Institute of Oceanology, Chinese Academy of Sciences
Article Information
- ZHANG Xiaosen, XU Xinyu, Jane M. REED
- Sedimentary diatom and pigment-inferred recent anthropogenic accelerated eutrophication of a Mediterranean lake (Lake Dojran, Republic of North Macedonia/Greece)
- Journal of Oceanology and Limnology, 38(6): 1787-1798
- http://dx.doi.org/10.1007/s00343-020-9320-5
Article History
- Received Dec. 25, 2019
- accepted in principle Feb. 17, 2020
- accepted for publication Apr. 11, 2020
2 Department of Geography, Geology and Environment, University of Hull, Hull HU6 7RX, UK
Anthropogenic nutrient pollution and lake eutrophication have become one of the most widespread, costly and challenging environmental and ecological problems. Nitrogen and phosphorus pollutions have exceeded the earth sustainable boundaries (Rockström et al., 2009; Steffen et al., 2015) and impacted economic growth and human health (Elser and Bennett, 2011; Sutton et al., 2011). Management measures, consumer actions and technological innovation are being made to control both anthropogenic phosphorus and nitrogen diffuse and attempt to mitigate harmful algal blooms (Conley et al., 2009; Paerl et al., 2016), and there are now many success stories (Verdonschot et al., 2013), for example, Lake Geneva (France/Switzerland) (Rimet et al., 2009). However, lack of apparent recovery following restricted nutrient loading is mostly common (Bennion et al., 2015), for example, Lake Taihu (China) (Wang et al., 2018; Wan et al., 2019), and more importantly, the target of lake ecological restoration and the timescale to achieve the expectation are still unclear, which is linked to individual unique lake properties and complex lake ecosystems (Bennion et al., 2015). It is thus of primary importance to understand the pathway of human-induced water quality degradation and define the lake reference condition.
Palaeolimnological analysis of lake sediment records can provide insights into tracking humaninduced changes of lake trophic status and defining the baseline condition and restoration target (Bennion and Battarbee, 2007; Bennion et al., 2011). Diatoms (Bacillariophyceae) are unicellular algae with a siliceous cell wall, and their silica frustules often preserve well in lake sediments. Due to their sensitivity to a wide range of water chemistry variables, diatoms are recognised as a powerful proxy for past environmental and ecological change (Smol and Stoermer, 2010), and in particular, they offer excellent potential for revealing recent humaninduced lake degradation (Bennion et al., 2010; Hall and Smol, 2010). Algae (diatoms, chlorophytes, cyanobacteria, etc.) are dominant primary producers in lake ecosystems, and as algal preservation in lake sediments can be severely biased by the dissolution of some algal groups while algal pigments can be well preserved, pigments are often taken as a strong proxy for both lake productivity change and the shift of algal structure composition.
The Mediterranean region is a climatically transitional zone under the influence of the interaction of the mid-latitude westerlies and the subtropical high-pressure system (i.e. the Azores High), with most of the region controlled by a dry Mediterraneantype climate (Lionello, 2012), and lakes in this region are usually shallow with high conductivity and even saline water (Roberts and Reed, 2009). The potential for palaeolimnological research on recent water quality degradation is largely untapped across the Mediterranean, and the exploration of integrating diatoms and pigments to elucidate recent lake trophic changes also has not been conducted so far in this region. Lake Dojran is a shallow and eutrophic lake in the north-eastern Mediterranean region, and it has been taken as a key site to investigate Mediterranean climate change during the Holocene period (i.e. the past 11 500 years) through palaeolimnological analysis (Francke et al., 2013; Zhang et al., 2014; Masi et al., 2018). Holocene diatom record reveals that both regional climate change and local catchment dynamics affect the changes of lake level and trophic status in Lake Dojran (Zhang et al., 2014); however, associated with intensified human impact and increased catchment influence during the late Holocene (Athanasiadis et al., 2000; Masi et al., 2018), diatom evidence for major lake ecological shift is weak (Griffiths et al., 2002; Zhang et al., 2014). Whereas, modern limnological surveys on algal composition indicate human-induced dramatic water level fluctuation and lake eutrophication in recent decades (Levkov and Stojanovski, 2000-2001; Griffiths et al., 2002; Sotiria and Petkovski, 2004). This thus emphasises the importance of palaeolimnological analysis in tracking thoroughly the path of human-induced lake trophic change in the past, but also it is essential to combine diatoms and pigments to investigate the change of lake ecological state.
2 SITE DESCRIPTIONLake Dojran (Fig. 1, 41°12′N, 22°44′E, 144 m a.s.l.) is a transboundary lake between the Republic of North Macedonia (i.e. North Macedonia) and the Hellenic Republic (i.e. Greece) in the north-eastern Mediterranean region. The lake receives its water from small rivers, creeks and springs, and loses it through evaporation and groundwater outflow today, but surface outflow (i.e. the Gjolaja River) was possible at the southern end of the lake in previous phases of high water level, which drained into the Vardar River, a major river in North Macedonia and Greece, and then entered the Aegean Sea (Sotiria and Petkovski, 2004). Maximum lake level was 10.0 m in 1951–1987, decreased to 3.7 m in 2002 due to water abstraction and agricultural practices (Griffiths et al., 2002), and recovered to 6.7 m in 2010 due to decreased water use and additional water transfer into the lake (Popovska and Bonacci, 2008; Stojov, 2012). Total phosphorus (TP) concentration was 15–130 μg/L in 1953–1960 (Sotiria and Petkovski, 2004), and the lake was consistently eutrophic with the minimum TP value >50 μg/L since 1996 (Temponeras et al., 2000; Lokoska et al., 2006; Tasevska et al., 2010). Due to the influence of the typical Mediterranean climate (hot, dry summers and mild, humid winters) and the very simple plate-shaped morphometry of the lake basin, Lake Dojran's water warms up easily and does not stratify in summer, but it can freeze in winter. Reed beds occupy the fringe of the lake, and submerged plants are common in the littoral zone of the lake. Lake Dojran is currently a eutrophic, shallow and well-mixed lake.

|
| Fig.1 The location of Lake Dojran in the north-eastern Mediterranean region (a) and the catchment of this lake between North Macedonia and Greece and the coring site Co1260 (Francke et al., 2013) (b; modified from Zhang et al., 2014) |
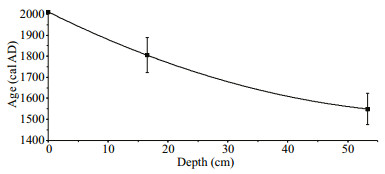
The present study is based on the topmost 55 cm section (undisturbed) of a long core Co1260 (Fig. 1; Francke et al., 2013; Zhang et al., 2014) in Lake Dojran, which was obtained in June 2011 from the deepest, south-central part of the lake using UWITEC gravity and piston coring equipments. The core was split into two halves in the laboratory. One half was subsampled and freeze-dried, and the other half was sealed airtight and stored in darkness at 4℃. The age model of this core section was estimated separately in this study. As 137Cs and 210Pb dating methods linked to nuclear bombing apply only to the past several decades, we relied on radiocarbon dating in spite of the uncertainty of 14C age calibration for recent samples. Two radiocarbon dates were obtained from the University of Cologne (Germany), of which one from terrestrial plant macrofossil at 53.3 cm depth is 360±70 14C BP (1 549±74 cal AD) and the other from bulk organic matter at 16.5 cm depth is 140±35 14C BP (1 805±83 cal AD). The 14C date from terrestrial plant remains provides reliable age control for the bottom of this core section, and the 14C date from bulk organic matter is possibly influenced by the carbon reservoir effect. We selected to generate a smooth curve between the two dated levels and the sampling date rather than simply using linear interpolation to develop the age model. The age model was established through quadratic polynomial interpolation (the function is y=0.1x2–14.2x+2011) between the two calibrated radiocarbon ages and the year 2011 AD of the surface sediment. We also consider the uncertainty of the age model when interpreting the diatom and pigment data below. The calibration of radiocarbon ages into calendar ages is based on OxCal 4.3 software (Bronk Ramsey, 2009) and IntCal13 calibration curve (Reimer et al., 2013). The age model of this sequence reflects the sedimentation during the past ca. 450 years (Fig. 2).
|
| Fig.2 The age-depth model of the 55 cm-long sequence in this study, which is developed through quadratic polynomial interpolation (the function is y=0.1x2–14.2x+2011) between the year 2011 AD of the surface sediment and two calibrated radiocarbon ages |
Diatom analysis was carried out on 14 freeze-dried sediment samples taken at 4-cm intervals. Standard techniques in Battarbee et al. (2001) were followed for diatom slide preparation. Approximately 0.1-g dry sediment was heated in 30-mL 30% H2O2 to oxidise organic matter, and then a few drops of concentrated HCl were added to remove carbonates (Battarbee et al., 2001). Diatom suspension was diluted to an appropriate concentration, and known quantities of microspheres were added for the calculation of absolute diatom valve concentration. Diatom slides were mounted using Naphrax™. Diatom valves were counted at ×1 000 magnification under oil immersion on an OLYMPUS BX51 light microscope, and more than 300 valves per slide were counted. Diatom fragments were also counted, and were used to potentially assess the quality of diatom preservation. Diatoms were identified mainly following four classic European diatom books published in German language in 1986–1991 (Krammer and LangeBertalot, 1986, 1988, 1991a, b), and the identification of diverse benthic Navicula and Cymbella sensu lato species was also based on Lange-Bertalot (2001) and Krammer (2002), respectively. To identify accurately the planktonic Stephanodiscus and Cyclotella sensu lato species in this study, we referred mainly to recent Václav Houk, Rolf Klee and Hiroyuki Tanaka's monographs (Houk et al., 2010, 2014). Stephanodiscus minutulus (Kützing) Cleve & Möller and Stephanodiscus parvus Stoermer & Håkansson are merged into S. minutulus/parvus. Stephanodiscus minutulus and S. parvus have similar morphological characteristics and ecological preferences, and it is difficult to separate them consistently under the light microscope (Hobbs et al., 2011; Bennion et al., 2012). It is also possible that S. parvus is a synonym of S. minutulus that is a polymorphic taxon (Scheffler and Morabito, 2003; Cruces et al., 2010). In addition, we referred to Zlatoko Levkov's taxonomical work in this region (Levkov et al., 2007; Levkov, 2009). We adopted the up-to-date nomenclature from the Catalogue of Diatom Names (Fourtanier and Kociolek, 2011) and the AlgaeBase database (Guiry and Guiry, 2019).
Pigment analysis was based on 14 wet sediment samples taken at 4-cm intervals, and for comparison with diatom data, these wet samples are taken from the same depths of the core section as the dry samples above for diatom analysis. They were stored in darkness at 4℃ after core recovery and freeze-dried shortly before analysis. Five dry sediment samples, taken at 12 cm intervals and corresponding to No. 1, 4, 7, 10 and 13 of the 14 wet pigment samples, were also analysed for pigments, and similar to diatom samples, they were freeze-dried immediately after core recovery and stored at room temperature. The pigment analysis of these five dry sediment samples is used to evaluate the degradation of pigments in referring to the results of the corresponding wet sediments. Approximately 0.2-g freeze-dried sediment was soaked in 5-mL extraction solvent (a mixture of 80% acetone, 15% methanol and 5% water) overnight for pigment extraction (McGowan, 2013). The solvent pigment extracts were dried under N2 gas, re-dissolved in 500-μL injection solvent (a 70꞉25꞉5 mixture of acetone, ion-pairing reagent and methanol), and then separated through the highperformance liquid chromatography (HPLC) based on pigment polarity and mass (McGowan, 2013). Pigments were identified according to their spectral characteristics and retention times, by referring to a range of standard phytoplankton pigment data and graphics (Jeffrey et al., 1997; Egeland et al., 2011). Pigment quantification is based on the conversion of the chromatogram peak area to the concentration, and pigment concentration is expressed as nanomoles per g total organic carbon (nmol/g TOC).
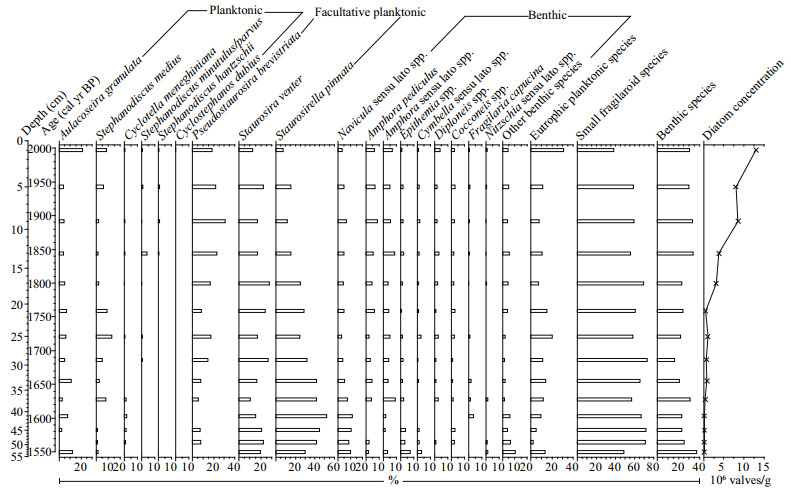
4 RESULT AND DISCUSSION 4.1 Diatom dissolution and preservationMore than 300 diatom valves are counted per sample, and diatom valve concentration is >105/g throughout the sequence. Small fragilaroid species, comprising Pseudostaurosira brevistriata (Grunow) Williams & Round, Staurosira venter (Ehrenberg) Cleve & Möller and Staurosirella pinnata (Ehrenberg) Williams & Round, are consistently abundant throughout the sequence, and benthic species are diverse and also consistently at relatively high abundance (Fig. 3). This is coherent with the currently shallow water state and extensively distributed reed beds of the lake. Planktonic taxa comprise mainly typical eutrophic species, and eutrophic Aulacoseira granulata (Ehrenberg) Simonsen, Stephanodiscus medius Håkansson, Cyclotella meneghiniana Kützing, Stephanodiscus minutulus/parvus, Stephanodiscus hantzschii Grunow and Cyclostephanos dubius (Hustedt) Round occur in this sequence (Fig. 3). The common occurrence of eutrophic planktonic diatom taxa is consistent with the currently eutrophic, shallow and well-mixed lake water state. Thus, the observed diatom assemblage composition in the lake sediment is closely linked with the limnological and ecological states of the lake, and it is possible that diatom dissolution is minor and diatoms can be considered to be well preserved in this sequence. A simple diatom dissolution index (F index; Ryves et al., 2001), i.e. the ratio of pristine valves to total valves, is often used to assess the preservation of diatoms, but this index is deemed inappropriate for this sequence, due to the shifts of main taxa and their different degrees of dissolution, i.e. taphonomic bias, and the fragmentation in some degree during the process of diatom slide preparation. However, both robust, highly-silicified taxa (A. granulata, S. medius, Epithemia species, etc.) and small taxa (P. brevistriata, S. venter, Amphora pediculus (Kützing) Grunow, etc.) are consistently observed throughout the sequence (Fig. 3). Thus, although diatom dissolution cannot be assessed quantitatively, empirical observation results show that the diatom data of this sequence can be reliably used as an indicator for recent environmental change.
|
| Fig.3 Summary diagram of sedimentary diatom assemblage composition with the groups of eutrophic planktonic species, small fragilaroid species and benthic species, and diatom concentration in this study |
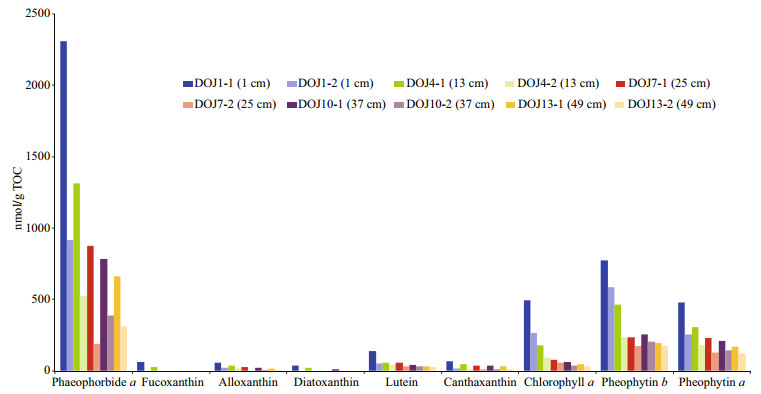
Totally nine pigments are identified in the chromatograms of these samples, and they are phaeophorbide a, fucoxanthin, alloxanthin, diatoxanthin, lutein, canthaxanthin, chlorophyll a, pheophytin b and pheophytin a (Figs. 4 & 5), which are listed here according to their retention times. Pigment concentrations are expressed in nanomoles per gram total organic carbon (nmol/g TOC) based on their peak areas in the chromatograms. The degradation of pigments is evaluated through the comparison of pigment concentrations in the five dry sediment samples, lyophilized immediately after core recovery and stored under ambient temperature, with their corresponding wet sediment samples that are kept in cold storage until the analysis. Compared with the wet sediment samples, no new pigment derivative occurs in the five dry samples, and the nine pigments exhibit different degrees of degradation with fucoxanthin and diatoxanthin declining even below the limit of detection (Fig. 4). Although this is related with the initial concentrations of pigments as in the wet samples, this shows clearly the different levels of stability of pigments and confirms the important role of sediment storage conditions in pigment analysis (Reuss and Conley, 2005). Exposure to heat, light and oxygen enhances pigment degradation, with the ambient condition increasing exposure to heat and light and the powdery texture of dry sediment maximizing exposure to oxygen (Leavitt and Hodgson, 2001), and Reuss and Conley (2005) suggested that freeze-dried sediments should be also stored in freezer. Fucoxanthin contains an epoxy group, which makes the pigment more susceptible to degradation (Reuss and Conley, 2005). Whereas, diatoxanthin, lutein, canthaxanthin, and alloxanthin do not have such an epoxy group and thus are more stable than fucoxanthin, although their degradation also occurs. This study shows that lutein is the most stable one of the five pigments above (Fig. 4). Carotenoids contain a long chain of alternating double bonds which makes them susceptible to degradation, and their breakdown products are colourless and undetectable; whereas, chlorophylls contain a tetrapyrrole ring, and their degradation products of different types (i.e. oxidation, loss of Mg and loss of phytol chain) are still coloured and can be detected (McGowan, 2013). Chlorophyll a is unstable and its degradation product pheophytin a is quite stable (Reuss and Conley, 2005). This study confirms the high stability of pheophytin a and shows that phaeophorbide a is a relatively unstable degradation product of chlorophyll a (Fig. 4). Veuger and van Oevelen (2011) suggested that a simple index, i.e. the ratio of chlorophyll a derivatives to chlorophyll a, could be used to indicate pigment preservation; however, due to the different levels of stability of phaeophorbide a and pheophytin a, this index is not suitable in this study. Chlorophyll b is not detected in this study and shows high instability in most cases, while its derivative pheophytin b is highly stable. Thus, sediment storage conditions are key factors for the accuracy of pigment analysis (Reuss and Conley, 2005), and the wet sediment samples freeze-dried just before the analysis in this study can be used as a proxy for recent environmental change.
|
| Fig.4 Comparison of pigment concentrations between five wet sediment samples in dark and cold storage before analysis (DOJ*-1) and their corresponding dry sediment samples lyophilized immediately after the recovery (DOJ*-2) * indicates the sample number with its depth followed in brackets. |
|
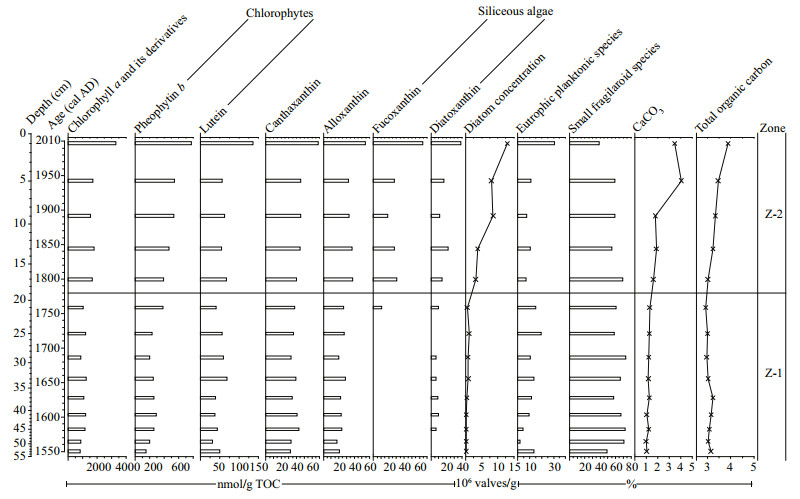
| Fig.5 Summary diagram of sedimentary chlorophyll and xanthophyll pigment concentrations in this study and comparison with diatom, carbonate and organic matter content data from the same sequence |
Based on the evaluations of diatom dissolution/ preservation and pigment degradation/preservation above, accurate and unbiased results about the change of lake ecological status can be obtained from this sequence. This sequence can be distinctly divided into two zones with a major ecological shift occurring at around 1770 cal AD based on pigment concentration data, which is consistent with the changes of diatom concentration and geochemical proxies (i.e. the contents of carbonates and organic matter in the sediments) (Fig. 5). Although there is also a clear shift from S. pinnata to P. brevistriata in the diatom assemblage composition (Fig. 3), it is well known that the two species have very wide nutrient tolerances and similar habitat preferences and thus the diatom composition data do not show a tipping point of ecological state in this lake during the past ca. 450 years.
Chlorophyll a is a widely distributed pigment that originates from all algae and aquatic plants as well as riverine terrestrial plant detritus (Leavitt and Hodgson, 2001). However, due to its exposure to oxygen at the soil surface, chlorophyll a is poorly preserved in the terrestrial detrital material (Lami et al., 2000), particularly under the influence of human-induced deforestation and agricultural development in the catchment of this lake (Athanasiadis et al., 2000; Masi et al., 2018). Thus, the concentration of chlorophyll a and its derivatives indicates primarily total algal production and can be used as a proxy to infer the change of lake productivity (Leavitt and Hodgson, 2001; McGowan, 2013). In Fig. 5, the lower concentration of chlorophyll a and its derivatives in Zone Z-1 (55–19 cm, ca. 1550–1770 cal AD) is largely attributed to lower lake productivity rather than higher pigment degradation, and their higher concentration in Zone Z-2 (19–0 cm, ca. 1770–2010 cal AD), particularly the higher concentration of phaeophorbide a that derives mainly from planktonic algae under the condition of zooplankton grazing (Guilizzoni and Lami, 2002), indicates higher lake productivity. The increase of lake productivity is probably linked to a human-induced dramatic lakelevel decline rather than climate change. The maximum lake level was approximately 15 m at the end of the 18th century, and in the year 1808, a channel was dug at the southern end of the lake to connect the Gjolaja River. This is the most influential water abstraction practice in this lake during the past 450 years, and in order to reduce the lake area for larger agricultural land and take more water for downstream irrigation, this channel was deepened gradually since then and the maximum lake level declined to less than 4 m in 2002 (Popovska et al., 2005; Stojov, 2012). The lake littoral zone and its macrophytic vegetation also reduced (Griffiths et al., 2002; Sotiria and Petkovski, 2004). Although recent climate warming can promote chlorophyll a concentration and lake algal production (Smol, 2019 and references therein), this rapid climate warming arose in the 20th century (IPCC, 2013) and is much later than the occurrence of the ecological shift in this lake. The slight chronological difference between the reconstructed lake ecological breakpoint and the documented causal human activity could be attributed to the possible carbon reservoir effect of the bulk organic matter date and the uncertainty of the age model. In all, the increase in the concentration of chlorophyll a and its derivatives is linked with increased algal blooms and accelerated lake eutrophication since the 19th century under the influence of intensified anthropogenic water reduction and agricultural nutrient diffuse.
Most xanthophylls are used as indicators for specific algal classes or functional groups (Leavitt and Hodgson, 2001). In Fig. 5, higher lutein and pheophytin b concentrations in Zone Z-2 (19–0 cm, ca. 1770–2010 cal AD) indicate increased chlorophyte production (McGowan et al., 2012). The slightly higher concentrations of canthaxanthin and alloxanthin in this zone indicate increased production of cyanobacteria and planktonic cryptophytes, respectively (Leavitt and Hodgson, 2001; Guilizzoni and Lami, 2002; McGowan, 2013). Distinctly increased fucoxanthin and diatoxanthin concentrations result from increased production of siliceous algae, particularly diatom production, rather than decreased diatom dissolution, and this is consistent with distinctly increasing diatom concentration in this zone. There is no clear changing trend of the relative abundances of individual eutrophic planktonic diatom species as well as the sum of these taxa in this study (Fig. 3), and another palaeolimnoligical analysis in this lake conducted by Griffiths et al. (2002) also does not show the response of diatom assemblage composition to the accelerated lake eutrophication. This can be probably attributed to the dominance of facultative planktonic fragilaroid taxa rather than planktonic taxa in the diatom assemblage composition and the wide environmental tolerances of the fragilaroid species. Small fragilaroid species can tolerate a wide range of trophic states and survive in a variety of benthic habitats consisting of lake sediments, submerged plants and the base of emergent plants (Sayer, 2001). Because non-planktonic diatoms can derive nutrients from sediments and macrophytes, they respond more directly and sensitively to habitat availability rather than water-column nutrient enrichment (Bennion et al., 2010; Hall and Smol, 2010). Although lake littoral zone and its macrophytic vegetation reduced under human agricultural activities as mentioned above, the decline of both lake area and water level would make the previous pelagic zone become shallow and available for the fragilaroid species to attach to the sediments, due to the simple plate-shaped morphometry of the lake basin. This is a possible reason that the fragilaroid species are consistently at high relative abundance and dominate the diatom assemblage composition in this sequence.
The chlorophyll and xanthophyll pigment concentration data and diatom concentration data show an increase in the production of diatoms, chlorophytes, cyanobacteria and planktonic cryptophytes, indicating increased algal biomass and lake productivity, which is linked to water abstraction practices and more intensive agricultural activities since the 19th century. This is also supported by increasing organic matter content (i.e. total organic carbon (TOC)) and distinctly increasing carbonate content from the same sequence, both of which are mainly derived from the in-lake authigenic origin (Francke et al., 2013). Although diatom assemblage composition does not show evidence for the accelerated lake eutrophication, other types of algae could respond and/or new toxic algal species could occur in such a nutrient-enriched environment, which is supported by the algal investigations in recent decades (Sotiria and Petkovski, 2004; Lešoski et al., 2010). Nutrient-polluted lakes often suffer from harmful cyanobacterial blooms and nuisance weeds. It is possible that chlorophytes and/or cyanobacteria override diatoms and become the most important primary producers in this lake at a higher trophic level, because chlorophytes and cyanobacteria are better competitors for low light and turbid status than diatoms under eutrophic, well-mixed water conditions (Tilman et al., 1986) and thus influence the response of diatoms. However, the increased production of chlorophytes and cyanobacteria does not restrain diatom growth through competition and/or allelopathy and diatoms can co-exist with other types of algae, because diatom concentration and diatom specific pigments (i.e. fucoxanthin and diatoxanthin) show a clear increasing trend (Figs. 3 & 5). Thus, the combined diatom and pigment analyses in this study provide clear evidence for recent human-induced accelerated lake eutrophication and a major ecological shift, and improves understanding of the predisturbance condition for current ecological restoration in this lake.
5 CONCLUSIONLake Dojran, a shallow and eutrophic lake across the Republic of North Macedonia and Greece, is a key site for understanding the environmental change in the Mediterranean region. This study conducted the diatom and pigment analyses of the Lake Dojran sediment sequence that spans the past ca. 450 years, and their preservation qualities were evaluated prior to palaeolimnological interpretation. For diatoms, good preservation is inferred from the co-existence of robust, highly-silicified taxa and small taxa throughout the sequence and also from the correspondence of the consistent diatom composition with the current limnological conditions. For pigment preservation, our comparison of wet sediment samples, which were in dark and cold storage until the analysis, with the corresponding dry sediment samples, which were lyophilized immediately after coring, reveals that sediment restoration conditions exert large influence on the accuracy of pigment analysis. In the palaeolimnological interpretation, the chlorophyll and xanthophyll pigment concentration and diatom concentration data provide clear evidence for accelerated lake eutrophication and ecological shift linked to intensified anthropogenic water reduction and agricultural nutrient diffuse in the late 18th to early 19th century; whereas, diatom assemblage composition does not show evidence, probably because widely-tolerant small fragilaroid species dominate the diatom composition and/or lightcompetitive chlorophytes and cyanobacteria override diatoms under eutrophic and turbid conditions. This study improves our understanding of recent humaninduced environmental change and current ecological restoration target in this lake.
6 DATA AVAILABILITY STATEMENTThe data that support the findings of this study are available in the Supplementary Information File.
7 ACKNOWLEDGMENTWe would especially like to thank Suzanne McGowan (University of Nottingham, UK) for providing help for pigment analysis. Many thanks are also due to Bernd Wagner (University of Cologne, Germany) and Alexander Francke (University of Wollongong, Australia) for providing samples for analysis. Two anonymous reviewers are also thanked for their detailed and constructive comments, suggestions and improvements on the manuscript.
Electronic supplementary materialSupplementary material (Supplementary Information File) is available in the online version of this article at https://doi.org/10.1007/s00343-020-9320-5.
Athanasiadis N, Tonkov S, Atanassova J, Bozilova E. 2000. Palynological study of Holocene sediments from Lake Doirani in northern Greece. Journal of Paleolimnology, 24(3): 331-342.
DOI:10.1023/A:1008161819212 |
Battarbee R W, Jones V J, Flower R J, Cameron N G, Bennion H, Carvalho L, Juggins S. 2001. Diatoms. In: Smol J P, Birks H J B, Last W. M eds. Tracking Environmental Change using Lake Sediments. Volume 3: Terrestrial, Algal, and Siliceous Indicators. Kluwer Academic Publishers, Dordrecht. p.155-202.
|
Bennion H, Battarbee R W, Sayer C D, Simpson G L, Davidson T. A. 2011. Defining reference conditions and restoration targets for lake ecosystems using palaeolimnology: a synthesis. Journal of Paleolimnology, 45(4): 533-544.
DOI:10.1007/s10933-010-9419-3 |
Bennion H, Battarbee R. 2007. The European Union Water Framework Directive: opportunities for palaeolimnology. Journal of Paleolimnology, 38(2): 285-295.
DOI:10.1007/s10933-007-9108-z |
Bennion H, Carvalho L, Sayer C D, Simpson G L, Wischnewski J. 2012. Identifying from recent sediment records the effects of nutrients and climate on diatom dynamics in Loch Leven. Freshwater Biology, 57(10): 2015-2029.
DOI:10.1111/j.1365-2427.2011.02651.x |
Bennion H, Sayer C D, Tibby J, Carrick H J. 2010. Diatoms as indicators of environmental change in shallow lakes. In: Smol J P, Stoermer E. F eds. The Diatoms: Applications for the Environmental and Earth Sciences. 2nd edn. Cambridge University Press, Cambridge. p.152-173.
|
Bennion H, Simpson G L, Goldsmith B. J. 2015. Assessing degradation and recovery pathways in lakes impacted by eutrophication using the sediment record. Frontiers in Ecology and Evolution, 3: 94.
DOI:10.3389/fevo.2015.00094 |
Bronk Ramsey C. 2009. Bayesian analysis of radiocarbon dates. Radiocarbon, 51(1): 337-360.
DOI:10.1017/S0033822200033865 |
Conley D J, Paerl H W, Howarth R W, Boesch D F, Seitzinger S P, Havens K E, Lancelot C, Likens G E. 2009. Controlling eutrophication: nitrogen and phosphorus. Science, 323(5917): 1014-1015.
DOI:10.1126/science.1167755 |
Cruces F, Rivera P, Urrutia R. 2010. Observations and comments on the diato. Stephanodiscus minutulus (Kützing) Cleve & Möller (Bacillariophyceae) found for the first time in Chile from bottom sediments collected in Lake Laja. Gayana Botánica, 67(1): 12-18.
DOI:10.4067/S0717-66432010000100002 |
Egeland E S, Garrido J L, Clementson L, Andresen K, Thomas C S, Zapata M, Airs R, Llewellyn C A, Newman G L, Rodríguez F, Roy S. 2011. Data sheets aiding identification of phytoplankton carotenoids and chlorophylls. In: Roy S, Llewellyn C A, Egeland E S, Johnsen G. eds. Phytoplankton Pigments: Characterization, Chemotaxonomy and Applications in Oceanography. Cambridge University Press, Cambridge. p.675-822.
|
Elser J, Bennett E. 2011. A broken biogeochemical cycle. Nature, 478(7367): 29-31.
DOI:10.1038/478029a |
Fourtanier E, Kociolek J. P. 2011. Catalogue of Diatom Names, On-line Version, updated 19 Sep. 2011. https://researcharchive.calacademy.org/research/diatoms/names/index.asp.
|
Francke A, Wagner B, Leng M J, Rethemeyer J. 2013. A Late Glacial to Holocene record of environmental change from Lake Dojran (Macedonia, Greece). Climate of the Past, 9(1): 481-498.
DOI:10.5194/cp-9-481-2013 |
Griffiths H I, Reed J M, Leng M J, Ryan S, Petkovski S. 2002. The recent palaeoecology and conservation status of Balkan Lake Dojran. Biological Conservation, 104(1): 35-49.
DOI:10.1016/S0006-3207(01)00152-5 |
Guilizzoni P, Lami A. 2002. Paleolimnology: use of algal pigments as indicators. In: Bitton G ed. Encyclopedia of Environmental Microbiology. John Wiley & Sons, New York. p.2306-2317.
|
Guiry M D, Guiry G. M. 2019. AlgaeBase. World-Wide Electronic Publication, National University of Ireland.
|
Hall R I, Smol J. P. 2010. Diatoms as indicators of lake eutrophication. In: Smol J P, Stoermer E. F eds. The Diatoms: Applications for the Environmental and Earth Sciences. 2nd edn. Cambridge University Press, Cambridge. p.122-151.
|
Hobbs W O, Fritz S C, Stone J R, Donovan J J, Grimm E C, Almendinger J E. 2011. Environmental history of a closed-basin lake in the US Great Plains: diatom response to variations in groundwater flow regimes over the last 8500 cal. yr BP. Holocene, 21(8): 1203-1216.
DOI:10.1177/0959683611405242 |
Houk V, Klee R, Tanaka H. 2010. Atlas of freshwater centric diatoms with a brief key and descriptions. Part Ⅲ. Stephanodiscaceae A. Cyclotella, Tertiarius, Discostella. Fottea, 10(Supplement): 1-498.
|
Houk V, Klee R, Tanaka H. 2014. Atlas of freshwater centric diatoms with a brief key and descriptions. Part IV. Stephanodiscaceae B. Stephanodiscus, Cyclostephanos, Pliocaenicus, Hemistephanos, Stephanocostis, Mesodictyon & Spicaticribra. Fottea, 14(Supplement): 1-530.
|
IP CC. 2013. Climate Change 2013. The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge and New York. 1535p.
|
Jeffrey S W, Mantoura R. F C, Bjørnland T. 1997. Data for the identification of 47 key phytoplankton pigments. In: Jeffrey S W, Mantoura R. F C, Wright S. W eds. Phytoplankton Pigments in Oceanography: Guidelines to Modern Methods. UNESCO Publishing, Paris. p.447-560.
|
Krammer K, Lange-Bertalot H. 1986. Süsswasserflora von Mitteleuropa. Band 2/1: Bacillariophyceae. Teil 1: Naviculaceae. Gustav Fischer Verlag, Stuttgart. 876p.
|
Krammer K, Lange-Bertalot H. 1988. Süsswasserflora von Mitteleuropa. Band 2/2: Bacillariophyceae. Teil 2: Epithemiaceae, Bacillariaceae, Surirellaceae. Gustav Fischer Verlag, Stuttgart. 596p.
|
Krammer K, Lange-Bertalot H. 1991a. Süsswasserflora von Mitteleuropa. Band 2/3: Bacillariophyceae. Teil 3: Centrales, Fragilariaceae, Eunotiaceae. Gustav Fischer Verlag, Stuttgart. 576p.
|
Krammer K, Lange-Bertalot H. 1991b. Süsswasserflora von Mitteleuropa. Band 2/4: Bacillariophyceae. Teil 4: Achnanthaceae. Gustav Fischer Verlag, Stuttgart. 437p.
|
Krammer K. 2002. Diatoms of Europe: Diatoms of the European Inland Waters and Comparable Habitats. Volume 3. Cymbella. Gantner Verlag, Ruggell. 584p.
|
Lami A N, Guilizzoni P, Marchetto A. 2000. High resolution analysis of fossil pigments, carbon, nitrogen and sulphur in the sediment of eight European Alpine lakes: the MOLAR project. Journal of Limnology, 59(S1): 15-28.
DOI:10.4081/jlimnol.2000.s1.15 |
Lange-Bertalot H. 2001. Diatoms of Europe: Diatoms of the European Inland Waters and Comparable Habitats. Volume 2. Navicula Sensu Stricto, 10 Genera Separated fro. Navicula Sensu Lato. Frustulia. Gantner Verlag, Ruggell. 526p.
|
Leavitt P R, Hodgson D. A. 2001. Sedimentary pigments. In: Smol J P, Birks H J B, Last W. M eds. Tracking Environmental Change using Lake Sediments. Volume 3: Terrestrial, Algal, and Siliceous Indicators. Kluwer Academic Publishers, Dordrecht. p.295-325.
|
Lešoski J, Zdraveski N, Kristić S. 2010. Preliminary results on cyanobacterial survey on Dojran Lake-the beginning of revealing of the ultimate truth about the lake's water quality. In: Conference on Balkan Water Observation and Information System for Decision Support (BALWOIS). Ohrid.
|
Levkov Z, Krstic S, Metzeltin D, Nakov T. 2007. Iconographia Diatomologica. Volume 16: Diatoms of Lakes Prespa and Ohrid. Gantner Verlag, Ruggell. 613p.
|
Levkov Z, Stojanovski P. 2000-2001. Changes in Doiran Lake's diatom flora, a 13 years study. Godisen zbornik Biologija, 53-54: 22-38.
|
Levkov Z. 2009. Diatoms of Europe: Diatoms of the European Inland Waters and Comparable Habitats. Volume 5. Amphora Sensu Lato. Gantner Verlag, Ruggell. 842p.
|
Lionello P. 2012. The Climate of the Mediterranean Region: From the Past to the Future. Elsevier, Amsterdam. 592p.
|
Lokoska L, Jordanoski M, Veljanoska-Sarafiloska E, Tasevska O. 2006. Water quality of Lake Dojran from biological and physical-chemical aspects. In: Conference on Balkan Water Observation and Information System for Decision Support (BALWOIS). Ohrid.
|
Masi A, Francke A, Pepe C, Thienemann M, Wagner B, Sadori L. 2018. Vegetation history and paleoclimate at Lake Dojran (FYROM/Greece) during the Late Glacial and Holocene. Climate of the Past, 14(3): 351-367.
DOI:10.5194/cp-14-351-2018 |
McGowan S, Barker P, Haworth E Y, Leavitt P R, Maberly S C, Pates J. 2012. Humans and climate as drivers of algal community change in Windermere since 1850. Freshwater Biology, 57(2): 260-277.
DOI:10.1111/j.1365-2427.2011.02689.x |
McGowan S. 2013. Palaeolimnology: pigment studies. In: Elias S A, Mock C. J eds. Encyclopedia of Quaternary Science. 2nd edn. Elsevier, Amsterdam. p. 326-338.
|
Paerl H W, Scott J T, McCarthy M J, Newell S E, Gardner W S, Havens K E, Hoffman D K, Wilhelm S W, Wurtsbaugh W A. 2016. It takes two to tango: when and where dual nutrient (N & P) reductions are needed to protect lakes and downstream ecosystems. Environmental Science & Technology, 50(20): 10805-10813.
DOI:10.1021/acs.est.6b02575 |
Popovska C, Bonacci O. 2008. Ecohydrology of Dojran Lake. In: Hlavinek P, Bonacci O, Marsalek J, Mahrikova I, eds. Dangerous Pollutants (Xenobiotics) in Urban Water Cycle. Springer, Dordrecht. p.151-160.
|
Popovska C, Gesovska V, Ivanoski D. 2005. Ecological and hydrological state of Dojran Lake. Vodoprivreda, 37(216-218): 175-180.
|
Reimer P J, Bard E, Bayliss A, Beck J W, Blackwell P G, Ramsey C B, Buck C E, Cheng H, Edwards R L, Friedrich M, Grootes P M, Guilderson T P, Haflidason H, Hajdas I, Hatté C, Heaton T J, Hoffmann D L, Hogg A G, Hughen K A, Kaiser K F, Kromer B, Manning S W, Niu M, Reimer R W, Richards D A, Scott E M, Southon J R, StaffR A, Turney C S M, van der Plicht J. 2013. IntCal13 and Marine13 radiocarbon age calibration curves 0-50, 000 years cal BP. Radiocarbon, 55(4): 1869-1887.
DOI:10.2458/azu_js_rc.55.16947 |
Reuss N, Conley D. J. 2005. Effects of sediment storage conditions on pigment analyses. Limnology and Oceanography: Methods, 3(10): 477-487.
DOI:10.4319/lom.2005.3.477 |
Rimet F, Druart J C, Anneville O. 2009. Exploring the dynamics of plankton diatom communities in Lake Geneva using emergent self-organizing maps (1974-2007). Ecological Informatics, 4(2): 99-110.
DOI:10.1016/j.ecoinf.2009.01.006 |
Roberts N, Reed J. M. 2009. Lakes, wetlands, and Holocene environmental change. In: Woodward J C ed. The Physical Geography of the Mediterranean. Oxford University Press, Oxford. p.255-286.
|
Rockström J, Steffen W, Noone K, Persson Å, Chapin F S, Lambin E F, Lenton T M, Scheffer M, Folke C, Schellnhuber H J, Nykvist B, de Wit C A, Hughes T, van der Leeuw S, Rodhe H, Sörlin S, Snyder P K, Costanza R, Svedin U, Falkenmark M, Karlberg L, Corell R W, Fabry V J, Hansen J, Walker B, Liverman D, Richardson K, Crutzen P, Foley J. A. 2009. A safe operating space for humanity. Nature, 461(7263): 472-475.
DOI:10.1038/461472a |
Ryves D B, Juggins S, Fritz S C, Battarbee R. W. 2001. Experimental diatom dissolution and the quantification of microfossil preservation in sediments. Palaeogeography, Palaeoclimatology, Palaeoecology, 172(1-2): 99-113.
DOI:10.1016/S0031-0182(01)00273-5 |
Sayer C D. 2001. Problems with the application of diatomtotal phosphorus transfer functions: examples from a shallow English lake. Freshwater Biology, 46(6): 743-757.
DOI:10.1046/j.1365-2427.2001.00714.x |
Scheffler W, Morabito G. 2003. Topical observations on centric diatoms (Bacillariophyceae, Centrales) of Lake Como (N. Italy). Journal of Limnology, 62(1): 47-60.
DOI:10.4081/jlimnol.2003.47 |
Smol J P, Stoermer E. F. 2010. The Diatoms: Applications for the Environmental and Earth Sciences. 2nd edn. Cambridge University Press, Cambridge. 686p.
|
Smol J P. 2019. Under the radar: long-term perspectives on ecological changes in lakes. Proceedings of the Royal Society B: Biological Sciences, 286: 20190834.
DOI:10.1098/rspb.2019.0834 |
Sotiria K, Petkovski S. 2004. Lake Dojran-An overview of the current situation. Greek Biotope/Wetland Centre (EKBY) and Society for the Investigation and Conservation of Biodiversity and the Sustainable Development of Natural Ecosystems (BIOECO), Thermi. 117p.
|
Steffen W, Richardson K, Rockström J, Cornell S E, Fetzer I, Bennett E M, Biggs R, Carpenter S R, de Vries W, de Wit C A, Folke C, Gerten D, Heinke J, Mace G M, Persson L M, Ramanathan V, Reyers B, Sorlin S. 2015. Planetary boundaries: guiding human development on a changing planet. Science, 347(6223): 1259855.
DOI:10.1126/science.1259855 |
Stojov V. 2012. Hydrological state of Dojran Lake related to tectonic, climatic and human impacts. In: Conference on Balkan Water Observation and Information System for Decision Support (BALWOIS). Ohrid.
|
Sutton M A, Oenema O, Erisman J W, Leip A, van Grinsven Grinsven H, Winiwarter W. 2011. Too much of a good thing. Nature, 472(7342): 159-161.
DOI:10.1038/472159a |
Tasevska O, Kostoski G, Guseska D. 2010. Rotifers based assessment of the Lake Dojran water quality. In: Conference on Balkan Water Observation and Information System for Decision Support (BALWOIS). Ohrid.
|
Temponeras M, Kristiansen J, Moustaka-Gouni M. 2000. Seasonal variation in phytoplankton composition and physical-chemical features of the shallow Lake Doïrani, Macedonia, Greece. Hydrobiologia, 424(1): 109-122.
DOI:10.1023/A:1003909229980 |
Tilman D, Kiesling R, Sterner R, Kilham S S, Johnson F. A. 1986. Green, bluegreen and d diatom algae: taxonomic differences in competitive ability for phosphorus, silicon and nitrogen. Archiv für Hydrobiologie, 106(4): 473-485.
|
Verdonschot P F M, Spears B M, Feld C K, Brucet S, Keizer-Vlek H, Borja A, Elliott M, Kernan M, Johnson R K. 2013. A comparative review of recovery processes in rivers, lakes, estuarine and coastal waters. Hydrobiologia, 704(1): 453-474.
DOI:10.1007/s10750-012-1294-7 |
Veuger B, van Oevelen. 2011. Long-term pigment dynamics and diatom survival in dark sediment. Limnology and Oceanography, 56(3): 1065-1074.
DOI:10.4319/lo.2011.56.3.1065 |
Wan L L, Chen X Y, Deng Q H, Yang L, Li X W, Zhang J Y, Song C L, Zhou Y Y, Cao X. Y. 2019. Phosphorus strategy in bloom-forming cyanobacteria. Dolichospermum and Microcystis) and its role in their succession. Harmful Algae, 84: 46-55.
DOI:10.1016/j.hal.2019.02.007 |
Wang S Y, Xiao J, Wan L L, Zhou Z J, Wang Z C, Song C L, Zhou Y Y, Cao X. Y. 2018. Mutual dependence of nitrogen and phosphorus as key nutrient elements: one facilitate. Dolichospermum flos-aquae to overcome the limitations of the other. Environmental Science & Technology, 52(10): 5653-5661.
DOI:10.1021/acs.est.7b04992 |
Zhang X S, Reed J M, Wagner B, Francke A, Levkov Z. 2014. Lateglacial and Holocene climate and environmental change in the northeastern Mediterranean region: diatom evidence from Lake Dojran (Republic of Macedonia/Greece). Quaternary Science Reviews, 103: 51-66.
DOI:10.1016/j.quascirev.2014.09.004 |
 2020, Vol. 38
2020, Vol. 38


