphological traits besides BL might be associated with BW. The optimal regression equations that we constructed for juvenile A. japonicus specimens at 60, 80, 100, and 130 dph provide a framework for the successful aquaculture and selective breeding of A. japonicus.
6 DATA AVAILABILITY STATEMENTThe datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
7 AUTHORS' CONTRIBUTIONY. Q. C., Y. Y. Z., and W. J. Z. conceived and designed the experiments. Experiments were performed by C. G, G. L., and J. S. Data were analyzed by C. G., W. J. Z., Y. Y. Z., and K. L. The paper was written by Y. Y. Z., W. J. Z., and K. L. All authors read and approved the manuscript.
Bahmani K, Izadi-Darbandi A, Noori S A S, Jafari A A, Moradi N. 2012. Determination of interrelationships among phenotypic traits of Iranian fennel (Foeniculum vulgare Mill.) using correlation, stepwise regression and path analyses. Journal of Essential Oil Bearing Plants, 15(3): 424-444.
DOI:10.1080/0972060X.2012.10644070 |
Chang Y Q, Ding J, Song J, Yang W. 2004. Biology and Aquaculture of Sea Cucumbers and Sea Urchins. China Ocean Press, Beijing, China.
(in Chinese) |
Chang Y Q, Feng Z G, Yu J P, Ding J. 2009. Genetic variability analysis in five populations of the sea cucumber Stichopus(Apostichopus) japonicus from China, Russia, South Korea and Japan as revealed by microsatellite markers. Marine Ecology, 30(4): 455-461.
|
Chang Y Q, Sui X L, Li J. 2006. The current situation, problem and prospect on the Apostichopus japonicus aquaculture. Fisheries Science, 25(4): 198-201.
(in Chinese with English abstract) |
Chen S G, Xue C H, Yin L A, Tang Q J, Yu G L, Chai W G. 2011. Comparison of structures and anticoagulant activities of fucosylated chondroitin sulfates from different sea cucumbers. Carbohydrate Polymers, 83(2): 688-696.
DOI:10.1016/j.carbpol.2010.08.040 |
Hu Y, Zhou C S, Yu H, Lu R M, Fang J, Tang M, Hu L H. 2017. Effect of density, size and shelter type on growth and growth coefficient of Apostichopus japonicus in indoor cement pond. Journal of Aquaculture, 38(7): 10-14.
(in Chinese with English abstract) |
Huo Z M, Yan X W, Zhao L Q, Zhang Y H, Yang F, Zhang G F. 2010. Effects of shell morphological traits on the weight traits of Manila clam (Ruditapes philippinarum). Acta Ecologica Sinica, 30(5): 251-256.
DOI:10.1016/j.chnaes.2010.08.004 |
Jiang S G, Chang Y Q, Tian Y, Song J, Ding J, Zhao C. 2009. Crossbreeding of different populations of sea cucumber Apostichopus japonicus. Journal of Dalian Fisheries University, 24(3): 247-250.
(in Chinese with English abstract) |
Liu C L, Chang Y M, Liang L Q, Hu L H, Liu J L, Chi B J, Wu X G. 2011. Correlation and path analyses of phenotypic traits and body mass of transgenic carp with growth hormone gene of salmon. Chinese Journal of Applied Ecology, 22(7): 1893-1899.
(in Chinese with English abstract) |
Liu H Z, Zheng F R, Sun X Q, Hong X G, Dong S L, Wang B, Tang X X, Wang Y Q. 2010. Identification of the pathogens associated with skin ulceration and peristome tumescence in cultured sea cucumbers Apostichopus japonicus(Selenka). Journal of Invertebrate Pathology, 105(3): 236-242.
DOI:10.1016/j.jip.2010.05.016 |
Liu H, Xu Q, Liu S L, Zhang L B, Yang H S. 2015. Evaluation of body weight of sea cucumber Apostichopus japonicus by computer vision. Chinese Journal of Oceanology and Limnology, 33(1): 114-120.
DOI:10.1007/s00343-015-4080-3 |
Liu R, Wu X, Wang C, Cui L B. 2017. Characteristics in sea cucumber Apostichopus japonicus culture ponds with three different sediments. Fisheries Science, 36(1): 29-35.
(in Chinese with English abstract) |
Liu X L, Chang Y Q, Xiang J H, Song J, Ding J. 2002. Analysis of effects of shell size characters on live weight in Chinese scallop Chlamys Farreri. Oceanologia et Limnologia Sinica, 33(6): 673-678.
(in Chinese with English abstract) |
Liu X L, Wu C G, Zhang Z H, Huang H, Li B, Zhang Y F, Sun C B, Xiang J H. 2004. Mathematical analysis of effects of morphometric attributes on body weight of Penaeus vannamei. Acta Ecologica Sinica, 24(4): 857-862.
(in Chinese with English abstract) |
Ministry of Agriculture of the People's Republic of China. 2013. China Fishery Statistical Yearbook 2013. China Agriculture Press, Beijing. 29pp. (in Chinese)
|
Ministry of Agriculture of the People's Republic of China. 2014. China Fishery Statistical Yearbook 2014. China Agriculture Press, Beijing. 29pp. (in Chinese)
|
Ministry of Agriculture of the People's Republic of China. 2015. China Fishery Statistical Yearbook 2015. China Agriculture Press, Beijing. 29pp. (in Chinese)
|
Purcell S W, Samyn Y, Conand C. 2012. Commercially Important Sea Cucumbers of the World. Commercially Important Sea Cucumbers of the World. p2.
|
Shao Y, Li C H, Zhang W W, Wang Z H, Lv Z M. 2016. The first description of complete invertebrate arginine metabolism pathways implies dose-dependent pathogen regulation in Apostichopus japonicus. Scientific Reports, 6: 23783.
DOI:10.1038/srep23783 |
Song J, Pang Z G, Chang Y Q, Zhao C. 2009. The method of inducing Apostichopus japonicus spawning in vivo. CHN: ZL 200910012325.0. (in Chinese)
|
Song J, Xiao D B, Zhang W J, Chang Y Q, Gong H N. 2012. The study of correlativity between Octopus variabilis body measurement and weight traits. Chinese Agricultural Science Bulletin, 28(23): 118-122.
(in Chinese with English abstract) |
Wang H H, Zhang W W, Li C H, Lv Z M, Jin C H. 2015. Identification and characterization of a novel Foxo transcription factors in Apostichopus japonicus. Fish & Shellfish Immunology, 44(1): 164-171.
|
Wang W, Ma C Y, Chen W, Ma H Y, Zhang H, Meng Y Y, Ni Y, Ma L B. 2016. Optimization of selective breeding through analysis of morphological traits in Chinese sea bass(Lateolabrax maculatus). Genetics and Molecular Research, 15(3).
DOI:10.4238/gmr.15038285 |
Wei J, Chang Y Q, Nie Z L, Ding J, Song J, Cao X B. 2007. The method for accurate measurement of the live body length of Apostichopus japonicus. CHN: CN200710010174.6 Aug 2007. (in Chinese)
|
Yücel C. 2004. Correlation and path coefficient analyses of seed yield components in the Narbon Bean (Vicia narbonensis L.). Turkish Journal of Agriculture & Forestry, 28(5): 371-376.
|
Zhang S C, Yang H S, Liu S L, Zhang L B, Ru X S, Xu D X. 2015. The influence of genetics factor on key growth traits and quantitative genetic analysis of sea cucumber Apostichopus japonicus (Selenka) heat-resistant and fastgrowing strain. Aquaculture International, 23(1): 219-233.
DOI:10.1007/s10499-014-9810-6 |
Zhang W J, Chang Y Q, Ding J, Xiao D B, Yu J Y, Zhou H S. 2013. Correlation and regression of shell size and weight of Dosinia japonica Reeve. Oceanologia et Limnologia Sinica, 44(3): 796-800.
(in Chinese with English abstract) |
Zhang W J, Chang Y Q, Zhao C, Liu P J, Song J. 2010. Effects of phenotypic traits on gonad traits in sea urchins Strongylocentrotus intermedius. Progress in Fishery Sciences, 31(3): 49-55.
(in Chinese with English abstract) |
Zhang W J, Zhao C, Liu X J, Luo SB, Chang Y Q. 2011. Evaluation of morphology traits to predict weight traits in the razor clam solen strictus. In: Proceedings of the 2011 5th International Conference on Bioinformatics and Biomedical Engineering. IEEE, Wuhan, China.
|
Zhao L Q, He Y Y, Yang F, Nie H T, Yan X W. 2014. Correlation and path analysis of morphological and weight traits in marine gastropod Glossaulax reiniana. Chinese Journal of Oceanology and Limnology, 32(4): 821-827.
DOI:10.1007/s00343-014-3290-4 |
Zhao Y Y, Zhu X C, Li Z, Jiang Y, Han Z B, Xu W B, Wang S Y, Wei H, Dong J, Bai W L, Li X D. 2016. Correlation analysis between body weight and morphometric traits of Palaemonetes sinensis. Journal of Shenyang Agricultural University, 47(6): 681-686.
(in Chinese with English abstract) |
Zhu L, Li C H, Su X R, Guo C Y, Wang Z, Jin C H, Li Y, Li T W. 2013. Identification and assessment of differentially expressed genes involved in growth regulation in Apostichopus japonicus. Genetics and Molecular Research, 12(3): 3028-3037.
DOI:10.4238/2013.August.20.4 |
 2019, Vol. 37
2019, Vol. 37


