Institute of Oceanology, Chinese Academy of Sciences
Article Information
- LIU Yuhao, ZHANG Guoliang, ZHANG Ji, WANG Shuai
- Geochemical constraints on CO2-rich mantle source for the Kocebu Seamount, Magellan Seamount chain in the western Pacific
- Journal of Oceanology and Limnology, 38(4): 1201-1214
- http://dx.doi.org/10.1007/s00343-020-0013-x
Article History
- Received Jan. 15, 2020
- accepted in principle Feb. 24, 2020
- accepted for publication Apr. 10, 2020
2 Laboratory for Marine Geology, Qingdao National Laboratory for Marine Science and Technology, Qingdao 266000, China;
3 University of Chinese Academy of Sciences, Beijing 100049, China;
4 Center for Ocean Mega-Science, Chinese Academy of Sciences, Qingdao 266071, China
Ocean island basalts (OIBs) are usually geochemically enriched with relatively higher concentration of incompatible trace elements than mid-ocean ridge basalts (MORBs). The OIBs are generally considered to have resulted from partial melting of a mantle source that has been affected by recycled materials, i.e., recycled oceanic crust through ancient plate subduction (Hofmann et al., 1986; Weaver, 1991). The partial melting of recycled oceanic crust usually produces melts with saturated silica (Hirose and Kushiro, 1993; Kawamoto and Holloway, 1997). However, the alkaline OIBs usually have under-saturated SiO2 and enriched alkaline content, which cannot be produced by partial melting of dry mantle peridotites (Hirose and Kushiro, 1993), but could be produced by partial melting of carbonated peridotites (Hirose, 1997; Dasgupta et al., 2007a) or carbonated eclogites (Dasgupta et al., 2006; Hammouda et al., 2009; Kiseeva et al., 2012). It has been shown by previous studies that the mantle melting with CO2 can reduce the content of SiO2 in the melt (Dasgupta et al., 2007b), which suggest that CO2 probably acts as a controlling factor for the generation of alkali basalt. Moreover, mantle xenoliths collected from the alkali basalts usually show obvious "carbonatitic fingerprints" (Coltorti et al., 1999; Kogarko et al., 2001; Neumann et al., 2002). Hence, study of the geochemistry of alkali basalts has important implications on both the genesis of OIBs and the deep carbon cycle.
The West Pacific Seamount Province is underlain by the oldest oceanic crust of the Pacific (Müller et al., 2008), and has the densest seamount distribution in the Pacific (Li, 2010). The alkali basalts are widely distributed in the West Pacific Seamount Province (Zhu, 2002; Wei et al., 2017a). However, the genesis of these alkali basalts and their mantle source remain debated. Therefore, study on the geochemistry of the alkali OIBs would help understand how the West Pacific Seamount Province was formed. The Magellan Seamounts Trail, as a part of the West Pacific Seamount Province, was widely known as an area of oceanic mineral resources (e.g., Co-crusts and rare earth elements-rich phosphorites) (Pan et al., 2002; Wei et al., 2017b). The previous studies in this area generally focused on the Co-crusts (Pan and Liu, 1999; Wang et al., 1999; Zhu et al., 2001; Sun et al., 2006; Bu et al., 2007; Ye et al., 2008; Song et al., 2009; Ren et al., 2011; Wang et al., 2016) or phosphorites (Pan et al., 2002; Wei et al., 2017b), thus, the distribution of mineral resources and their genesis have been extensively studied. The previous studies have investigated the seamount topography based on the fractal methods (Zhang et al., 2009) and traced the origin of the seamounts trail based on geophysics methods (Zhao et al., 2010).
To understand petrology and petrogeochemistry, Smith et al. (1989) and Koppers et al.(1998, 2003) studied six seamounts from the Magellan Seamounts Trail based on mineral 40Ar/39Ar ages and Sr and Pb isotopes, and ascribed their genesis as the result of activities of deep mantle-rooted South Pacific Isotopic and Thermal Anomaly (SOPITA). However, Tang et al. (2019) found that, at the western part of Magellan Seamounts, some alkali basalts have magma sources ranging from EMI to EMII, probably suggesting that the Magellan Seamounts have more than one mantle source. Moreover, Zhu (2002), and Wei (2017a) collected bedrock samples from Ita Mai Tai, Gelenddzhink, Govorov, Il'ichev and Pallada seamounts, and the relevant geochemistry data were reported. However, previous studies mostly focused on regional tectonic genesis and evolution of the seamounts (Koppers et al., 1998, 2003; Tang et al., 2019), rock composition and mineral composition (Zhu, 2002; Wei et al., 2017a), hence the genesis of alkali basalts still remains unclear. The unique characteristics of the alkali basalts (for example, high P2O5 content) of the Magellan Seamounts Trail have been interpreted by most previous studies as result of secondary minerals by alteration (e.g., Zhu, 2002; Wei et al., 2017a) without a detailed study of the chemical composition and genesis of the primary rock-forming minerals. Thus, it is necessary to constrain the genesis of alkali basalts of Magellan Seamounts based on detailed study of whole-rock geochemistry and mineral chemistry. In this study, we provide new whole-rock geochemistry data and the electronic microprobe analysis (EPMA) data of mineral chemical composition from the Kocebu Seamount of this seamount chain, which has not been previously sampled. The objectives are to examine the genesis of the alkali basalts of the Magellan Seamounts Trail and their possible relationship with CO2 in the deep mantle source.
2 GEOLOGICAL SETTING AND SAMPLEThe Magellan Seamounts Trail is located to the east of the Mariana Trench and the west of Pigafetta Basin. It has a number of seamounts distributed along the trend from northwest to southeast with a length of about 1 200 km. The basement rocks of this area include pillow basalts, Jurassic tholeiites and Cretaceous alkali basalts (Liang and Zhu, 2002). The ages of the seamounts range mostly 120–80 Ma and generally decrease from northwest to southeast (Koppers et al., 2003). Twenty samples were obtained from the Kocebu Seamount, where igneous rock samples have never been collected previously. Some of the samples in this study are covered with Fe-Mn crusts. Most samples are moderately altered, while porphyritic structure can be observed in the slightly altered samples.
The phenocrysts are dominated by clinopyroxene and plagioclase in this study, while fresh olivine phenocryst has not been observed. Clinopyroxenes and plagioclases are mostly euhedral crystals. Apatites and ilmenites can be observed as auxiliary minerals in most samples. Apatites have two types of crystal, anhedral apatites mostly spread at the edge of the plagioclases or filling the cells among whole rocks, euhedral apatites mostly are granular or needle crystals. Ilmenites mostly are granular crystals.
3 METHODMajor elements and some trace elements (Cr, V, etc.) were analyzed on fused glass discs with PANalytical AXIOS Minerals instrument at the Rock-Mineral Preparation and Analysis Lab, the Institute of Geology and Geophysics (IGG; Beijing), Chinese Academy of Sciences (CAS). Samples were analyzed on fused glass discs with an Axios sequential X-ray Fluorescence Spectrometer, and glass discs were produced at 1 000℃ using a lithium tetraborate flux (Li2B4O7) in a mixture consisting of 0.5 g of sample and 5 g of lithium tetraborate. The loss on ignition (LOI) of sample powders was determined at a temperature of 1 000℃. The measurements were monitored using standards BCR-2 and BHVO-2. The accuracy of the analytical results was controlled by measuring standard reference material BCR-2 and BHVO-2 (Supplementary Table S1).
Trace elements analyses were determined using an inductively coupled plasma mass spectrometry (ICP-MS) at the Second Institute of Oceanography, Ministry of Nature Resources (MNR). For trace element analyses, 50 mg of sample powder was precisely weighed and transferred into Teflon capsule; sample powder was moisturized by 0.1-mL Mili-Q water; 1.5-mL HF, 1.5-mL HNO3 and 0.1-mL HClO4 were added in turn; the Teflon capsule was sealed in stainless steel bomb jacket; and lastly the unit was heated in oven at 190℃ for 48 h. When the units cooled down, the Teflon capsules were taken out; opened and heated on a hotplate at 120℃ to evaporate the dissolutions to dryness; 1.5-mL HClO4 was added, and heated on a hotplate at 120℃ until the white smoke was totally dispersed; 3-mL HNO3 was added, and heated on a hotplate at 120℃ to evaporate the dissolutions to dryness. After that, 3-mL 1:1 HNO3 was added, sealed in stainless steel bomb jacket, and heated in oven at 190℃ for 24 h to re-dissolve the residues. Finally, the solutions were diluted by 2 000 times using Mili-Q water and analyzed by ICP-MS using Rh and Re as internal standards. The standard solutions were diluted to 1, 5, 10, 20, 50, and 80 μg/L to produce the calibration curve with linear regression coefficients all above 0.999. To evaluate the accuracy, BHVO-2 and BCR-2 were run as external standards, and the results are shown in Supplementary Table S2.
The in-situ microanalysis was conducted at the Institute of Oceanology, Chinese Academy of Sciences (CAS) on a JXA-8230 EPMA. The analyses were performed under conditions of accelerating voltage of 20 kV and sample current of 10 nA for clinopyroxene and feldspar, accelerating voltage of 15 kV and sample current of 10 nA for apatite. The data were calibrated by using an oxide-ZAF correction program.
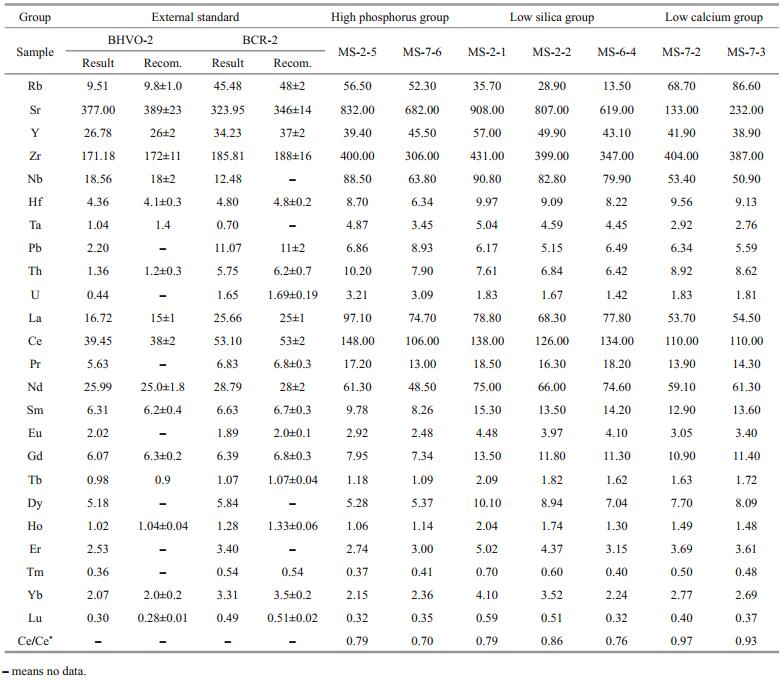
4 RESULT 4.1 Major elementsThe results of major elements are shown in Table 1. According to the nomenclature of Le Maitre (1984), the igneous rocks of Magellan Seamounts are trachy basalt, basanite tephrite, tephriphonolite and foidite, and all igneous rocks are alkaline rock (Fig. 1). Compared with typical OIBs, e.g., Hawaii OIBs, the igneous rocks of the Magellan Seamounts have obviously lower SiO2 contents, lower MgO and higher CaO, TiO2, and P2O5 contents. These results are similar to those of the experimentally determined carbonated eclogite-derived melt (except for P2O5) and the nephelinites of Canary Islands (Fig. 2), where carbonatites extruded (Hoernle et al., 2002) and the mantle xenoliths show metasomatism by carbonated melts (Neumann et al., 2002). These igneous rock samples can be divided into three groups, high phosphorus group, low calcium group, and low silica group, based on their SiO2, CaO, and P2O5 contents (Fig. 2). Samples of high phosphorus group have obviously higher P2O5 contents (4.2–4.7 wt%) and relatively higher CaO contents (9.8–10 wt%) than those of the other groups, and have SiO2 contents of between 44.5–46.9 wt% (Fig. 2). The samples of low calcium group have lower CaO content (1.6%–2.2%), higher SiO2 (47.2–54.6 wt%) and lower P2O5 (0.89– 0.92 wt%) contents than other groups. Samples of low silica group have lower SiO2 content (36.9%–46.9%), higher CaO (8.9–11.8 wt%) and P2O5 (1.4–2.0 wt%) contents than the other groups (Fig. 2).

|
| Fig.1 Total Alkali Silica (TAS) diagram (after Le Maitre, 1984) Previous data are from http://georoc.mpch-mainz.gwdg.de/georoc/Entry.html, Zhu (2002), and Wei et al. (2017a). |
|
| Fig.2 Major elements-SiO2 diagram of Magellan Seamounts Trail Previous data are from http://georoc.mpch-mainz.gwdg.de/georoc/Entry.html, Zhu (2002), and Wei et al. (2017a), carbonated peridotite melt data are from Hirose (1997) and Dasgupta et al. (2007a), carbonated eclogite melt data are from Hammouda et al. (2009), Gerbode and Dasgupta (2010), Kiseeva et al. (2012), and data of Hawaii and Canary Island nephelinite are from http://georoc.mpch-mainz.gwdg.de/georoc/Entry.html. |
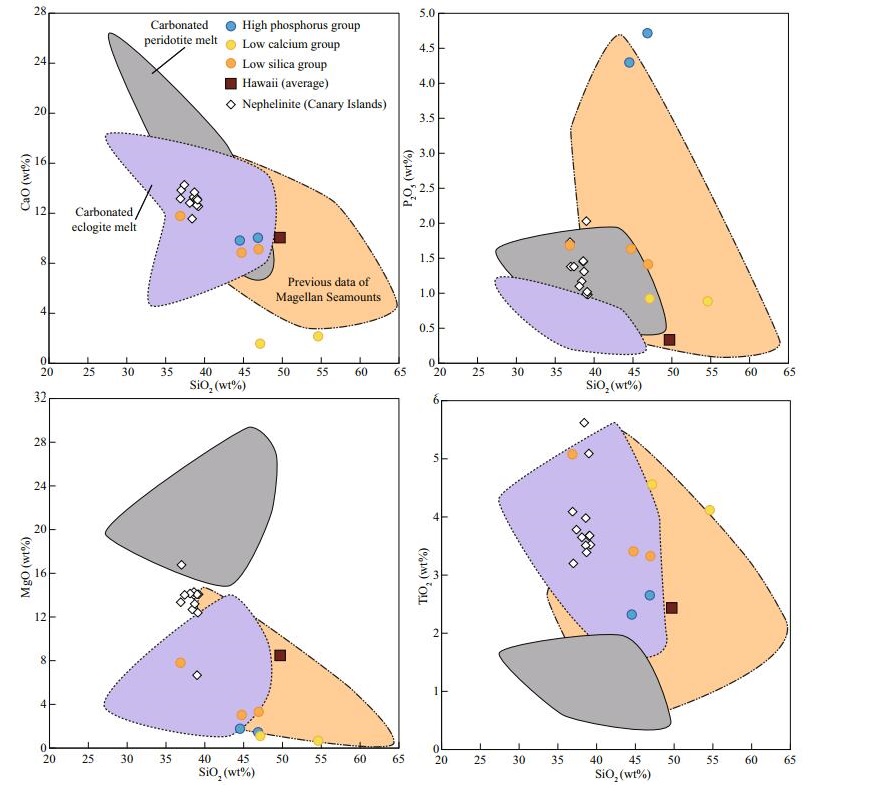
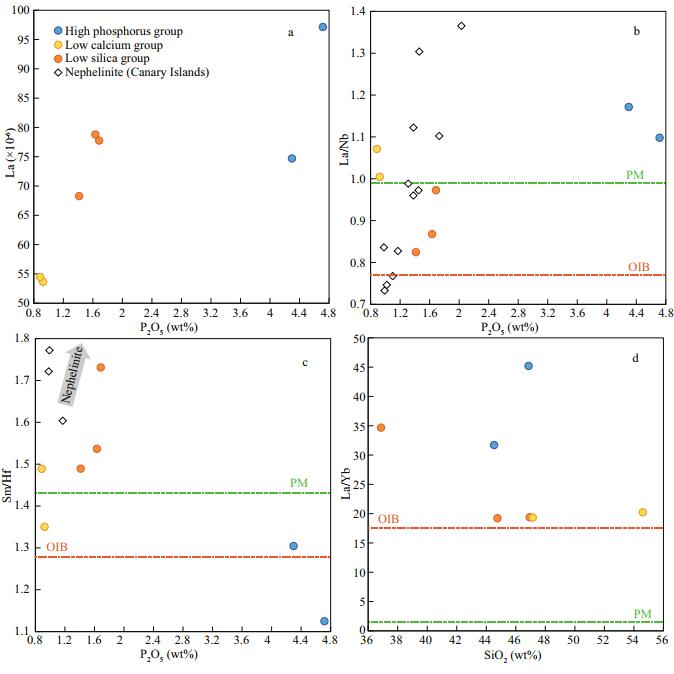
The results of trace elements are shown in Table 2. To avoid the effect of seafloor alteration, we eliminated samples that have obvious Ce anomalies. All samples of the Magellan Seamounts have OIB-type rare earth elements (REEs) pattern, but the different groups have variable trace elements patterns (Fig. 3). Compared with typical OIBs, the high phosphorus group has slight Nb and Ta negative anomalies and P obvious positive anomaly (Fig. 3). High phosphorus group has higher La/Nb and La/Yb ratio than OIBs, even higher than the primitive mantle (PM) (Fig. 4b & c). The low calcium group has an obvious K positive anomaly (Fig. 3). The low calcium group has higher La/Nb ratio, similar to the PM, but La/Yb ratio higher than PM, similar to the OIBs (Fig. 4b & c). Compared with typical OIBs, the low silica group samples have slight Zr and Hf negative anomalies, and K has negative anomaly (Fig. 3). For the relations between major and trace elements, there are obvious correlations between not only P2O5 and La, but also P2O5 and La/Nb ratio (Fig. 4a & b). The low silica group has similar La/Nb ratio with nephelinites of Canary Islands, and the highest P2O5 sample of low silica group has higher La/Nb ratio than OIBs, even higher than PM (Fig. 4b). Samples of the low silica group have higher La/Yb ratio than typical OIBs, especially for the samples with low SiO2 or high P2O5 contents of this group (Fig. 4c).
|
| Fig.3 REE and trace elements patterns of three groups Data of Cl chondrite and primitive mantle for normalization are from McDonough and Sun (1995). Data of OIB are from Sun and McDonough (1989). Data of oceanic carbonatite are from Hoernle et al. (2002). |
|
| Fig.4 La-P2O5, La/Nb-P2O5, Sm/Hf-P2O5, and La/Yb-SiO2 diagram OIB and PM data are from Sun and McDonough (1989), and Canary Island nephelinite data are from http://georoc.mpch-mainz.gwdg.de/georoc/entry.html. |
The results of in-situ microanalysis on apatites, clinopyroxenes and feldspars are shown in Supplementary Tables S2–S4, respectively, and Supplementary Table S1 shows the range, average and number of analyzed. High phosphorus group has two samples: MS-2-5 and MS-7-6 (Fig. 5a: MS-2-5, b: MS-7-6). The main minerals are potassium feldspars, plagioclases, apatites and ilmenite. Apatites mostly are anhedral crystal. Plagioclases enclosed within by potassium feldspars, and the An# plagioclases are mostly between 50 to 70, the highest value is 75, and the average is 64. Low silica group has five samples: MS-2-1, MS-2-2 and MS-6-4 (Fig. 5c: MS-6-4, d: MS-2-1). The main minerals are clinopyroxenes, plagioclases, apatites and ilmenite, wherein MS-6-4 does not contain plagioclases. Mineral compositions between samples in this group are different, with the decrease of SiO2 and CaO content (from Fig. 5c to 5d), clinopyroxenes are decreasing, plagioclases was increasing, and crystal forms of apatites vary from euhedral crystal to anhedral crystal. The An# plagioclases are mostly between 60 to 70, with the highest value of 76 and the average value of 68. Low calcium group has two samples: MS-7-2 and MS-7-3 (Fig. 5e: MS-7-2, b: MS-7-3). The main minerals are potassium feldspars, plagioclases, apatites, and ilmenite. Apatites mostly are euhedral crystal. Plagioclases were wrapped by potassium feldspars, and the An# of plagioclases are mostly between 10 to 50, with the highest value of 57, and the average value of 29.

|
| Fig.5 Back Scattered Electron (BSE) photographs of typical samples from three groups a. MS-2-5; b. MS-7-6, both are from high phosphorus group; c. MS-6-4; d. MS-2-1, both are from low silica group; e. MS-7-2; f. MS-7-3, both are from low calcium group. Kfs: potassium feldspar; Pl: plagioclase; Ap: apatite; Ilm: ilmenite; Cpx: clinopyroxene. |
Previous studies suggest that phosphorites are widely distributed along the Magellan Seamounts Trail (Pan et al., 2002; Zhu, 2002; Wei et al., 2017a). The enrichment phosphorus in igneous rocks has usually been described as the effects of secondary phosphate as indicated by the obvious Ce anomaly (Zhu et al., 2001; Zhu, 2002; Wei et al., 2017a). However, as shown in Fig. 3, the samples without Ce anomalies from the Magellan Seamounts Trail in this study have higher P2O5 contents than typical OIBs. Here we analyzed the chemical compositions of apatites by EPMA to decipher the origin of phosphorus enrichment. We found two types of apatites in these samples: allotriomorphic apatites (Fig. 5a & b) that mostly exist in the samples of high phosphorus group, and idiomorphic apatites (Fig. 5c, e, & f) that mostly exist in low silica group and low calcium group. It is shown in Fig. 6 that the P2O5 contents and CaO/P2O5 ratio are different among different types of apatites. Idiomorphic apatites show relatively high P2O5 content (30–42 wt%) and low CaO/P2O5 ratio (ca. 1.3). Allotriomorphic apatites show relatively low P2O5 content (24–32 wt%) and CaO/P2O5 ratios higher than 1.6.

|
| Fig.6 Apatites CaO/P2O5-P2O5 diagram Data of apatites from igneous rocks are from http://georoc.mpch-mainz.gwdg.de/georoc/Entry.html, and data of phosphorites are from Pan et al. (2002). |
In marine environment, natural crystallized or metasomatic apatites are all francolites (Ye et al., 1989). Due to the replacement between CO32- and PO42-, the CaO/P2O5 ratio of francolites could be higher than 1.32 (calculated as fluorapatite: Ca5(PO4)3F). Previous experimental study suggested that, in marine environment, secondary phosphates probably mixed with calcite (Ye et al., 1989), which would enhance the CaO/P2O5 ratio. Hence, the allotriomorphic apatites from the high phosphorus group probably are secondary phosphate, the higher P2O5 content than other groups probably are the effect of secondary phosphate. The idiomorphic apatites with CaO/P2O5 ratios similar to fluorapatite suggest that these idiomorphic apatites are original magmatic minerals. Even in the samples with the lowest P2O5 content, the content of phosphorus is obviously higher than typical OIBs (Fig. 2). Therefore, the obviously higher phosphorus of these samples could be features of magmas that formed alkaline rocks in this study area.
5.2 Effect of seafloor alterationThe composition of rocks collected from the ocean would be more or less affected by seawater alteration. It is necessary to discuss the changes of whole rock chemistry caused by alteration. For the seawater alteration, previous studies have been carried out in detail. Hart (1973) believes that for major elements, the content of K2O will increase significantly, the content of MgO, SiO2, CaO, and MnO would decrease slightly, while the content of Na2O, Al2O3, and TiO2 would remain unchanged. For trace elements, Ba, Rb, Sr, V, Zr, Cs, and U would increase, Co and Cr would decrease, while Cu, Ga, Sc, and Zn would remain basically unchanged. Staudigel et al. (1981) reported that seawater alteration will not affect Fe2O3(T) and TiO2, but the content of K2O, Rb, and Cs will increase; the content of REEs will decrease, but their patterns will not generally change. Zhang and Smith-Duque (2014) showed that seawater alteration would not affect the TiO2 content, but it may cause a small decrease in CaO, MgO, and Na2O content and a significant increase in K2O content. In terms of trace elements, seawater alteration will cause the increase of Rb, Cs, Ba, and U content, and decrease of Th content. It has little influence on REEs and high field strength elements (HFSEs) (e.g., Nb, Hf). Summarizing the previous research results, the main result of seawater alteration is increase of K2O content, increase or decrease of large ion lithophile elements (LILEs) (for example, Rb, Ba), without significant influences on HSFEs and REEs.
For rocks in Magellan Seamounts, phosphatization also widely exists. The effects of phosphatization on the composition of whole rocks have also been studied in detail. Pan et al. (2007), collected a large number of seamount phosphates in the Western Pacific, and found that most of these phosphates are accessory minerals that appear in needle, interlayers, or veins. The average of CaO/P2O5 ratio was 1.60. Ren and Wei (2007) studied REEs in phosphates, and found that oceanic phosphates will inherit the characteristics of REEs in seawater, which are mainly manifested by obvious negative Ce anomalies and higher Y/Ho ratios. The distribution characteristics are common in Pacific seamount phosphates (Pan et al., 2002). Summarizing previous research results, secondary phosphates in the Pacific Ocean are mainly characterized by high CaO/P2O5, Y/Ho ratio and obvious negative Ce anomalies.
For this study, results of the EPMA analysis suggest that the secondary phosphate exists in some samples, indicating that some of the samples in this study were affected by phosphate. The high Y/Ho ratio may have multiple origins (e.g., magma generated by partial melting of the CO2 and halogen rich source may also have a high Y/Ho ratio (Zhang et al., 2017)), in order to exclude the effect of secondary phosphate, we chose Ce/Ce* (CeN2/ (LaN×PrN)) as an indicator to measure the effect of the sample after the phosphate, and exclude samples with Ce/Ce* < 0.70 to eliminate the effect of phosphatization (Supplementary Table S2). For seawater alteration, we select elements that are insensitive to seawater alteration (such as HFSEs and REEs) as the indicators of discussion in order to exclude the effect of seawater alteration.
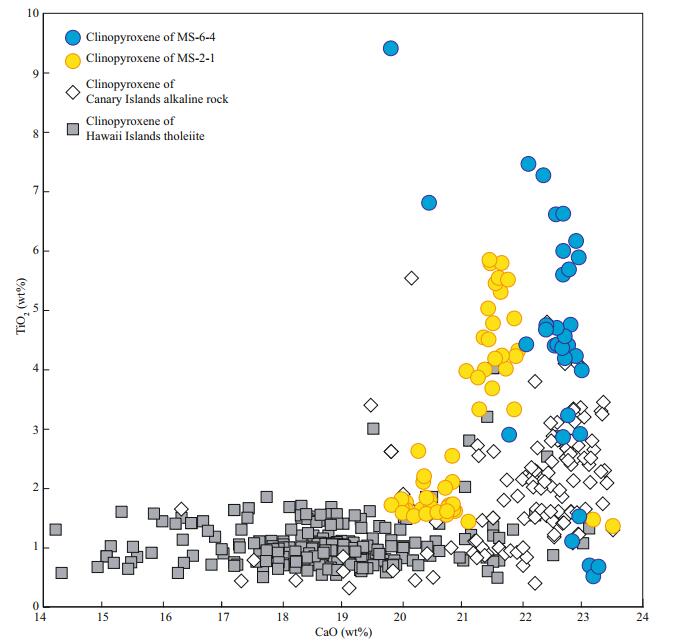
5.3 Effect of magma fractional crystallizationBased on major element compositions, the low silica group samples have highly variable bulk rock SiO2 contents, and their SiO2 contents show correlations with other elements (e.g., Ca, P). Their clinopyroxene CaO contents also decrease with increasing bulk rock SiO2 (Fig. 7, bulk rock SiO2 increases from 36.87 wt% for MS-6-4 to 44.76 wt% for MS-2-1), similar to the bulk rock data, suggesting that the bulk rock CaO decreased as clinopyroxene crystallization proceeded. Thus, the samples of the low silica group have undergone fractional crystallization of clinopyroxenes and apatites.
|
| Fig.7 Clinopyroxenes CaO-TiO2 diagram Data of clinopyroxenes from Hawaii tholeiites and Canary Island alkaline rocks are from http://georoc.mpch-mainz.gwdg.de/georoc/entry.html. |
Some elements, especially light rare earth elements (LREEs) and middle rare earth elements (MREEs), are strongly enriched in apatite (Chazot et al., 1996), hence crystallization of apatite probably causes the positive anomalies of Nb, Ta, Zr, and Hf (e.g., Zhang et al., 2017). In this study, LREEs (for example, La) and La/Nb, Sm/Hf ratios have negative correlations with P2O5, probably suggests the control of apatite on REEs (Fig. 4a, b, c). Moreover, the sample with the highest P2O5 content has higher La/Nb and Sm/Hf ratios than typical OIBs, similar to or even higher than PM (Fig. 4b, c). These features probably show that the samples of this study have obviously higher initial La/Nb and Sm/Hf ratios than typical OIBs, suggesting that the primary magmas probably have negative anomalies of Nb and Hf. The samples of low CaO and high SiO2 from the low silica group probably evolved from high CaO and low SiO2 magmas. Thus, the low silica group is the most suitable group for discussing the mantle source.
5.4 Mantle sourceCompared with typical OIBs, the samples from the low silica group have obviously higher CaO and lower SiO2 contents that are similar to the nephelinites of Canary Islands (Fig. 2). The low silica group samples have obviously higher P2O5 contents than typical OIBs, similar to the nephelinite that has the highest P2O5 content (Fig. 2). Previous studies suggested that the high P2O5 content and the appearance of apatite are usually the features of Si-under-saturated rocks that were formed from carbonated silicate melt (Green and Wallace, 1988; Hauri et al., 1993; Zhang et al., 2017). The negative anomalies of HFSEs (e.g., Nb, Ta, Zr, Hf) (Figs. 3, 4b & c) and K (Fig. 3) are obviously different from the lavas from typical mantle peridotites, but similar to those of the oceanic carbonatites (Fig. 3).
Clinopyroxene, as the early stage magmatic minerals of carbonated silicate melts, probably bears important information on mantle source, because it tends to crystallize from high Ca magmas, such as carbonated melts (Beccaluva et al., 1992; Ivanikov et al., 1998; Lee and Wyllie, 1998). The compositions of clinopyroxene crystalized from different magmatic systems are different. Figure 7 shows the compositional difference of clinopyroxene crystallized from different magmatic systems. Compared with typical OIBs (e.g., Hawaiian OIBs), the clinopyroxenes of this study have higher CaO and TiO2 contents, similar to the OIBs of the Canary Island, where carbonatites were found. The partition coefficients of Ti in clinopyroxene in equilibrium with carbonate melts is generally higher than those in equilibrium with silicate melts (Rudnick et al., 1993; Klemme et al., 1995), thus, the existence of high-Ti clinopyroxene in these samples suggest that they were derived from a carbonated primary magma.
Carbonated peridotites and carbonated eclogites are usually considered as the mantle sources of carbonated silicate melts (Hirose, 1997; Dasgupta et al., 2007a; Hammouda et al., 2009; Gerbode and Dasgupta, 2010; Kiseeva et al., 2012). For this study, the anomalously high TiO2 and the relatively low MgO contents are obviously different from the primary carbonated peridotite melts (Fig. 2), but similar to the carbonated eclogite melts. The very high La/Yb ratio (Fig. 4d) probably suggests the effects of a high proportion of residual garnet in the source mantle. Hence, we suggest that carbonated eclogites, probably derived from subduction-related oceanic crust, are the mantle source of carbonated silicate melts that contributed to the alkaline volcanic rocks of the Kocebu Seamount.
6 CONCLUSIONThe volcanic rocks of Magellan Seamounts are alkaline and have higher CaO, P2O5 and lower SiO2 contents than typical OIBs. Igneous rocks of Magellan Seamounts can be divided into three groups, and the samples that have low CaO and high SiO2 contents probably are evolved rocks. Primary minerals of samples are clinopyroxenes, feldspars, apatites, and ilmenites. The existence of primary magmatic apatites suggests the high phosphorus contents for alkaline primary magmas of the Magellan Seamounts. Clinopyroxenes in this study mostly are titanaugites, whose contents of CaO and TiO2 are higher than the clinopyroxenes from typical tholeiites, similar to clinopyroxenes from carbonatites. The obviously higher CaO and P2O5, lower SiO2 contents than typical OIBs, and HFSE negative anomalies and widespread titanaugites and apatites probably suggested a carbonated mantle source. The relatively higher TiO2, lower MgO contents and relatively higher La/Yb ratio suggest that the volcanic rocks of Magellan Seamounts might have carbonated eclogite in the source mantle.
7 DATA AVAILABILITY STATEMENTAll data generated and/or analyzed during this study are available from the corresponding author upon reasonable request.
8 ACKNOWLEDGMENTWe are thankful to the crew and scientists involved with the Magellan Seamounts cruise. We appreciate the help of XUE Dingshuai and LIU Yanhong of Institute of Geology and Geophysics, the Chinese Academy of Sciences, for the major elements analysis. We appreciate the help of ZHU Jihao of Second Institute of Oceanography, Ministry of Nature Resources, for the trace elements analysis. Thanks to the two reviewers for their valuable suggestions.
Electronic supplementary material
Supplementary material (Supplementary Tables S1–S4) is available in the online version of this article at https://doi.org/10.1007/s00343-020-0013-x.
Beccaluva L, Barbieri M, Born H, Brotzu P, Coltorti M, Conte A, Garbarino C, G C B G, M A, Morbidelli L, Ruberti E, Siena F, Traversa G. 1992. Fractional crystallization and liquid immiscibility processes in the alkaline-carbonatite complex of Juquiá (São Paulo, Brazil). Journal of Petrology, 33(6): 1371-1404.
DOI:10.1093/petrology/33.6.1371 |
Bu W R, Shi X F, Zhang M J, Liu J H, Glasby G P. 2007. He, Ne and Ar isotopic composition of Fe-Mn crusts from the western and central Pacific Ocean and implications for their genesis. Science in China Series D: Earth Sciences, 50(6): 857-868.
DOI:10.1007/s11430-007-0011-2 |
Chazot G, Menzies M A, Harte B. 1996. Determination of partition coefficients between apatite, clinopyroxene, amphibole, and melt in natural spinel lherzolites from Yemen: implications for wet melting of the lithospheric mantle. Geochimica et Cosmochimica Acta, 60(3): 423-437.
DOI:10.1016/0016-7037(95)00412-2 |
Coltorti M, Bonadiman C, Hinton R W, Siena F, Upton B G J. 1999. Carbonatite metasomatism of the oceanic upper mantle: evidence from clinopyroxenes and glasses in ultramafic xenoliths of Grande Comore, Indian Ocean. Journal of Petrology, 40(1): 133-165.
DOI:10.1093/petroj/40.1.133 |
Dasgupta R, Hirschmann M M, Smith N D. 2007b. Water follows carbon: CO2 incites deep silicate melting and dehydration beneath mid-ocean ridges. Geology, 35(2): 135-138.
DOI:10.1130/G22856A.1 |
Dasgupta R, Hirschmann M M, Smith N D. 2007a. Partial melting experiments of peridotite + CO2 at 3 GPa and genesis of alkalic ocean island basalts. Journal of Petrology, 48(11): 2093-2124.
DOI:10.1093/petrology/egm053 |
Dasgupta R, Hirschmann M M, Stalker K. 2006. Immiscible transition from carbonate-rich to silicate-rich melts in the 3 GPa melting interval of eclogite + CO2 and genesis of silica-undersaturated ocean island lavas. Journal of Petrology, 47(4): 647-671.
DOI:10.1093/petrology/egi088 |
Gerbode C, Dasgupta R. 2010. Carbonate-fluxed melting of MORB-like pyroxenite at 2·9 GPa and genesis of HIMU Ocean Island Basalts. Journal of Petrology, 51(10): 2067-2088.
DOI:10.1093/petrology/egq049 |
Green D H, Wallace M E. 1988. Mantle metasomatism by ephemeral carbonatite melts. Nature, 336(6198): 459-462.
DOI:10.1038/336459a0 |
Hammouda T, Moine B N, Devidal J L, Vincent C. 2009. Trace element partitioning during partial melting of carbonated eclogites. Physics of the Earth and Planetary Interiors, 174(1-4): 60-69.
DOI:10.1016/j.pepi.2008.06.009 |
Hart R A. 1973. A model for chemical exchange in the basaltseawater system of oceanic layer ò. Canadian Journal of Earth Sciences, 10(6): 799-816.
DOI:10.1139/e73-074 |
Hauri E H, Shimizu N, Dieu J J, Hart S R. 1993. Evidence for hotspot-related carbonatite metasomatism in the oceanic upper mantle. Nature, 365(6443): 221-227.
DOI:10.1038/365221a0 |
Hirose K, Kushiro I. 1993. Partial melting of dry peridotites at high pressures: determination of compositions of melts segregated from peridotite using aggregates of diamond. Earth and Planetary Science Letters, 114(4): 477-489.
DOI:10.1016/0012-821X(93)90077-M |
Hirose K. 1997. Partial melt compositions of carbonated peridotite at 3 GPa and role of CO2 in alkali-basalt magma generation. Geophysical Research Letters, 24(22): 2837-2840.
DOI:10.1029/97GL02956 |
Hoernle K, Tilton G, Le Bas M J, Duggen S, Garbe-Schönberg D. 2002. Geochemistry of oceanic carbonatites compared with continental carbonatites: mantle recycling of oceanic crustal carbonate. Contributions to Mineralogy and Petrology, 142(5): 520-542.
DOI:10.1007/s004100100308 |
Hofmann A W, Jochum K P, Seufert M, White W M. 1986. Nb and Pb in oceanic basalts: new constraints on mantle evolution. Earth and Planetary Science Letters, 79(1-2): 33-45.
DOI:10.1016/0012-821X(86)90038-5 |
Ivanikov V V, Rukhlov A S, Bell K. 1998. Magmatic evolution of the melilitite-carbonatite-nephelinite dyke series of the Turiy Peninsula (Kandalaksha Bay, White Sea, Russia). Journal of Petrology, 39(11-12): 2043-2059.
DOI:10.1093/petroj/39.11-12.2043 |
Kawamoto T, Holloway J R. 1997. Melting temperature and partial melt chemistry of H2O-saturated mantle peridotite to 11 gigapascals. Science, 276(5310): 240-243.
DOI:10.1126/science.276.5310.240 |
Kiseeva E S, Yaxley G M, Hermann J, Litasov K D, Rosenthal A, Kamenetsky V S. 2012. An experimental study of carbonated eclogite at 3·5-5·5 GPa-implications for silicate and carbonate metasomatism in the cratonic mantle. Journal of Petrology, 53(4): 727-759.
DOI:10.1093/petrology/egr078 |
Klemme S, van der Laan S R, Foley S F, Günther D. 1995. Experimentally determined trace and minor element partitioning between clinopyroxene and carbonatite melt under upper mantle conditions. Earth and Planetary Science Letters, 133(3-4): 439-448.
DOI:10.1016/0012-821X(95)00098-W |
Kogarko L, Kurat G, Ntaflos T. 2001. Carbonate metasomatism of the oceanic mantle beneath Fernando de Noronha Island, Brazil. Contributions to Mineralogy and Petrology, 140(5): 577-587.
DOI:10.1007/s004100000201 |
Koppers A A P, Staudigel H, Pringle M S, Wijbrans J R. 2003. Short-lived and discontinuous intraplate volcanism in the South Pacific: hot spots or extensional volcanism?. Geochemistry, Geophysics, Geosystems, 4(10): 1089.
DOI:10.1029/2003GC000533 |
Koppers A A P, Staudigel H, Wijbrans J R, Pringle M S. 1998. The Magellan seamount trail: implications for cretaceous hotspot volcanism and absolute Pacific Plate motion. Earth and Planetary Science Letters, 163(1-4): 53-68.
DOI:10.1016/S0012-821X(98)00175-7 |
Le Maitre R W. 1984. A proposal by the IUGS subcommission on the systematics of igneous rocks for a chemical classification of volcanic rocks based on the total alkali silica (TAS) diagram. Australian Journal of Earth Sciences, 31(2): 243-255.
DOI:10.1080/08120098408729295 |
Lee W J, Wyllie P J. 1998. Petrogenesis of carbonatite magmas from mantle to crust, constrained by the system CaO-(MgO+FeO*)-(Na2O+K2O)-(SiO2+Al2O3+TiO2)-CO2. Journal of Petrology, 39(3): 495-517.
DOI:10.1093/petroj/39.3.495 |
Li D P. 2010. In-depth discussion of tectonic geological characteristics of the Pacific Ocean and the spatial and temporal distribution of seamounts. Science and Technology Innovation Herald, (25): 127-128.
(in Chinese) DOI:10.3969/j.issn.1674-098X.2010.25.105.(inChinese) |
Liang D H, Zhu B D. 2002. The region geological bachground of cobalt-rich crust formation form the Magellanean Seamount chain. Geological Research of South China Sea, 14: 84-90.
(in Chinese with English abstract) |
McDonough W F, Sun S S. 1995. The composition of the earth. Chemical Geology, 120(3-4): 223-253.
DOI:10.1016/0009-2541(94)00140-4 |
Müller R D, Sdrolias M, Gaina C, Roest W R. 2008. Age, spreading rates, and spreading asymmetry of the worldos ocean crust. Geochemistry, Geophysics, Geosystems, 9(4): Q04006.
DOI:10.1029/2007GC001743 |
Neumann E R, Wulff-Pedersen E, Pearson N J, Spencer E A. 2002. . Mantle xenoliths from Tenerife (Canary Islands):evidence for reactions between mantle peridotites and silicic carbonatite melts inducing Ca metasomatism. Journal of Petrology, 43(5): 825-857.
DOI:10.1093/petrology/43.5.825 |
Pan J H, Liu S Q, Luo Z H, You G Q. 2007. Modes of occurrence and characteristics of phosphorates on Pacific Guyots and their genetic significance. Mineral Deposits, 26(2): 195-203.
(in Chinese with English abstract) DOI:10.3969/j.issn.0258-7106.2007.02.006. |
Pan J H, Liu S Q, Yang Y, Liu X Q. 2002. Research on geochemical characteristics of major, trace and rare-earth elements in phosphates from the west pacific seamounts. Geological Review, 48(5): 534-541.
(in Chinese with English abstract) DOI:10.3321/j.issn:0371-5736.2002.05.012. |
Pan J H, Liu S Q. 1999. Distribution, composition and element geochemistry of Co-rich crusts in the Western Pacific. Acta Geoscientia Sinica, 20(1): 47-54.
(in Chinese with English abstract) DOI:10.3321/j.issn:1006-3021.1999.01.007. |
Ren J B, Wei Z Q. 2017. The discovery and implication of REE-rich phosphate in deep-sea. Geological Review, 63(S1): 13-14.
(in Chinese with English abstract) |
Ren X W, Liu J H, Shi X F, Cui Y C, Lin X H. 2011. Genesis and ore-forming stages of co-rich ferromanganese crusts from seamount M of Magellan Seamounts: evidence from geochemistry and Co chronology. Marine Geology & Quaternary Geology, 31(6): 65-74.
(in Chinese with English abstract) |
Rudnick R L, McDonough W F, Chappell B W. 1993. Carbonatite metasomatism in the Northern Tanzanian mantle: petrographic and geochemical characteristics. Earth and Planetary Science Letters, 114(4): 463-475.
DOI:10.1016/0012-821X(93)90076-L |
Smith W H F, Staudigel H, Watts A B, Pringle M S. 1989. The Magellan Seamounts: early cretaceous record of the south Pacific isotopic and thermal anomaly. Journal of Geophysical Research, 94(B8): 10501-10523.
DOI:10.1029/JB094iB08p10501 |
Song Z Z, Yang Y, Gao H Y. 2009. Determination and implication for paleoceanography study of fluorine of iron-manganese crusts from the Magellan Seamounts. Geological Science and Technology Information, 28(5): 91-95.
(in Chinese with English abstract) DOI:10.3969/j.issn.1000-7849.2009.05.013. |
Staudigel H, Hart S R, Richardson S H. 1981. Alteration of the oceanic crust: processes and timing. Earth and Planetary Science Letters, 52(2): 311-327.
DOI:10.1016/0012-821X(81)90186-2 |
Sun S S, McDonough W F. 1989. Chemical and isotopic systematics of oceanic basalts: implications for mantle composition and processes. In: Saunders A D, Norry M J eds. Magmatism in the Ocean Basins. Geological Society, London, Special Publications. p.313-345, https://doi.org/10.1144/GSL.SP.1989.042.01.19.
|
Sun X M, Xue T, He G W, Ye X R, Zhang M, Lu H F, Wang S W. 2006. Noble gases isotopic compositions and sources of cobalt-rich crusts from west Pacific Ocean seamounts. Acta Petrologica Sinica, 22(9): 2331-2340.
(in Chinese with English abstract) DOI:10.3969/j.issn.1000-0569.2006.09.008. |
Tang L M, Dong Y H, Chu F Y, Chen L, Ma W L, Liu Y G. 2019. Geochemistry and age of seamounts in the west Pacific: mantle processes and petrogenetic implications. Acta Oceanologica Sinica, 38(1): 71-77.
DOI:10.1007/s13131-019-1371-0 |
Wang L L, Zhong H X, Zeng F C, Li Y, Zhu K C. 1999. Interelement relationships and their implications for crust genesis in cobalt-rich ferromanganese crusts from the MA and Mc seamounts of the Magellan Seamounts. Gresearch of Eological South China Sea: 26-46.
(in Chinese with English abstract) |
Wang Y M, Zhang H D, Liu J H, Zhang X H, Zhu B D. 2016. Abundances and spatial distributions of associated useful elements in co-rich crusts from Caiwei Seamount in Magellan Seamounts. Marine Geology & Quaternary Geology, 36(2): 65-74.
(in Chinese with English abstract) DOI:10.16562/j.cnki.0256-1492.2016.02.008. |
Weaver B L. 1991. The origin of ocean island basalt endmember compositions: trace element and isotopic constraints. Earth and Planetary Science Letters, 104(2-4): 381-397.
DOI:10.1016/0012-821X(91)90217-6 |
Wei Z Q, Deng X G, Zhu K C, Yao H Q, Yang Y, Ren J B. 2017a. Characteristic of substrate rocks of Caiwei Seamounts in the West Pacific Ocean. Marine Geology Frontiers, 33(12): 1-6.
(in Chinese with English abstract) DOI:10.16028/j.1009-2722.2017.12001. |
Wei Z Q, He G W, Deng X G, Yao H Q, Liu Y G, Yang Y, Ren J B. 2017b. The progress in the study and survey of oceanic cobalt-rich crust resources. Geology in China, 44(3): 460-472. (in Chinese with English abstract)
|
Ye L J, Chen Q Y, Zhao D X, Chen Z M, Chen Y M, Liu K W. 1989. The Phosphorites of China. Science Press, Beijing, China. p.169-196. (in Chinese)
|
Ye X R, Fang N Q, Ding L, Zhang M J. 2008. The noble gas contents and helium and argon isotopic compositions in the cobalt-rich crusts from the Magellan Seamounts. Acta Petrologica Sinica, 24(1): 185-192.
(in Chinese with English abstract) |
Zhang G L, Chen L H, Jackson M G, Hofmann A W. 2017. Evolution of carbonated melt to alkali basalt in the South China Sea. Nature Geoscience, 10(3): 229-235.
DOI:10.1038/ngeo2877 |
Zhang G L, Smith-Duque C. 2014. Seafloor basalt alteration and chemical change in the ultra thinly sedimented South Pacific. Geochemistry, Geophysics, Geosystems, 15(7): 3066-3080.
DOI:10.1002/2013GC005141 |
Zhang W Y, Zhang F Y, Zhu K C, Yang K H, Hu G D, Cheng Y S, Li S J, Zhao H Q. 2009. Fractal research on seamount topography in the West Pacific Ocean. Geoscience, 23(6): 1138-1146.
(in Chinese with English abstract) DOI:10.3969/j.issn.1000-8527.2009.06.020. |
Zhao L H, Jin X L, Gao J Y, Li J B, Chu F Y. 2010. The research on the drifting history and possible origin of the Magellan Seamount Trail. Acta Oceanologica Sinica, 32(3): 60-66.
(in Chinese with English abstract) |
Zhu K C, Zhao Z B, Li Y. 2001. Cobalt-rich ferromanganese crusts from the MA, ME and MF seamounts of the Magellan Seamounts. Marine Geology & Quaternary Geology, 21(1): 33-38.
(in Chinese with English abstract) DOI:10.16562/j.cnki.0256-1492.2001.01.006. |
Zhu K C. 2002. Petrology of the substrate in seamounts MA, MC, MD, ME and MF from Magellan Seamounts. Marine Geology & Quaternary Geology, 22(1): 49-56.
(in Chinese with English abstract) DOI:10.16562/j.cnki.0256-1492.2002.01.008. |
 2020, Vol. 38
2020, Vol. 38