Institute of Oceanology, Chinese Academy of Sciences
Article Information
- LIU Qi, LI Wenjun, LU Lina, LIU Bin, DU Zhenning, QIN Song
- Phycocyanin attenuates X-ray-induced pulmonary inflammation via the TLR2-MyD88-NF-κB signaling pathway
- Journal of Oceanology and Limnology, 37(5): 1678-1685
- http://dx.doi.org/10.1007/s00343-019-8196-8
Article History
- Received Aug. 15, 2018
- accepted in principle Nov. 10, 2018
- accepted for publication Jan. 3, 2019
2 School of Pharmacy, Yantai University, Yantai 264005, China;
3 School of Nuclear Science and Technology, Lanzhou University, Lanzhou 730000, China
Lungs are highly sensitive to ionizing radiation and are more vulnerable than other organs (Demirel et al., 2016). The most common radiation toxicities are pneumonitis and fibrosis, known as radiation-induced lung injury (Shen et al., 2013). In recent years, radiation therapy has been the main treatment for lung tumors, so increasing numbers of patients are receiving radiotherapy. An increase in the radiation dose enhances the therapeutic effect, but the side effects of radiation also increase which limits the possible rise in dose (Li, 2015). The rate of radiation pneumonitis after chest irradiation was reported to be approximately 15% and can be fatal in some cases (Mehta, 2005). Although the risk of severe radiation pneumonitis can be reduced by controlling the dose or reducing the volume of the lung body exposed to radiation (Mehta, 2005), this weakens the treatment effect. Therefore, as an alternative, radiation protection drugs have become a research hotspot. Current anti-radiation drugs include aminoguanidines, superoxide dismutases, nitrogen oxides, cytokines, and immunomodulators. However, most of these are associated with side effects or only have single targets. Because ideal radiation protection drugs do not exist (Hosseinimehr, 2007), there is an urgent need to develop a safe and effective radiation protective agent.
Phycocyanin (PC) is extracted mainly from the cyanobacterium Spirulina platensis and is a watersoluble pigment protein complex (Wu et al., 2016). PC has good anti-oxidative (Li et al., 2016) and antiinflammatory effects (Chang et al., 2011), and free radical scavenging characteristics. It was also shown to remove hydrogen peroxide, hydroxyl radicals, and superoxide radicals. Additionally, PC inhibited the lipid peroxidation induced by carbon tetrachloride in the rat liver and exhibited anti-inflammatory properties by inhibiting cyclooxygenase-2 (Bhat and Madyastha, 2000). PC may become a drug candidate for the treatment of radiation pneumonitis.
Toll-like receptor (TLR) is an important pathogen recognition receptor, and TLR2 plays a crucial role in the pathogenesis of various lung diseases (Liu et al., 2010). Myeloid differentiation factor MyD88, a key connector molecule in the TLR signaling pathway, transmits upstream information and is involved in disease development. The nuclear factor NF-κB family is widespread in mammalian cells and is a vital nuclear transcription regulator. It binds to the B sequence of promoters and activates a variety of transcription factors that promote cell growth or apoptosis. Indeed, the expression of many cytokines depends on activation of the TLR-induced NF-κB pathway (Carmody and Chen, 2007). Kim et al. found that the BLM-induced pulmonary inflammation reduced when the TLR2 was blocked (Kim et al., 2011). Li et al. (2017) indicated that the PC alleviates pneumonia and pulmonary fibrosis by blocking TLR2 signaling pathway. These studies suggest that pneumonia induced be radiation may be alleviated by inhibiting the TLRs signaling pathway.
Based on the above studies, PC can control inflammation and have an impact on the TLR pathway. However, the effect of PC treatment on radiationinduced lung injury has not been studied. We hypothesized that PC has preventive and therapeutic effects on radiation-induced lung injury and would alleviate lung inflammation induced by X-rays through blocking the TLR2-MyD88-NF-κB signaling pathway.
2 MATERIAL AND METHOD 2.1 AnimalMale C57BL/6 mice were purchased from the Pengyue Co. Ltd. (Jinan, China Animal license number: SCXK(Lu)2014-007). There is 1 week of acclimation before animal experiments and then performed according to protocols in accordance with institutional guidelines. The mice were housed in an environmentally controlled room at 23±2℃ under a 12 h:12 h light-dark cycle with free access to water and food.
2.2 ChemicalThe following reagents were purchased from the indicated sources: Phycocyanin (Xindaze Spirulina Co. Ltd., China), and the purity (OD620/280) of PC is 3.7, BCA kit (Nanjing Jiancheng Bioengineering, China), LPS ELISA kit (Mlbio Bioengineering, China) RNA and RT-PCR kit (TaKaRa, Japan); TLR2 rabbit mAb (CST, USA); NF-kB rabbit mAb (CST, USA); and Myd88 rabbit mAb (SAB, USA); HRPconjugated secondary antibody (Proteintech, USA). All other reagents were of analytical grade.
2.3 Experimental design32 C57 BL/6 male mice were randomly divided into normal control group (Control group), irradiation model group (TBI group), PC pretreatment group (these 6 mice were gavaged with 200 mg/kg PC for 7 consecutive days before irradiation, PC+TBI group) and PC treatment group (6 mice were gavaged 200 mg/kg PC consecutive 7 days after irradiation, TBI+PC group). On the 8th day of intragastric administration, except for the normal control group, the rest of the mice were subjected to total body irradiation to a dose of 6 Gy for the preparation of radiation damage model. The plasma and tissues were collected 7 days after irradiation.
2.4 Histological assessmentThree mice from each group were sacrificed for lung histology analysis. After the mice were anesthetized, the left-lung of the mice was taken out and collected, then these lungs of the mice were fixed with a 4% paraformaldehyde solution, respectively. The lungs are then removed and placed in a fixative as previously described. The fixed lung tissue was processed into paraffin. The 4 μm sections were prepared for analysis of histological changed. Paraffin sections were first decolorized with xylene, then dehydrated using different concentrations of alcohol, and finally, histomorphology was observed by hematoxylin and HE staining.
2.5 PC inhibits radiation-induced cytokine release.Lung tissues were removed and weighed, a ninefold volume of normal saline was added and the tissues were homogenized using an ultrasonic. And samples were centrifuged at 1 200 r/min at 4℃ for 15 min, and the supernatants were used for detecting IL-6 and TNF-α and LPS levels. The total protein concentration in lung tissue was determined by BCA Protein Assay Kit (Solarbio, Beijing, China). The concentration of cytokines such as IL-6 and TNF-α was determined by ELISA using the Mlbio kit.
2.6 Fluorescence microscopy of in vivo ROSReactive oxygen species (ROS) oxidizes dihydroethidium (DHE) to produce fluorescent ethidium that subsequently binds to nucleic acids, resulting in nuclei stained with bright red fluorescence. Therefore, DHE is used as a fluorescent probe for detecting intracellular ROS (Rong et al., 2012). Frozen lung was cut into 5-μm sections, then 5 mmol/L DHE (Beyotime Institute of Biotechnology, Haimen, China) was added and incubated at 37℃ for 30 min. After appropriate washing, the fluorescence intensity of tissue sections was observed under a fluorescence microscope. The maximum excitation wavelength of DHE is 370 nm, and its maximum emission wavelength is 420 nm, while the maximum excitation wavelength of red fluorescence produced after dehydrogenation of DHE combined with DNA and RNA is 370 nm, and its maximum emission wavelength is 600 nm. For actual observations, 535 nm can be used as the excitation wavelength. Three samples from each group were examined by fluorescence microscopy, and the mean fluorescence density (MFD) was estimated as the fluorescence intensity per unit sphere as assayed by Image-ProPlus 6.0 software (Media Cybernetics, Rockville, MD).
2.7 Real-time reverse transcription-polymerase chain reaction (RT-PCR)Firstly, the total RNA of lung tissue was extracted by Trizol routine one step method. The OD values of A260 and A280 were measured by ultraviolet spectrophotometer respectively, and the value of RNA A260/A280 in each group was 1.8–2.0. Quantitative real-time RT-PCR using SYBR Premix Ex Taq probes and primers (TaKaRa, Dalian, China) was used for gene expression analyses as described previously. According to the instructions of the kit, the genomic DNA reaction and the reverse transcription reaction were carried out to obtain cDNA respectively. Takara Gene Expression Assay Reagents for mouse SOD, GSH-PX, TLR2, Myd88, and NF-kB, glyceraldehydes-3-phosphate dehydrogenase (GAPDH) were used for specific probes and primers of PCR amplifications. GAPDH used as a reference. The threshold cycle (CT) was determined, the comparative CT method was used to calculate relative quantification. Primer sequences TLR2, Myd88, and NF-κB are presented in Table 1. Target gene expression was normalized to gene expression.
Lung tissues were prepared using RIPA buffer (Suolaibao) with 10% protease inhibitors and protein concentrations measured by BCA kit (Nanjing Jiancheng). In order to analize the TLR2, Myd88, and NF-Kb expression, 30-μg protein samples were loaded on 8%, 10%, 12% SDS-PAGE respectively followed by transfer of protein onto a polyvinylidene fluoride membrance (100 V, 75 min). At room temperature, the membrane was placed in 5% skim milk for 4 h, then incubated overnight at 4℃ with primary antibodies including TLR2 (1:1 000), Myd88 (1:1 000), NF-kb (1:1 000) GADPH (1:1 000). Finally, the membrane was incubated with the secondary antibody (1:5 000) for 2 h at room temperature and an enhanced chemiluminescent reagent was used. Membranes were observed using a ChemiScope Mini 3100 (Clinx, China), and the relative expression of the various protein to GADPH was analized by ImageJ 5.0 software.
2.9 Statistical analysisThe analysis was performed using software SPSS19. The data obtained are expressed as mean±SD. If the P value is less than 0.05 or 0.01, the difference between the groups is considered to be statistically significant. Comparisons between groups were performed using the test of homogeneity of variances and one-way analysis of variance (ANOVA).
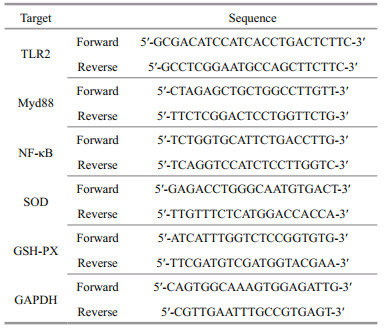
3 RESULT 3.1 PC attenuated radiation induced histological changes in miceLung injury was analyzed by HE staining (Fig. 1). After HE staining, the alveolar wall of the lung tissue of the control group was thin and the structure was clear. On the 7th day after irradiation, the radiation group showed changes of acute interstitial pneumonitis. The main manifestations of the lesions were exudation, alveolar epithelial hyperplasia, alveolar septum widening, partial alveolar cavities fusion, and small blood vessel congestion and dilation. The pathological changes of edema, hyperplasia, and hyperemia in the PC prevention group and the treatment group were significantly reduced compared with the model group. The quantitative histological of pneumonia changes are analyzed depending on Ashcroft scale (Arrieta et al., 2009). According to A's research, the scoring criteria are as follows: 0, normal; 1–2, alveolar walls slightly broadening, < 10 inflammatory cells under high power field of vision; 3–4, alveolar walls significantly broadening, 10–20 inflammatory cells under high power field of vision; 5–6, alveolar walls significantly broadening, inflammatory cells infiltration full of alveolar walls. Through pathological analysis, the inflammation score of groups TBI was higher than that of PC prevention group and PC treatment group (Fig. 2). This evidence confirms that PC can reduce pneumonia caused by radiation.
|
| Fig.1 Effect of PC on hepatic histopathological changes of radiation induced lung injury in mice HE stain, magnification 400×. |

|
| Fig.2 PC attenuated X-ray induced histological changes in mice Lung tissue sections of the control group, TBI group, PC+TBI group and TBI+PC group in 7 days after radiation separately. The severity of lung injury was analyzed by the related scoring system. Each bar represents the mean±SD of 6-8 mice. * represents a significant difference compared with the TBI (P < 0.05); ** represents an extremely significant difference compared with the TBI (P < 0.01); # represents a significant difference compared with the Control (P < 0.05); ## represents an extremely significant difference compared with the Control (P < 0.01). |
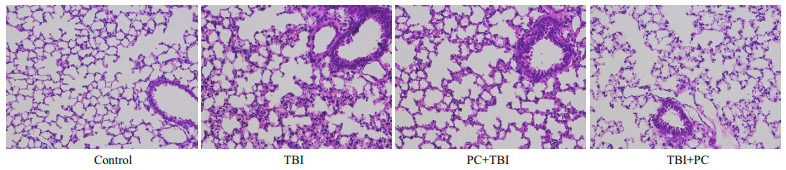
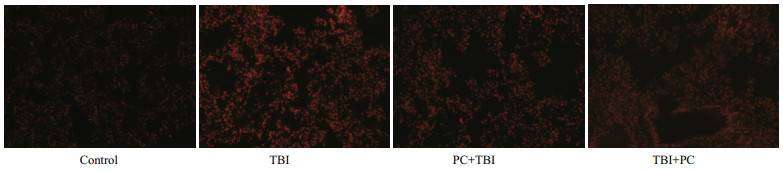
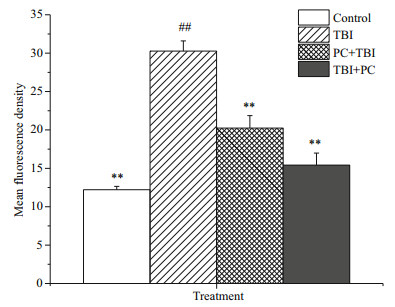
The defense response of the living tissue with the vascular system to the injury factor is inflammation. Inflammation occurs with oxidative stress processes caused by the accumulation of reactive oxygen species (ROS). Protective effect of PC on lung tissue in radioactive ROS environment is shown in Fig. 3. X ray exposure enhanced the red fluorescence which implied increase ROS content in the lung, but much less can be observed in the lung of mice in PC+TBI and TBI+PC groups. The MFD (Mean Fluorescence Density) in PC supplementation significantly decreased as compared to the TBI group both on days 7 after irradiation exposure (Fig. 4).
|
| Fig.3 Effect of PC on radiation induced ROS generation in the liver Fluorescence was monitored using an OLYMPUS fluorescence microscope equipped with a fluorescein isothiocyanate filter. |
|
| Fig.4 Fluorescence intensity in control, TBI, PC+TBI, TBI+PC lung Values obtained from 3 independent experiments and presented as mean±SD. * represents a significant difference compared with the TBI (P < 0.05); ** represents an extremely significant difference compared with the TBI (P < 0.01); # represents a significant difference compared with the Control (P < 0.05); ## represents an extremely significant difference compared with the control (P < 0.01). |
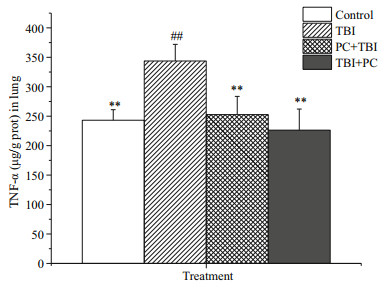
According to the researches, IL-6 and TNF-α are now considered to be important mediators of radiation-induced pneumonitis. Inflammatory signaling pathway of nuclear factor kappa B (NFkappa B) is activated by irradiation to normal lung tissue activates the, after then it promotes the release of IL-6 and TNF-α secreted by inflammatory cells such as macrophages and lymphocytes, resulting in the expansion of inflammatory cascade reaction. In this study, we detected the concentration of IL-6 and TNF-α in the lung of mice. The results showed that the IL-6 and TNF-α contents of mice in the radiation group increased significantly (Figs. 5, 6). It suggests that radiation induces lung inflammation. However, the content of IL-6 and TNF-α in the PC+TBI and TBI+PC were significantly lower than that in the radiation group, which to some extent proved that phycocyanin could reduce pneumonia caused by radiation.

|
| Fig.5 PC inhibits IL-6 release IL-6 levels in the lung measured by ELISA. Data were expressed as mean±SD, (n=6). * represents a significant difference compared with the TBI (P < 0.05); ** represents an extremely significant difference compared with the TBI (P < 0.01); # represents a significant difference compared with the Control (P < 0.05); ## represents an extremely significant difference compared with the control (P < 0.01). |
|
| Fig.6 PC inhibits cytokines release TNF-α levels in the lung were measured by ELISA. Data were expressed as mean±SD, (n=6). * represents a significant difference compared with the TBI (P < 0.05); ** represents an extremely significant difference compared with the TBI (P < 0.01); # represents a significant difference compared with the Control (P < 0.05); ## represents an extremely significant difference compared with the control (P < 0.01). |
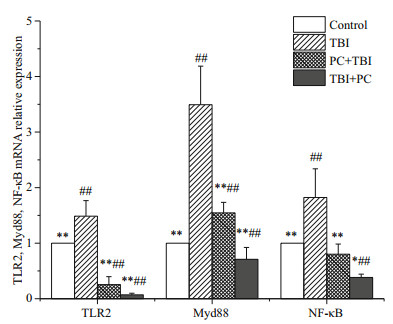
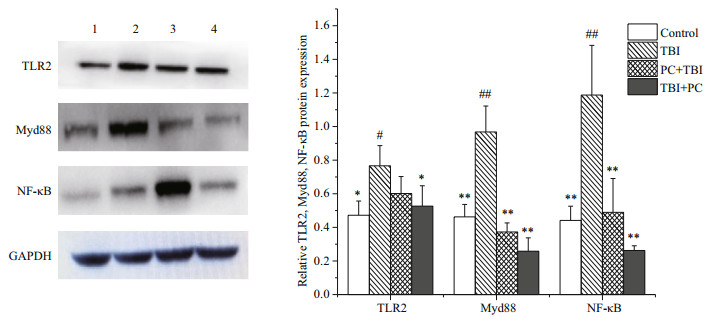
Animal studies have shown that oral administration of PC can reduce the expression of NF-κB in injured lungs. LI Chengcheng and other reports suggest that phycocyanin can reduce lung oxidative stress, reduce the release of inflammatory factors and weaken pulmonary fibrosis through the TLR2-MyD88-NF- κB signaling pathway. While it is not clear whether PC can play a role in preventing radiation pneumonitis through the TLR dependent mechanism. First of all, we studied the effects of PC on gene transcription level of TLR2, Myd88, and NF-κB. As shown in Fig. 7, RT-PCR results confirmed that PC reduced the expression levels of TLR2, MyD88, and NF-κB in X-ray exposed mice on day 7. In addition, the relative expression of mRNA in TBI+PC are lower than it in the PC+TBI. Subsequently, we further verify TLR2 at the level of translation. The relative expression of three proteins was measured by Western blot. Figure 8 shows the level of protein expression. The relative expression of the protein is similar to that of mRNA. In summary, PC can attenuate radiation-induced lung inflammation by blocking the TLR2-MyD88-NF-κB signaling pathway.
|
| Fig.7 Effects of phycocyanin on the transcriptional levels of TLR2, Myd88, and NF-κB We determined TLR2, Myd88 and NF-κB mRNA by RT-PCR. Data were expressed as mean±SD, (n=6). * represents a significant difference compared with the TBI (P < 0.05); ** represents an extremely significant difference compared with the TBI (P < 0.01); # represents a significant difference compared with the control (P < 0.05); ## represents an extremely significant difference compared with the control (P < 0.01). |
|
| Fig.8 Effects of PC on TLR2, Myd88 and NF-κB proteins expression in lung These proteins expression on Day 7 after radiation were analyzed by Western blot, and band (1) represents control group, and band (2) represents TBI group; band (3) represents PC+TBI group; band (4) represents TBI+PC group. Images are representative of the three independent studies. Data were expressed as mean±SD. * represents a significant difference compared with the TBI (P < 0.05); ** represents an extremely significant difference compared with the TBI (P < 0.01); # represents a significant difference compared with the Control (P < 0.05); ## represents an extremely significant difference compared with the Control (P < 0.01). |
Radiation-induced lung injury has been described as a continuous multicellular process involving the immediate release of cytokines from specific cells, rather than their sequential release during the incubation period, pneumonia, and later stages of injury (Verma et al., 2017). The oxidative stress induced by radiation is closely associated with inflammation, causing cell damage and inducing changes in the expression of cytokines and growth factors in epithelial and endothelial cells, fibroblasts, and alveolar macrophages. Some proinflammatory cytokines such as interleukin IL-1, IL-6, and tumor necrosis factor TNF-α are released by the alveoli and interstitium, resulting in lung injury (Ding et al., 2013).
The outbreak of inflammation is always accompanied by oxidative stress caused by the accumulation of ROS (Rong et al., 2012). ROS accumulation in vivo often plays a role in different pathological processes, including inflammatory responses, by driving multiple molecular pathways. Previous studies have shown that PC effectively scavenges free radicals, so we explored whether PC can alleviate pneumonia caused by radiation in the present study. We detected ROS in the mouse lung after exposure to X rays and found that this dramatically reduced following PC gavage.
Pneumonia and fibrosis are the main consequences of lung radiation (Verma et al., 2017), and inflammation is known to play a crucial role in the development of fibrosis. Moreover, reducing the extent of early stage radiation pneumonia effectively prevents the occurrence and development of pulmonary fibrosis. Therefore, we investigated the protective effect of PC on early stage radiationinduced pneumonia using IL-6 and TNF-α as indicators of radiation-induced inflammation (Williams et al., 1998). We detected inflammatory markers in the plasma, lung, and intestine, and detected a significant increase in their levels in mice treated with radiation. However, the concentration of IL-6 in the PC preventive group was significantly lower than in the radiation group, suggesting that PC reduced radiation-induced pneumonia.
To further study the role of PC in reducing lung injury caused by radiation, we explored its effect on signaling pathways. The transmembrane receptor TLR functions in immune and inflammatory processes by interacting with conserved adaptor proteins such as MyD88 and TRIF (Medzhitov, 2001; Zhang et al., 2005; Ogawa et al., 2007). The TLR signaling pathway also plays an important role in acute lung injury and radiation damage (Roses et al., 2008). Under normal conditions, TLR signal activation activated the anti-microbial defense system, stimulates neutrophils and phagocytes, and causes the production of inflammatory mediators such as IL-1, IL-6, IL-8, and TNF-α to trigger inflammation. This enables the removal of invasive pathogenic microorganisms and promotes the repair of damaged tissues, but can also cause damage to the body's own tissues and organs. Damaged cells or necrotic cells release large amounts of danger-associated molecular patterns, which act as endogenous ligands to further activate the TLR/NF- κB signaling pathway, release inflammatory mediators, and recruit inflammatory cells. This positive feedback cycle eventually leads to cascade amplification and uncontrollable persistence of the inflammatory response, resulting in pathological changes in lung function (Doz et al., 2008). Therefore, we assumed that PC can block TLR/NF-κB to alleviate pneumonia induced by radiation. We previously showed that PC treated pulmonary fibrosis by attenuating the expression of molecules in the TLR2-MyD88-NF-κB pathway (Li et al., 2017), so herein we examined whether PC alleviated radiationinduced lung injury in the same way. The expression of TLR, Myd88, and NF-κB in groups PC+TBI and group TBI+PC was significantly lower than in group TBI, suggesting that blockage of the TLR2-MyD88- NF-κB pathway was at least partially responsible for alleviating radiation-induced pneumonia.
5 CONCLUSIONPC reduced the ROS produced in the lung and reduced the release of inflammatory factors. PC has a certain relieving effect on pneumonia caused by radiation. A part of the reason may be blocking TLR2- MyD88-NF-κB pathway.
6 DATA AVAILABILITY STATEMENTThe datasets generated during the current study are available in NCBI.
Arrieta O, Gallardo-Rincón D, Villarreal-Garza C, Michel R M, Astorga-Ramos A M, Martínez-Barrera L, De La Garza J. 2009. High frequency of radiation pneumonitis in patients with locally advanced non-small cell lung cancer treated with concurrent radiotherapy and gemcitabine after induction with gemcitabine and carboplatin. J.Thorac. Oncol., 4(7): 845-852.
DOI:10.1097/JTO.0b013e3181a97e17 |
Bhat V B, Madyastha K M. 2000. C-phycocyanin:a potent peroxyl radical scavenger in vivo and in vitro. Biochem.Biophys. Res. Commun., 275(1): 20-25.
DOI:10.1006/bbrc.2000.3270 |
Carmody R J, Chen Y H. 2007. Nuclear factor-κB:activation and regulation during toll-like receptor signaling. Cell.Mol. Immunol., 4(1): 31-41.
|
Chang C J, Yang Y H, Liang Y C, Chiu C J, Chu K H, Chou H N, Chiang B L. 2011. A novel phycobiliprotein alleviates allergic airway inflammation by modulating immune responses. Am. J. Respir. Crit. Care. Med., 183(1): 15-25.
DOI:10.1164/rccm.201001-0009OC |
Demirel C, Kilciksiz S C, Gurgul S, Erdal N, Yigit S, Tamer L, Ayaz L. 2016. Inhibition of radiation-induced oxidative damage in the lung tissue:may acetylsalicylic acid have a positive role?. Inflammation, 39(1): 158-165.
DOI:10.1007/s10753-015-0234-x |
Ding N H, Li J J, Sun L Q. 2013. Molecular mechanisms and treatment of radiation-induced lung fibrosis. Curr. Drug Targets, 14(11): 1347-1356.
DOI:10.2174/13894501113149990198 |
Doz E, Noulin N, Boichot E, Guenon I, Fick L, Le Bert M, Lagente V, Ryffel B, Schnyder B, Quesniaux V F J, Couillin I. 2008. Cigarette smoke-induced pulmonary inflammation is TLR4/MyD88 and IL-1R1/MyD88 signaling dependent. J. Immunol., 180(2): 1169-1178.
DOI:10.4049/jimmunol.180.2.1169 |
Hosseinimehr S J. 2007. Trends in the development of radioprotective agents. Drug Discov. Today, 12(19-20): 794-805.
DOI:10.1016/j.drudis.2007.07.017 |
Kim H S, Go H, Akira S, Chung D H. 2011. TLR2-mediated production of IL-27 and chemokines by respiratory epithelial cells promotes bleomycin-induced pulmonary fibrosis in mice. J. Immunol., 187(8): 4007-4017.
DOI:10.4049/jimmunol.1101654 |
Li C C, Yu Y, Li W J, Liu B, Jiao X D, Song X Y, Lv C J, Qin S. 2017. Phycocyanin attenuates pulmonary fibrosis via the TLR2-MyD88-NF-κB signaling pathway. Sci. Rep., 7(1): 5843.
DOI:10.1038/s41598-017-06021-5 |
Li J, Mu S, Mu L, Zhang X, Pang R, Gao S. 2015. Transforming growth factor-beta-1 is a serum biomarker of radiationinduced pneumonitis in esophageal cancer patients treated with thoracic radiotherapy:preliminary results of a prospective study. Onco. Targets Ther., 8: 1129-1136.
|
Li Y J, Han Z, Ge L, Zhou C J, Zhao Y F, Wang D H, Ren J, Niu X X, Liang C G. 2016. C-phycocyanin protects against low fertility by inhibiting reactive oxygen species in aging mice. Oncotarget, 7(14): 17393-17409.
DOI:10.18632/oncotarget.8165 |
Liu H Z, Yang H Z, Mi S, Cui B, Hua F, Hu Z W. 2010. Toll like receptor 2 mediates bleomycin-induced acute lung injury, inflammation and fibrosis in mice. Acta Pharm.Sinica, 45(8): 976-986.
|
Medzhitov R. 2001. Toll-like receptors and innate immunity. Nature Reviews Immunology, 1: 135-145.
DOI:10.1038/35100529 |
Mehta V. 2005. Radiation pneumonitis and pulmonary fibrosis in non-small-cell lung cancer:pulmonary function, prediction, and prevention. Int. J. Radiat. Oncol. Biol. Phys., 63(1): 5-24.
DOI:10.1016/j.ijrobp.2005.03.047 |
Ogawa Y S, Tasaka W, Yamada F, Saito N, Hasegawa T, Miyasho, Ishizaka A. 2007. Role of Toll-like receptor 4 in hyperoxia-induced lung inflammation in mice. Inflammation Research, 56: 334-338.
DOI:10.1007/s00011-007-7052-z |
Rong S, Zhao Y T, Bao W, Xiao X, Wang D, Nussler A K, Yan H, Yao P, Liu L G. 2012. Curcumin prevents chronic alcohol-induced liver disease involving decreasing ROS generation and enhancing antioxidative capacity. Phytomedicine, 19(6): 545-550.
DOI:10.1016/j.phymed.2011.12.006 |
Roses R E, Xu M, Koski G K, Czerniecki B J. 2008. Radiation therapy and Toll-like receptor signaling:implications for the treatment of cancer. Oncogene, 27: 200-207.
DOI:10.1038/sj.onc.1210909 |
Shen W B, Zhu S C, Gao H M, Li Y M, Liu Z K, Li J, Su J W, Wan J. 2013. Low dose volume histogram analysis of the lungs in prediction of acute radiation pneumonitis in patients with esophageal cancer treated with threedimensional conformal radiotherapy. Chin. J. Oncol., 35(1): 45-49.
|
Verma S, Kalita B, Bajaj S, Prakash H, Singh A K, Gupta M L. 2017. A combination of podophyllotoxin and rutin alleviates radiation-induced pneumonitis and fibrosis through modulation of lung inflammation in mice. Front.Immunol., 8: 658.
DOI:10.3389/fimmu.2017.00658 |
Williams M A, Withington S, Newland A C, Kelsey S M. 1998. Monocyte anergy in septic shock is associated with a predilection to apoptosis and is reversed by granulocytemacrophage colony-stimulating factor ex vivo. J. Infect.Dis., 178(5): 1421-1433.
DOI:10.1086/314447 |
Wu Q H, Liu L, Miron A, Klímová B, Wan D, Kuča K. 2016. The antioxidant, immunomodulatory, and antiinflammatory activities of Spirulina:an overview. Arch.Toxicol., 90(8): 1817-1840.
DOI:10.1007/s00204-016-1744-5 |
Zhang X C, Shan P Y, Qureshi S, Homer R, Medzhitov R, Noble P W, Lee P J. 2005. Cutting edge:TLR4 deficiency confers susceptibility to lethal oxidant lung injury. J.Immunol., 175(8): 4834-4838.
DOI:10.4049/jimmunol.175.8.4834 |
 2019, Vol. 37
2019, Vol. 37