Institute of Oceanology, Chinese Academy of Sciences
Article Information
- FANG Jinghui, FANG Jianguang, CHEN Qionglin, MAO Yuze, JIANG Zengjie, DU Meirong, GAO Yaping, LIN Fan
- Assessing the effects of oyster/kelp weight ratio on water column properties: an experimental IMTA study at Sanggou Bay, China
- Journal of Oceanology and Limnology, 38(6): 1914-1924
- http://dx.doi.org/10.1007/s00343-019-9109-6
Article History
- Received Apr. 17, 2019
- accepted in principle Jul. 8, 2019
- accepted for publication Oct. 25, 2020
2 Key Laboratory of Marine Biotechnology of Zhejiang Province, School of Marine Sciences, Ningbo University, Ningbo 315211, China
Aquaculture is a fast-growing food-producing sector in the world, which is crucial to meet the need of growing human population on the Earth. The State of the World Fisheries and Aquaculture (SOFIA) states that "in terms of global production volume, that of farmed fish and aquatic plants combined surpassed that of capture fisheries in 2013. In terms of food supply, aquaculture provided more fish than capture fisheries in 2014" (FAO, 2016). Driven by the growing demand for aquaculture, many environmental problems appeared, which hampered the sustainable development of aquaculture, particularly for the species that need feed input. Only 17% of the nitrogen in fish feed can be retained in finfish, while the rest 83% are wasted in the environment (Wen et al., 2007). Furthermore, Wu (1995) found that 1%–15% of formulated fish feed and 40% of fresh feed are wasted. Half of sedimentary organic carbon is originated from aquaculture fish feed (Gillibrand et al., 1996). The nitrogen input from the feed in aquaculture pond or cage could amounted to 54.7%–95% of the total nitrogen input in the system (Briggs and Fvnge-Smith, 1994; Chang et al., 2006; Zhang, 2012). Therefore, designing a system to reduce the negative effects in aquaculture production is an urgent task.
To solve the pollution problem, we developed a novel concept: the integrated multi-trophic aquaculture (IMTA). An IMTA system in essence is a multi-species co-culture at different trophic levels within a certain area to maintain optimal production while reduce the negative effects of feed loss (Chopin et al., 2010; Fang et al., 2014, 2016). In the system, waste nutrients produced from higher trophic species can be re-used by lower trophic species. In the design, to seek and combine different species farmed is complicated. There are several successful practices, e.g., fish-seaweed (Stenton-Dozey, 2007; Abreu et al., 2009), finfish-shellfish (Mazzola and Sarà, 2001; Langan, 2004), abalone-kelp, finfish-bivalve-kelp, abalone-sea-cucumber-clam-seaweed (Fang et al., 2016), and shellfish-seaweed (Chopin et al., 2008), etc. The combination of shellfish-seaweed is a basic IMTA mode. However, there is no additional biogenic element input through the management in a filterfeeding shellfish-seaweed mode. Moreover, it would be an important mode if is beneficial to the environment (Tang et al., 2011). Seaweed can absorb dissolved biogenic elements, such as carbon dioxide (CO2), dissolved inorganic carbon (DIC), ammonia (NH3), nitrite (NO2-), nitrate (NO3-) and phosphorous (P), and at the same time, produce oxygen, while the filter-feeding shellfish can utilize organic particles and produces ammonia nitrogen and CO2 for seaweed. Meanwhile, shellfish can absorb carbon for building shells in the form of CaCO3. Thus, seaweed-shellfish co-culturing could benefit each other in the IMTA system. The question is: what would be the optimal ratio of the two species in eco-friendly practice.
Sanggou Bay (37°01′N–37°09′N, 122°24′E–122°35′E), located in Shandong Province, China, is a typical IMTA area that houses more than 30 important aquaculture species, including kelp (Saccharina japonica), oyster (Crassostrea gigas), mussel (Mytilus edulis), scallop (Chlamys farreri), abalone (Haliotis discus hannai), and sea cucumber (Apostichopus japonicus) (Zhang et al., 2007). Some of them have been successfully co-cultured in IMTA mode. Shellfish-seaweed collocation is the base of the IMTA modes (Tang et al., 2013; Fang et al., 2016). In 2015, 65 000 ton of oyster (C. gigas), one of the mostly cultured shellfish, was produced in Sanggou Bay (data from local government), and kelp (S. japonica) is the main cultured seaweed in Sanggou Bay, and 84 500 ton was produced in 2015 (Ning et al., 2016). In order to determine the optimal ratio of oyster to kelp co-culture in Sanggou Bay at the beginning of winter (the kelp culturing period), water quality and carbonate system were investigated within the frame of different oyster-kelp modes.
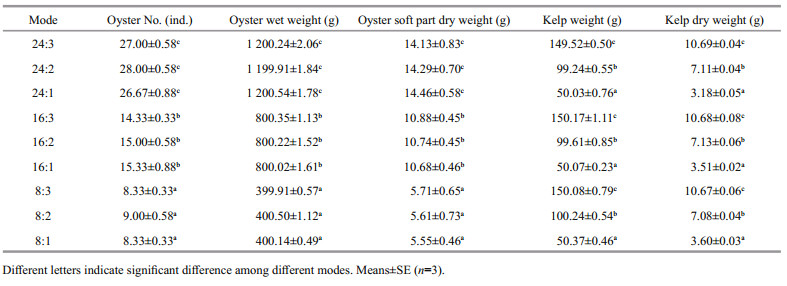
2 MATERIAL AND METHOD 2.1 Experimental design and managementThe experiment was conducted in Sanggou Bay, China, in December 2017 (Fig. 1). Kelp S. japonica (whole plant wet weight: 1.65±0.76 g, mean±SD, n=30) and oyster C. gigas (wet weight: 36.26±3.89 g, mean±SD, n=30) were collected locally. Kelps were tied on ropes and oysters were cleaned and put into a net bag. Both organisms were hung onto a transparent polyethylene plastic bag (1.0 m3) that was securely sealed with a cap, after expelling all air bubbles. Each bag had a special cap that can be opened and closed easily. Nine collocation modes in oyster꞉kelp weight ratio were tested showing as 24꞉3, 24꞉2, 24꞉1, 16꞉3, 16꞉2, 16꞉1, 8꞉3, 8꞉2, and 8꞉1 (Table 1). The number of oysters is shown in Table 2. Each treatment included 30, 60, and 90 kelps and 3 replications in each treatment.
|
| Fig.1 The study site |

|
The bags were randomly tied onto the floating frame (10 m2) in open sea and filled with natural seawater. The bags were placed 10–20 cm above the surface. During the experiment, samples were collected at 17:00 on Day 1 (D1), and 6:00 and 17:00 on Days 2 and 3. The time was adjusted according to local sunrise at 6:00 and sunset at 17:00. Water temperature, salinity, pH, and dissolved oxygen (DO) were measured by YSI Professional Plus (Pro Plus, USA) and pH meter (Orion 9107 BNMD, USA). Measurement work took 1.5 h each time. One liter of water sample was collected from each bag stirred in each sampling period and particles in the water samples were collected with cellulose acetate filters (0.45 μm, Whatman) and preserved at -20℃. An amount of 150-mL filtered seawater was mixed with 0.02% HgCl2 saturated solution and stored and sealed in 150-mL borosilicate bottles at 4℃ for total alkalinity (TA) analysis (Dickson and Goyet, 1994).
2.2 Sample analysisFluorescence analysis was used to determine chlorophyll-a (chl-a) content. Total alkalinity (TA) was measured using an automatic titrator (848 Titrino plus, Switzerland). DIC, [CO2], [CO32-] and [HCO3-] concentration in seawater carbonate system were calculated using CO2SYS_V2, 1 software based on the TA, pH, salinity and temperature. All oyster and kelp samples were removed from the bags and cleaned with sterile gauze to eliminate surface seawater, and then dried separately in an oven at 70℃ for 48 h. Finally, the dry weight of the oyster's shell and tissue part, following the field experiment, were determined.
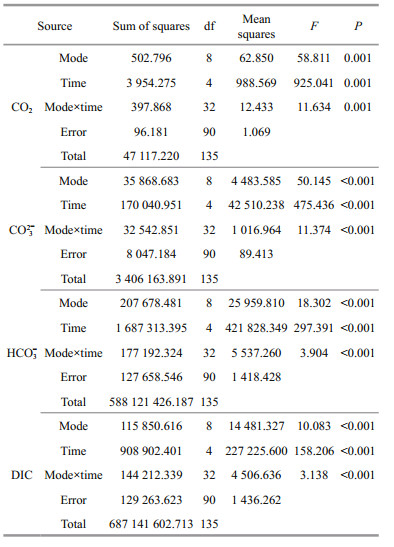
2.3 Data analysisData were analyzed using SPSS 13.0 for Windows (SPSS, USA). Prior to statistical analysis, all data were checked for homogeneity and normality of variance assumptions by (i) visually examining standardized residuals versus predicted values plots and Q-Q plots of residuals, (ii) Shapiro-Wilk tests, and (iii) Levene tests. Data were log-transformed if a normal distribution assumptions were not met. Oyster and kelp in each mode were considered together as a combined system (mode). Interaction between modes and experiment time on DO, pH, chl a, and parameters of the carbonate system were tested using two-way repeated ANOVA measurements. Statistically significant influences, proven by ANOVA, were followed by a Tukey test to determine the withintreatment effects. The difference in the organism's initial wet weight and dry weight and biomass after experiment among treatments was tested using oneway ANOVA. All differences were considered statistically significant at P < 0.05.
3 RESULTDuring the experiment, water temperature ranged 9.0–10.2℃, salinity 31.30–31.57, and no dead animals were found. The biomass of the organisms was allocated as the experiment design showed in Table 2.
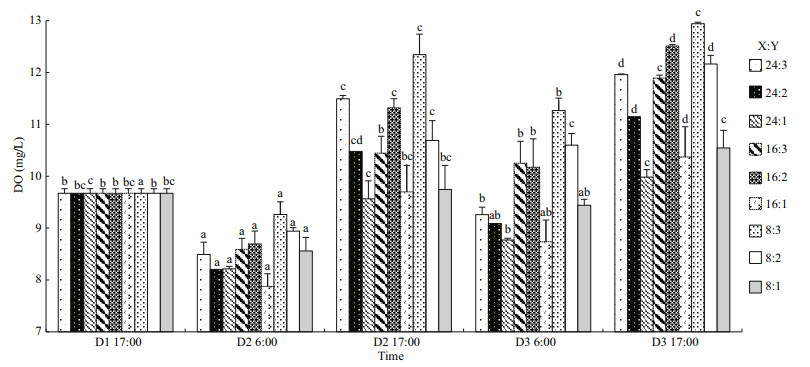
3.1 Dissolved oxygenTwo-way ANOVA showed that DO was significantly affected by oyster-kelp modes and sampling time, and that interaction between both factors was significant (P < 0.05, Table 3). DO was higher in the modes with higher kelp ratio (P < 0.05, Fig. 2). At the end of the experiment, DO was lowest in the 24꞉1 mode and highest in the 8꞉3 mode (P < 0.05).
|
|
| Fig.2 The dissolved oxygen of oyster-kelp modes at different time D1, D2, and D3 (D=Day); X:Y=oyster and kelp ratios in weight (Table 1). The different letters indicate a significant difference of DO in the same mode at different detecting time. Means±SE (n=3). |
The pH values in this experiment are often in parallel to those of the DO results (Fig. 3).

|
| Fig.3 The pH of oyster-kelp modes at different time D1, D2, and D3 (D=Day); X:Y=oyster and kelp weight ratios (Table 1). The different letters indicate a significant difference of pH within mode at different detecting time. Means±SE (n=3). |
Results of two-way ANOVA show that pH was significantly different among the oyster-kelp modes and sampling time, and interaction between both factors was significant (P < 0.05, Table 3). pH was higher in the modes of more kelp (P < 0.05). At the end of the experiment, pH was the lowest in the 16꞉1 mode and highest in the 8꞉3 mode (P < 0.05).
pH fluctuated significantly during day and night, but the means were lower in the morning than those in the afternoon on the same day (P < 0.05, one-way ANOVA). At the end of the experiment, pH was significantly higher in all mode than the initial value (P < 0.05).
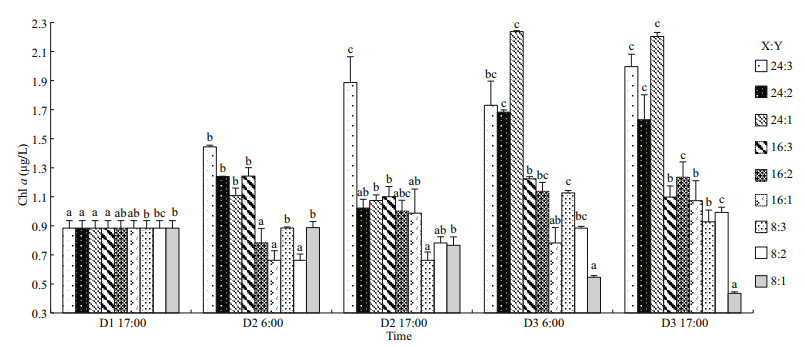
3.3 Chl aAs two-way ANOVA statistics show, chl-a concentration was significantly different among the oyster-kelp modes and sampling time. Interaction between both factors was significant (P < 0.05, Table 3). Chl-a concentration demonstrated significantly higher values in the modes in higher ratio of oyster꞉kelp (P < 0.05). The chl-a concentration was continuously increasing over the time (P < 0.05).
By the end of the experiment, the modes with "24" oysters contained highest chl-a concentrations (P < 0.05, Fig. 4). There was a significant increase in 24꞉1 and 24꞉2 modes on 6:00 am of D3 (P < 0.05). Chl-a concentrations increased in all modes compared with those at D1 17:00 excepting for the 8꞉1 mode at the end of the experiment. However, no significant difference was found between D1 17:00 and D3 17:00 in the 8꞉2, 8꞉3 or 16꞉1 modes (P > 0.05).
|
| Fig.4 The chl a of oyster-kelp modes at different time D1, D2, and D3 (D=Day); X:Y=oyster and kelp weight ratios (Table 1). The different letters indicate a significant difference of chl a within mode at different detecting time. Means±SE (n=3). |
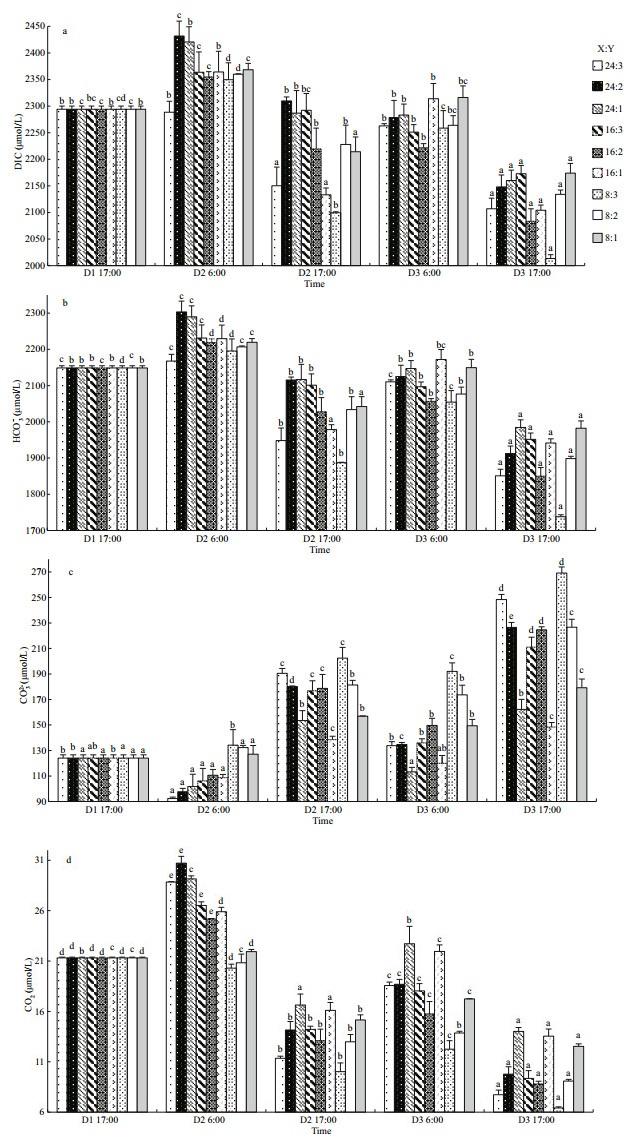
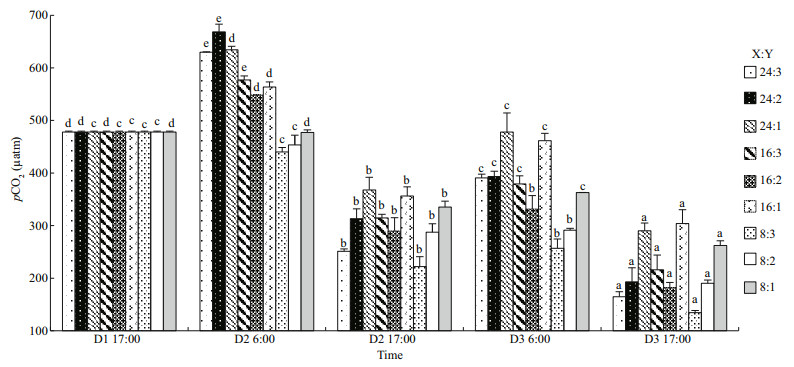
The carbonate system and pCO2 were significantly related to the oyster-kelp modes and sampling time and their interactions were also significant (P < 0.05, two-way ANOVA, Tables 3 & 4). DIC, HCO3-, CO2 concentrations and pCO2 were the lowest on D3 in the 8꞉3 mode at 17:00 (P < 0.05, Figs. 5a, b, d, & 6). In contrast, the CO32- concentration was highest on D3 in the 8꞉3 mode at 17:00 (P < 0.05, Fig. 5c). Thus, throughout the experiment, the CO32- concentration was increasing, while the DIC, HCO3-, CO2 concentrations, and pCO2 were decreasing.
|
|
| Fig.5 The variation of carbonate system in different oyster-kelp modes a. DIC; b. HCO3-; c. CO32-; d. CO2. D1, D2, and D3 (D=Day); X:Y=oyster and kelp weight ratios (Table 1). The different letters indicate significant difference of DIC, HCO3-, CO32-, and CO2 within mode at different detecting time, respectively. Means±SE (n=3). |
|
| Fig.6 The fluctuations of pCO2 in oyster-kelp modes at different time D1, D2, and D3 (D=Day); X:Y=oyster and kelp weight ratios (Table 1). The different letters indicate a significant difference of pCO2 within mode at different detecting time. Means±SE (n=3). |
Variation of DIC is very similar to that of HCO3- concentration in the same mode (Fig. 5a & b). The DIC was the lowest (2 013.53 μmol/L) on D3 in the 8꞉3 mode at 17:00 (P < 0.05, one-way ANOVA), and the highest (2 431.65 μmol/L) on D2 in the 24꞉2 mode at 6:00. Similarly, the HCO3- concentration reached its lowest (1 738.10 μmol/L) (P < 0.05) and highest (2 303.22 μmol/L) point at the same day, mode, and time as DIC. By the end of the experiment, DIC and HCO3- had decreased significantly in all the oysterkelp modes (P < 0.05).
In contrast, by the end of the experiment, the CO32- concentration had significantly increased in all the oyster-kelp modes (P < 0.05, one-way ANOVA, Fig. 5c). At the end of the experiment, the highest CO32- concentration was 269.13 μmol/L in the 8꞉3 mode (P < 0.05), and the lowest was 148.35 μmol/L in 16꞉1 mode. CO32- concentration was lower in the morning than those in the afternoon on the same day (P < 0.05). More kelp ratio induced arising CO32- concentration in the modes with the same amount of oyster.
The fluctuations of CO2 concentration and pCO2 were very similar. Both of them fluctuated significantly during day and night, but means were lower in the afternoon than those in the morning on the same day (P < 0.05, one-way ANOVA, Figs. 5d & 6). The higher CO2 concentration and pCO2 were in the modes with higher oyster ratio. Both of them decreased significantly at the end of the experiment in all oysterkelp modes (P < 0.05). pCO2 dropped as low as 134.31 μatm in the 8꞉3 mode.
4 DISCUSSIONIMTA initiated a new direction for sustainable aquaculture (Troell et al., 2009) in which different trophic species could benefit to each other. Fed aquaculture species (e.g. finfish) produce high quality protein, bringing excessive nutrients into the aquatic ecosystem through feeding. Wu (1995) reviewed the impact of marine fish farming on the marine environment and determined that 85% P, 80%–88% C, and 52%–95% N could be lost in the environment, due to uneaten feed, fish excretion, and respiration, and feces production. The suspended particles can be consumed by filter feeders (e.g., shellfish), which are called organic extractive species in the IMTA system. Jiang et al., 2013, determined that 34.86% of fish feces and fish feed from the sea bass (Lateolabrax japonicus) cage were consumed by Pacific oysters (C. gigas). Furthermore, 23% C, 21% N and 53% P of feed input accumulated in the sediment underneath the fish farm (Wu, 1995), most of which was consumed by the deposit-feeders, such as sea cucumbers and polychaete. Sea cucumber can consume 441–3 500 g/(ind.∙a) of sea mud (Inui et al., 1991). C and N contents in the sediment beneath a combined Japanese flounder (Paralichthys olivaceus) and polychaete (Perinereis albuhitensis) system were reduced by 30.68% and 51.89%, respectively, compared with control treatment after 40 days (Fang et al., 2014). However, dissolved inorganic nutrients from aquaculture animals (e.g. fed aquaculture species and organic extractive species) could be absorbed by inorganic extractive aquaculture species (e.g. seaweeds), which is the necessary and vital part of the IMTA system with respect to keeping the environment healthy. In this study, there is no fed aquaculture species. The shellfish-seaweed IMTA mode is removing bio-elements from the ocean ecosystem without any element input. The dissolved inorganic carbon (DIC), HCO3-, CO2 concentrations and pCO2 were reduced by the end of the experiment in all treatments. It is definitely a carbon sink IMTA system and should be considered one of the most ecofriendly IMTA modes. Eutrophication in coastal area is a serious problem all around the world. Fortunately, waste nutrients in the aquatic environment can be absorbed by seaweeds to recover water quality, a process that has been well studied in many previous works (Ahn et al., 1998; Phillips and Hurd, 2003; Pedersen et al., 2004; Lartigue and Sherman, 2005; Mao et al., 2009). Mao et al. (2006) reported that NH4+ and PO43- produced by scallop could be absorbed by Gracilaria lemaneiformis. The maximum reduction efficiency of NH4+ and PO43- were 83.17% and 70.14%, respectively. Therefore, it is well known that seaweeds in the IMTA system can remove dissolved nutrients produced from cultured animals and weaken eutrophication in coastal areas. The more seaweed included in the system, the more dissolved nutrients can be absorbed. The same trend was found from our study, even if the oysters were to be fed, the excretion nutrients of the oyster would be absorbed by the kelp in the experimental system. Thus, additional work is needed to explore the detailed fluctuations of different nutrients in the shellfish-seaweed IMTA system.
Furthermore, IMTA system may affects the seawater's carbonate system, which plays an important role in the coastal carbon cycle (Tang et al., 2011). It has been reported that the shellfish culture, without macroalgae, could be an appreciable net carbon source (Han et al., 2013). At the end of a 36 h-experiment, pH, and DO significantly reduced and CO2 concentration significantly increased in shellfish in treatment with no seaweed. However, shellfish integrated with seaweed could generate a carbon sink system, which would aid in offsetting the negative environmental effects facilitated by shellfish culture (Han et al., 2013). Yet, finding the optimal proportions of different trophic species for particular IMTA systems is difficult. Jiang et al. (2010) published good suggestions for optimal co-cultivation proportion of caged fish and Laminaria or Gracilaria based on the nitrogen balance in Nansha Bay, China. However, the IMTA system is a complex ecological system containing many environmental parameters. The optimal proportion of different trophic species in the IMTA system depends on numerous variables that show fluctuations during the culturing period in every mode in this study. Based on our results with respect to the variation of DO, carbonate system and pCO2, the optimal proportion of oyster and kelp was below 24꞉1. pH continued to increase over the time, indicating that the appropriate oyster-kelp combination could reduce CO2 concentration and create a carbon sink to resist ocean acidification. However, as the chl-a concentration changes, keeping an oyster꞉kelp ratio of 24꞉1–24꞉3 would induce a phytoplankton bloom in the system.
Chl-a content represents phytoplankton biomass, and phytoplankton is the main food for suspension feeders like shellfish (Sarà and Mazzola, 2004; Rueda et al., 2005; Liu et al., 2011). In our study, chl-a concentration significantly increased in the 24꞉1, 24꞉2, and 24꞉3 modes. It also increased in other modes by the end of the experiment, excepting for the 8꞉1 mode. Furthermore, the chl-a concentration increased with increasing oyster biomass. The conflicts with the concept that oysters feed on the phytoplankton and occurs because the growth condition for the smaller phytoplankton improved when the oysters (C. gigas) feed on larger phytoplankton (> 5 μm) as opposed to smaller ones (< 5 μm). Dupuy et al. (2000) found that the percentage of picoeukaryotes retained by filtering oysters (C. gigas) was less than 2%, which means that oyster filtered the larger phytoplankton leaving the smaller cells in the column. Correspondingly, nutrients produced from the oysters could increase the phytoplankton growth. Lu et al. (2015) found that the biomass of Synechococcus (< 2 μm) increased significantly after 36 h of culturing with scallop (Chlamys farreri) in transparent polyethylene bags. P-enrichment released by scallops significantly stimulated the picoplankton growth (< 2 μm) by providing a food source for nanoflagellates as the < 5 μm treatment also excludes the predators of 2–5 μm nanoflagellates. Thus, it is indicated that the increase in chl -a content was due to picophytoplankton growth in the system. Therefore, in our study, nutrients released by oysters were enough for kelp absorption in different modes except for the 8꞉1 mode. In order to simultaneously avoid eutrophication and food shortage for shellfish, the chl-a concentration should be kept at a stable level. Thus, the optimal proportion of oyster and kelp for IMTA is 8꞉2–8꞉3 at the beginning of winter in Sanggou Bay.
5 CONCLUSIONIn conclusion, the IMTA system is an important framework for aquaculture sustainability, as it mitigates the negative effects from fed aquaculture species and organic extractive species. Seaweeds, as an inorganic extractive species, play an important role in the IMTA system. More seaweeds in the IMTA system result in more nutrients and carbon source being removed from water column. The proportion of different trophic species in the IMTA system is difficult to determine due to the fluctuations of different environmental parameters. According to the previous references and results of this study, the optimal proportion of oyster and kelp is 8꞉2–8꞉3 at the beginning of winter in Sanggou Bay, which will aid government in optimally adjusting the Sanggou Bay aquaculture structure.
6 DATA AVAILABILITY STATEMENTThe datasets during and/or analyzed during the current study available from the corresponding author on reasonable request.
Abreu M H, Varela D A, Henríquez L, Villarroel A, Yarish C, Sousa-Pinto I, Buschmann A H. 2009. Traditional vs. integrated multi-trophic aquaculture of Gracilaria chilensis C. J. Bird, J. McLachlan & E. C. Oliveira:productivity and physiological performance. Aquaculture, 293(3-4): 211-220.
DOI:10.1016/j.aquaculture.2009.03.043 |
Ahn O, Petrell R J, Harrison P J. 1998. Ammonium and nitrate uptake by Laminaria saccharina and Nereocystis luetkeana originating from a salmon sea cage farm. J. Appl. Phycol., 10(4): 333-340.
DOI:10.1023/A:1008092521651 |
Briggs M R P, Fvnge-Smith S. 1994. A nutrient budget of some intensive marine shrimp ponds in Thailand. Aquac. Res., 25(8): 789-811.
DOI:10.1111/j.1365-2109.1994.tb00744.x |
Chang J, Tian X L, Dong S L, Wang D P, Bao J, Ma S, Sun Y C, Sun J. 2006. An experimental study on nitrogen and phosphorus budgets in polyculture of shrimp, bivalve and seaweed. Period. Ocean Univ. China, 36(S1): 33-39.
(in Chinese with English abstract) DOI:10.3969/j.issn.1672-5174.2006.z1.006 |
Chopin T, Robinson S M C, Troell M, Neori A, Buschmann A H, Fang J. 2008. Multitrophic integration for sustainable marine aquaculture. In: Jørgensen S E, Fath B D eds. Encyclopedia of Ecology. Elsevier, Amsterdam. p.2 463-2 475, https://doi.org/10.1016/B978-008045405-4.00065-3.
|
Chopin T, Troell M, Reid G K, Knowler D, Robinson S M C, Neori A, Buschmann A H, Pang S J. 2010. Integrated multi-trophic aquaculture. Part Ⅰ. Responsible practice provides diversified products, biomitigation. Global Aquac. Advocate, 5: 38-39.
|
Dickson A G, Goyet C. 1994. Handbook of Methods for the Analysis of the Various Parameters of the Carbon Dioxide System in Sea Water, version 2. ORNL/CDIAC-74 US Department of Energy, Washington, DC. p.4-26.
|
Dupuy C, Vaquer A, Lam-Höai T, Rougier C, Mazouni N, Lautier J, Collos Y, Le Gall S. 2000. Feeding rate of the oyster Crassostrea gigas in a natural planktonic community of the Mediterranean Thau Lagoon. Mar. Ecol. Prog. Ser., 205: 171-184.
DOI:10.3354/meps205171 |
Fang J G, Zhang J, Xiao T, Huang D J, Liu S M. 2016. Integrated multi-trophic aquaculture (IMTA) in Sanggou Bay, China. Aquac. Environ. Interact., 8: 201-205.
DOI:10.3354/aei00179 |
Fang J H, Zhang J H, Wu W G, Mao Y Z, Jiang Z J, Fang J G. 2014. Carbon and nitrogen budget and environmental optimization in an integrated cage culture model of Japanese flounder with Perinereis aibuhitensis. J. Fish. Sci. China, 21(2): 390-397.
(in Chinese with English abstract) |
FAO. 2016. The State of World Fisheries and Aquaculture 2016 (SOFIA): Contributing to Food Security and Nutrition for All. The State of World Fisheries and Aquaculture, Rome.
|
Gillibrand P A, Turrell W R, Moore D C, Adams R D. 1996. Bottom water stagnation and oxygen depletion in a Scottish sea loch. Estuar., Coast. Shelf. Sci., 43(2): 217-235.
DOI:10.1006/ecss.1996.0066 |
Han T, Jiang Z, Fang J, Zhang J, Mao Y, Zou J, Huang Y, Wang D. 2013. Carbon dioxide fixation by the seaweed Gracilaria lemaneiformis in integrated multi-trophic aquaculture with the scallop Chlamys farreri in Sanggou Bay, China. Aquacult Int., 21(5): 1 035-1 043.
DOI:10.1007/s10499-012-9610-9 |
Inui M, Itsubo M, Iso S. 1991. Creation of a new nonfeeding aquaculture system in enclosed coastal seas. Mar. Pollut. Bull., 23: 321-325.
DOI:10.1016/0025-326X(91)90694-N |
Jiang Z J, Fang J G, Mao Y Z, Wang W. 2010. Eutrophication assessment and bioremediation strategy in a marine fish cage culture area in Nansha Bay, China. J. Appl. Phycol., 22(4): 421-426.
DOI:10.1007/s10811-009-9474-1 |
Jiang Z J, Wang G H, Fang J G, Mao Y Z. 2013. Growth and food sources of Pacific oyster Crassostrea gigas integrated culture with Sea bass Lateolabrax japonicus in Ailian Bay, China. Aquac. Int., 21(1): 45-52.
DOI:10.1007/s10499-012-9531-7 |
Langan R. 2004. Balancing marine aquaculture inputs and extraction: combined culture of finfish and bivalve molluscs in the open ocean. Bull. Fish. Res. Agen., (S1): 51-58.
|
Lartigue J, Sherman T D. 2005. Response of Enteromorpha sp.(Chlorophyceae) to a nitrate pulse:nitrate uptake, inorganic nitrogen storage and nitrate reductase activity. Mar. Ecol. Prog. Ser., 292: 147-157.
DOI:10.3354/meps292147 |
Liu Z L, Chen J F, Liu Y L, Gao S Q, Li H L, Zhang H S. 2011. The size-fractionated chlorophyll a concentration and primary productivity in the Bering Sea in the summer of 2008. Acta Oceanol. Sin., 33(3): 148-157.
(in Chinese with English abstract) |
Lu J C, Huang L F, Xiao T, Jiang Z J, Zhang W C. 2015. The effects of Zhikong scallop (Chlamys farreri) on the microbial food web in a phosphorus-deficient mariculture system in Sanggou Bay, China. Aquaculture, 448: 341-349.
DOI:10.1016/j.aquaculture.2015.06.021 |
Mao Y Z, Yang H S, Zhou Y, Hu Z F, Yuan X T, You K, Wang R C. 2006. Studies on growth and photosynthesis characteristics of Gracilaria lemaneiformis and its capacity to uptake ammonium and phosphorus from scallop excretion. Acta Ecol. Sin., 26(10): 3 225-3 231.
(in Chinese with English abstract) |
Mao Y Z, Yang H S, Zhou Y, Ye N H, Fang J G. 2009. Potential of the seaweed Gracilaria lemaneiformis for integrated multi-trophic aquaculture with scallop Chlamys farreri in North China. J. Appl. Phycol., 21(6): 649-656.
DOI:10.1007/s10811-008-9398-1 |
Mazzola A, Sarà G. 2001. The effect of fish farming organic waste on food availability for bivalve molluscs (Gaeta Gulf, central Tyrrhenian, MED):stable carbon isotopic analysis. Aquaculture, 192(2-4): 361-379.
DOI:10.1016/S0044-8486(00)00463-4 |
Ning Z M, Liu S M, Zhang G L, Ning X Y, Li R H, Jiang Z J, Fang J G, Zhang J. 2016. Impacts of an integrated multitrophic aquaculture system on benthic nutrient fluxes:a case study in Sanggou Bay, China. Aquac. Environ. Interact., 8: 221-232.
DOI:10.3354/aei00144 |
Pedersen A, Kraemer G, Yarish C. 2004. The effects of temperature and nutrient concentrations on nitrate and phosphate uptake in different species of Porphyra from Long Island Sound (USA). J. Exp. Mar. Biol. Ecol., 312(2): 235-252.
DOI:10.1016/j.jembe.2004.05.021 |
Phillips J C, Hurd C L. 2003. Nitrogen ecophysiology of intertidal seaweeds from New Zealand:N uptake, storage and utilisation in relation to shore position and season. Mar. Ecol. Prog. Ser., 264: 31-48.
DOI:10.3354/meps264031 |
Rueda J L, Smaal A C, Scholten H. 2005. A growth model of the cockle (Cerastoderma edule L.) tested in the Oosterschelde estuary (The Netherlands). J. Sea Res., 54(4): 276-298.
DOI:10.1016/j.seares.2005.06.001 |
Sarà G, Mazzola A. 2004. The carrying capacity for Mediterranean bivalve suspension feeders:evidence from analysis of food availability and hydrodynamics and their integration into a local model. Ecol. Modell., 179(3): 281-296.
DOI:10.1016/j.ecolmodel.2004.03.005 |
Stenton-Dozey J. 2007. Finding hidden treasure in aquaculture waste. Water Atmos, 15(4): 10-11.
|
Tang Q S, Fang J G, Zhang J H, Jiang Z J, Liu H M. 2013. Impacts of multiple stressors on coastal ocean ecosystems and integrated multi-trophic aquaculture. Prog. Fish. Sci., 34(1): 1-11.
(in Chinese with English abstract) DOI:10.3969/j.issn.1000-7075.2013.01.001 |
Tang Q S, Zhang J H, Fang J G. 2011. Shellfish and seaweed mariculture increase atmospheric CO2 absorption by coastal ecosystems. Mar. Ecol. Prog. Ser., 424: 97-104.
DOI:10.3354/meps08979 |
Troell M, Joyce A, Chopin T, Neori A, Buschmann A H, Fang J G. 2009. Ecological engineering in aquaculture-potential for integrated multi-trophic aquaculture (IMTA) in marine offshore systems. Aquaculture, 297(1-4): 1-9.
DOI:10.1016/j.aquaculture.2009.09.010 |
Wen Y M, Wei X G, Shu T F, Zhou J F, Yu G H, Li F, Huang Y Y. 2007. Forms and balance of nitrogen and phosphorus in cage culture waters in Guangdong Province, China. Chin. Geogr. Sci., 17(4): 370-375.
DOI:10.1007/s11769-007-0370-9 |
Wu R S S. 1995. The environmental impact of marine fish culture:towards a sustainable future. Mar. Pollut. Bull., 31(4-12): 159-166.
DOI:10.1016/0025-326X(95)00100-2 |
Zhang Y. 2012. Comparison of culture effect, discharge of nitrogen and phosphorus and environmental influence for three kinds of cages. Huazhong Agriculture University, Wuhan. (in Chinese with English abstract)
|
Zhang Z H, Lü J B, Ye S F, Zhu M Y. 2007. Values of marine ecosystem services in Sanggou Bay. Chin. J. Appl. Ecol., 18(11): 2 540-2 547.
(in Chinese with English abstract) |
 2020, Vol. 38
2020, Vol. 38